Atoms Atoms are the basic unit of matter

Atoms � � � Atoms are the basic unit of matter Atoms are composed of protons, neutrons and electrons These subatomic particles are charged….

� � Atoms make elements… Elements can’t be broken down into anything simpler. Examples: � Carbon= C � Sodium = Na � Hydrogen= H � Nitrogen= N

Subatomic particles have specific locations… • Protons and the neutrons are found in the nucleus of the atom and electrons whizz around the outside of the nucleus • Electrons are always moving. • The atomic shell or orbital is the distance from the nucleus that the electron spins.

Valence shells… • The outermost shell is called the valence shell. • To be “happy” and full, the valence shell must have 8 electrons. • The electrons in the outermost shell are the only ones that react with other atoms. If the valence shell is full, the atom does not react. • Carbon only has four valence electrons (8 is the max) so carbon can bond with up to four other atoms.

Ions � � � A normal atom is neutral (same number of electrons and protons) Ions have a different number of electrons than the neutral atom Sodium has only one valence electron… it would like to give that up so it can have a full outer shell. Chloride needs one electron to have a full outer shell. Sodium loses an electron and becomes Na+ Chloride becomes negative when it gains an electron… Cl-

Bonding= trying to be a happy, FULL atom Some atoms can easily lose electron s to be full Some atoms can easily gain electrons to become full
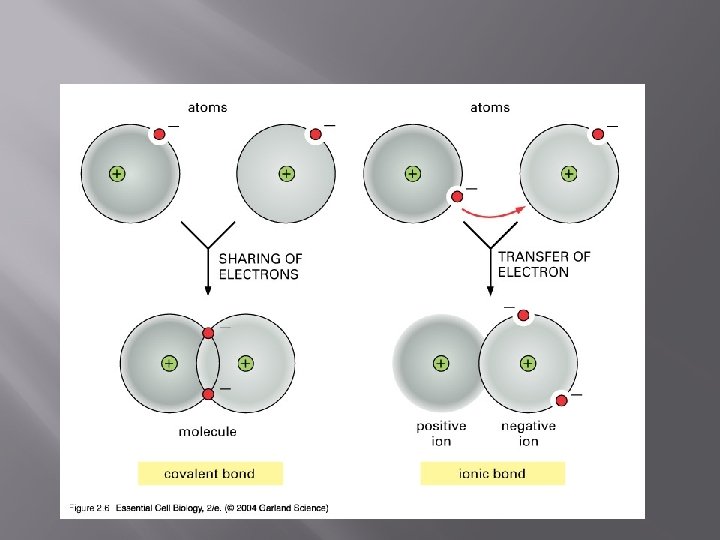

Ionic bonds � � � A good example of an ionic bond is Na. Cl. Sodium really wants to lose an electron and becomes a positive ion (cation). Chloride really wants to gain an electron (high electronegativity) and becomes a negative ion (anion).
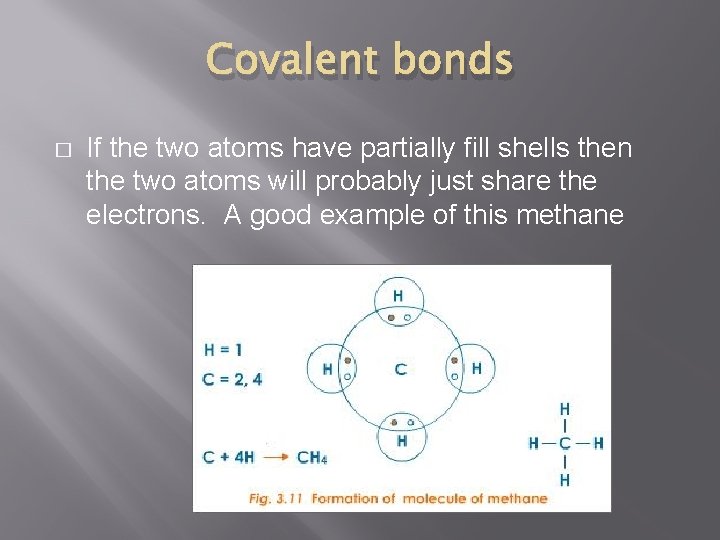
Covalent bonds � If the two atoms have partially fill shells then the two atoms will probably just share the electrons. A good example of this methane
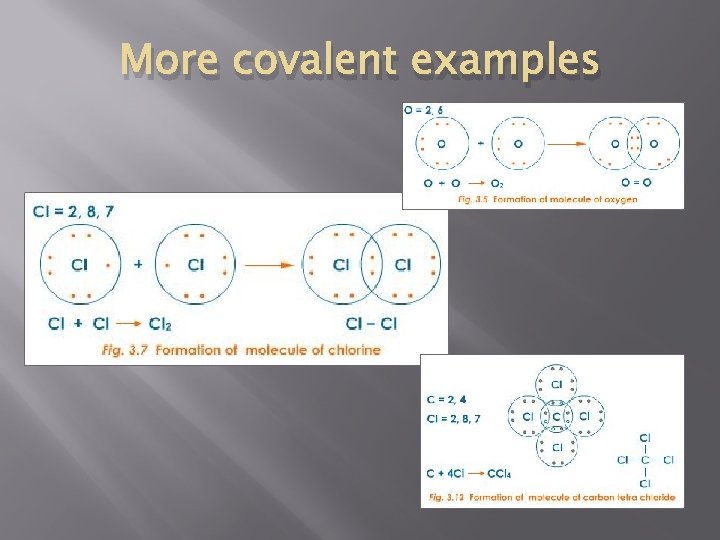
More covalent examples

