ATOMS Atoms are the building blocks of all

ATOMS Atoms are the building blocks of all matter. We know the atom is made up of smaller bits called… Name Protons Charge +ve Electrons Neutrons Position 1 Outside neutral All atoms have a defined number of protons. Mass Nucleus 0
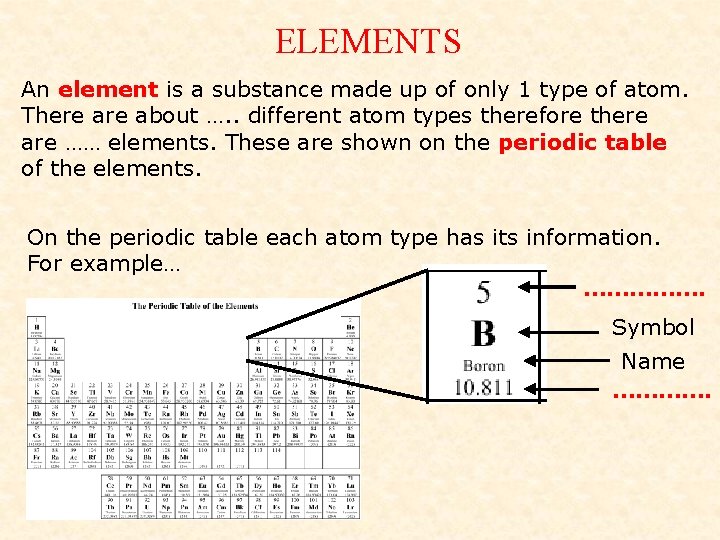
ELEMENTS An element is a substance made up of only 1 type of atom. There about …. . different atom types therefore there are …… elements. These are shown on the periodic table of the elements. On the periodic table each atom type has its information. For example… ……………. Symbol Name ………….
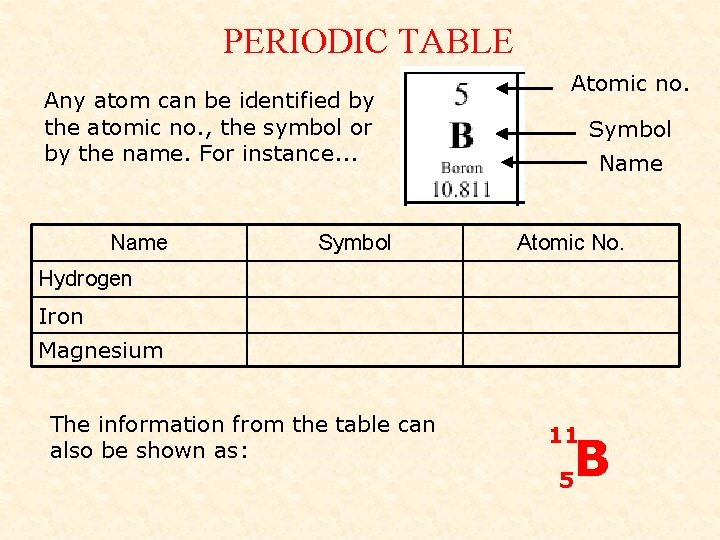
PERIODIC TABLE Any atom can be identified by the atomic no. , the symbol or by the name. For instance. . . Name Symbol Atomic no. Symbol Name Atomic No. Hydrogen Iron Magnesium The information from the table can also be shown as: 11 B 5

WHAT IT MEANS The Atomic Number: 11 B = number of p…………. 5 = number of e…………. (as an atom has the same of each) The Mass Number: = number of p………. + n………. - why are electrons not included in the mass no? They have no mass! So for Boron… What about Phosphorus? Protons = 5 Protons = Electrons = 5 Electrons = 15 Neutrons = 5. 811 Neutrons = 16 15

Learning Check Select the correct symbol for each: A. Calcium 1) C 2) Ca 3) CA B. Sulfur 1) S 2) Sl 3) Su C. Iron 1) Ir 2) FE 3) Fe

Learning Check Select the correct name for each: A. N 1)neon 2)nitrogen 3)nickel B. P 1)potassium 2)phlogiston 3)phosphorus C. Ag 1) silver 2)agean 3)gold

Isotopes Atoms of Elements can occur with different mass numbers. E. g. Carbon • Carbon can have 6, 7 and 8 …………… • Carbon has … isotopes

Test Yourself • Explain why 12 C and 13 C are neutral and consider the atomic structure to discuss how the mass of each atom differs. 12 C 13 C neutral 6+ and 6 - protons 6+ 6+ neutrons 6 no charge 7 no charge electrons 6 - 6 - mass 6 protons +6 neutrons 6 protons +7 neutrons

ELECTRON ARRANGEMENT Electrons are very fast moving. They are arranged in shells around the nucleus. The first shell fits… 2 e The second fits… 8 e So the electron shell for The third fits… 8 e 12 Mg would be… 2, 8, 2 What is the electron configuration of Li (2, 1) Ca (2, 8, 8, 2) P (2, 8, 5) What element is represented by 2, 8, 7 (Cl) 2, 6 (O) 2, 8, 1 (Na)

GROUPS of the Periodic Table The columns in the periodic table are called g…. . . Groups of elements share similar reactivity. This is because they have the same number of valence electrons. There are effectively 8 groups going from left to right As you go down a group the reactivity increases. Group 1 are the alkali metals properties: good conductors solid at room temperatures can be cut with a knife low densities and melting points Group 17 are the Halogens

Periods • The rows in the periodic table are called p……………. • Atom’s size decreases from left to right in a given period.

Learning Check A. Element in Group XVII, Period 4 1) Br 2) Cl 3) Mn B. Element in Group II, Period 3 1) beryllium 2) magnesium 3) boron

IONS An ion is an atom that has ……… Or …………. electrons meaning it is negatively or positively charged. Atoms do this to get a ……. outer (valence) electron shell and so becomes more ………… (less reactive). The atom will get a full outer shell the simplest way it can, e. g. for 12 Mg: - Electron arrangement of 2, 8, 2 - it will LOSE 2 electrons (become 2, 8) - Now it has 10 electrons, but still has 12 protons. It has a 2+ charge. The ion is called Mg 2+. Superscript is used for ion charges

Cations are ions that have lost electrons to become …………… ions. E. g. Na+, Mg 2+, Al 3+ Na has lost 1 electron so it is said to have a +1 charge or it has a valency of +1. Valency is a measure of its charge it has gained or lost. It is often referred to as the ………. . …………. . … ………. Metals have valencys of +1, +2 or +3. That is they lose 1 electron 2 electrons or 3 Electrons

So Why? The outer shell of an atom is called the …………. . shell (note the spelling). The outer shell can have 1, 2 , 3, 4, 5 , 6 , 7 or 8 electrons. When the outer shell has 8 electrons the atom is ……………. . and very stable. These atoms don’t need to lose or gain electrons. They are chemically very stable and thus wont react with other atoms. On the periodic table the elements that are stable are called the ………… or inert gases. They are found on the far right column of the periodic table. Group 8 All other elements can react with other elements.

Metals have 1 , 2 or 3 electrons in the outer shell. The atom wants to be stable so they will lose the electrons to become stable. They have to give them to other atoms who need electrons. These atoms are the …-………. . .

Anions The metal atom electrons go to the non-metal atoms. The gain of electrons to the non-metal atom changes the charge and makes it ………………. The negatively charged non-metal atom is called an ………………. . . E. g. Cl-, O 2 -, N 3 Each of the above examples has gained 1 , 2 and 3 electrons respectively. Or they have valencys of …. , ……, and ………

What About Carbon? Some elements have 4 electrons in their outer shell. They could gain 4 electrons or lose 4 electrons. …………. . is an example. Usually they tend to gain electrons. That is they are. . -………. . .

Could Sodium gain 7 electrons rather than lose 1 electron? …. . . It is much easier for metals to lose electrons and it is much easier for nonmetals to gain electrons. As a result metals and non-metals react to form ionic compounds. It’s a bit like a marriage.

IONIC COMPOUNDS The ions that have been formed are now electrically attracted to …………. . charged ions. So Mg 2+ will be attracted to Cl-. This forms an ionic ………………. . Naming rules: - The positive ion is first, and the negative second. - The negative ion ends in –ide, e. g. The sulfur atom becomes the sulfide ion. Exceptions are complex anions that are madeof 2 or 3 types of atoms: NO 3 - (nitrate) SO 42 - (sulfate) CO 32 - (carbonate) HCO 3 - (hydrogen carbonate)
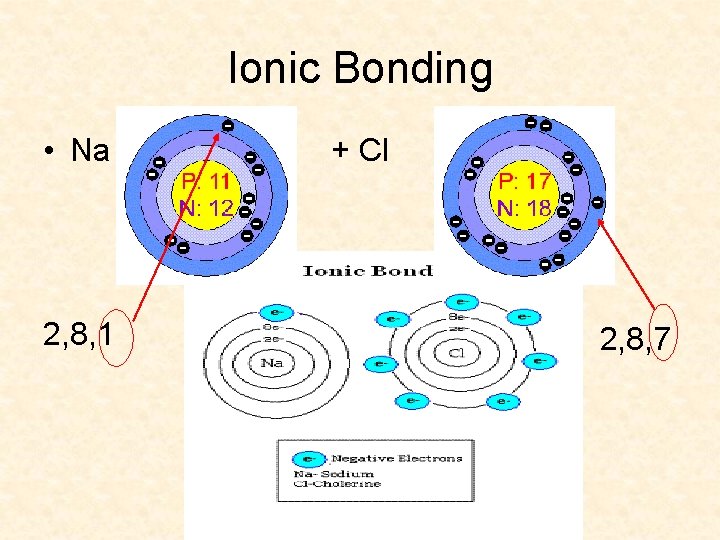
Ionic Bonding • Na 2, 8, 1 + Cl 2, 8, 7

IONIC FORMULAE So Mg 2+ will be attracted to Cl-. Because Mg is 2+ and Cl is only 1 -, Mg will attract 2 Cl’s. The compound formed will be Mg. Cl 2. The subscript shows that the are 2 Cl’s for each Mg. If the starting ions were Cu 2+ and S 2 -, the 2 ions have the same charge. So each Cu will only attract 1 S. The compound formed will be Cu. S. There is never any charges on the final product they balance out

DIFFICULT ONES What is the formula for Magnesium Nitrate? Find the 2 ions on your table of ions… Mg 2+ and NO 3 - This means that there are 3 oxygens attached to the Nitrogen – don’t let them get lost! So for each Mg we will need 2 NO 3’s: The shorthand way NO 3 of writing this is: Mg NO 3 Mg(NO ) 3 2 NH 4+ and S 2 - = (NH 4)2 S Fe 3+ and OH- = Fe(OH)3 Al 3+ and SO 42 - = Al 2(SO 4)3 The brackets are needed to show that we want 2 of the whole thing – you always need them if you have 2 of an ion with more than 1 bit. (polyatomic ions)

Physical Properties The characteristics of a substance that can be observed without changing the substance. n Conductivity (thermal/electrical) n Color n Malleability n Lustre n Density n Freezing and Boiling Points n Odour

Metals and Nonmetals Metals n Located to the left of the heavy line n Shiny, ductile n Good conductors of heat and electricity Nonmetals n Located to the right of the heavy line n Dull and brittle n Poor conductors, good insulators

Metal reactions General Equation • Metal + Oxygen → Metal oxide Word Equation • Aluminium + Oxygen → Formula Equation Aluminium oxide • 4 Al + 3 O 2 → 2 Al 2 O 3 • Metal + Water → Metal hydroxide + Hydrogen • Aluminium + Water → Aluminium Hydroxide + Hydrogen • 2 Al + 6 H 2 O → 2 Al(OH)3 + 3 H 2 • Metal + Acid → Metal salt + Hydrogen • Magnesium + Hydrochloric acid → Magnesium Chloride + Hydrogen • Mg + 2 HCl → Mg. Cl 2 + H 2
- Slides: 26