Polar Covalent Bonds � Polar Covalent means that two atoms share electrons but the electrons may spend a majority of their time with one particular atom. (joint custody) � An example: WATER
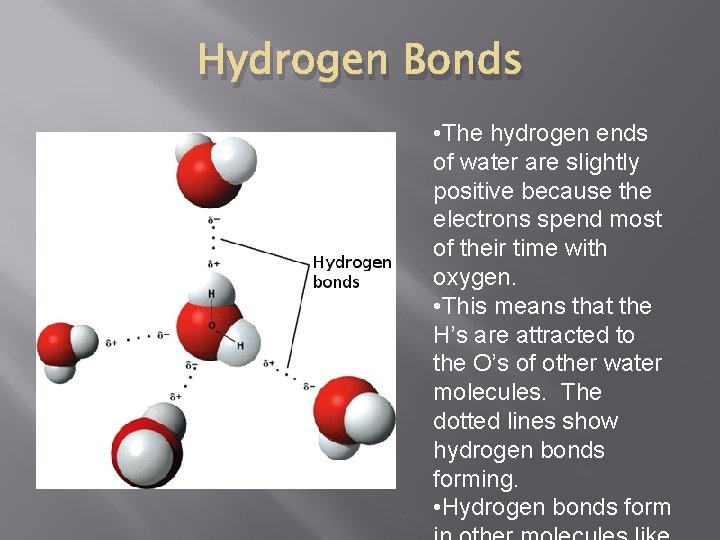
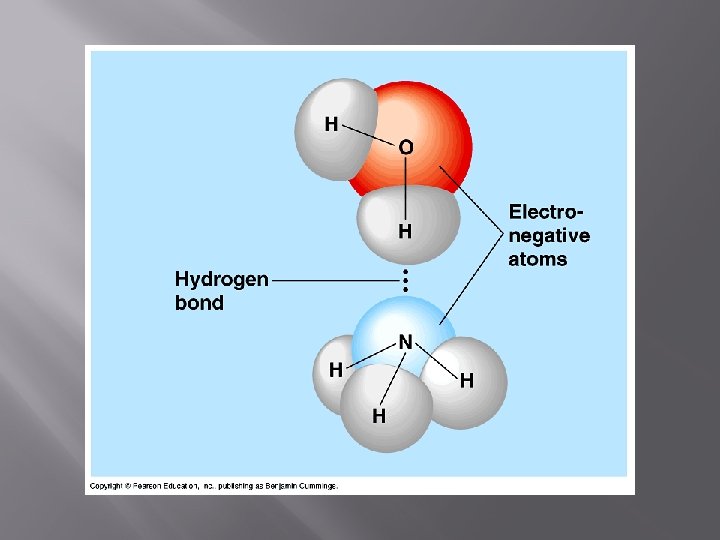
Hydrogen Bonds • The hydrogen ends of water are slightly positive because the electrons spend most of their time with oxygen. • This means that the H’s are attracted to the O’s of other water molecules. The dotted lines show hydrogen bonds forming. • Hydrogen bonds form
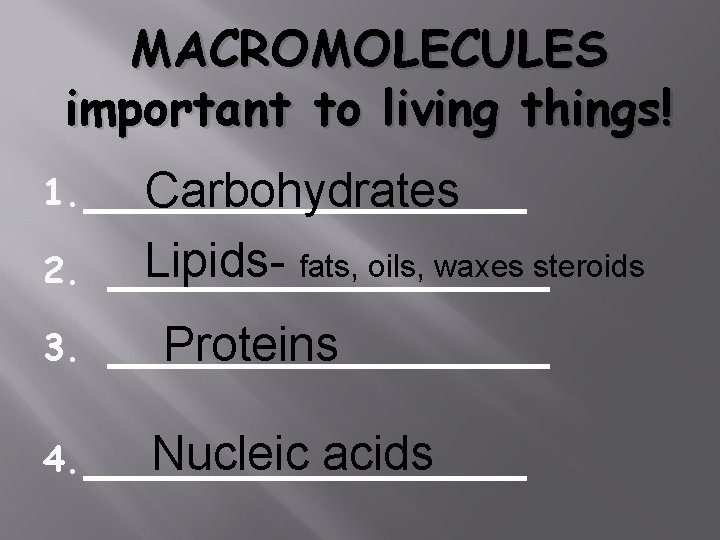
MACROMOLECULES important to living things! 1. _______ Carbohydrates 2. 3. Lipids- fats, oils, waxes steroids _____________ Proteins Nucleic acids 4. _______

Introductory Terms � � Macromolecule = “giant” molecule of living matter Monomer = subunits that are the building blocks of a polymer � Example: � Lego block Polymer = monomers linked together � Example: Lego castle

Macromolecular Reactions � Dehydration Synthesis: 2 monomers become bonded to each other to make a polymer � Lose water � � Hydrolysis: Water is used to break up polymers into monomers � Using water to break bonds �

Carbohydrates � Structure: � C: H: O in a 1: 2: 1 Ratio � Example: C 6 H 12 O 6 which is glucose � Functions: � Short-term energy-Pasta dinner before the big race, game, etc. � Structural Support

Lipids � Structure: � Made up of carbon, hydrogen, and a few oxygens � Do not dissolve in water � Functions: � Long-term energy storage � Chemical messengers (hormones) � Insulation around your nerve cells and to keep you warm! � Waxes (plant leaves to prevent water loss)

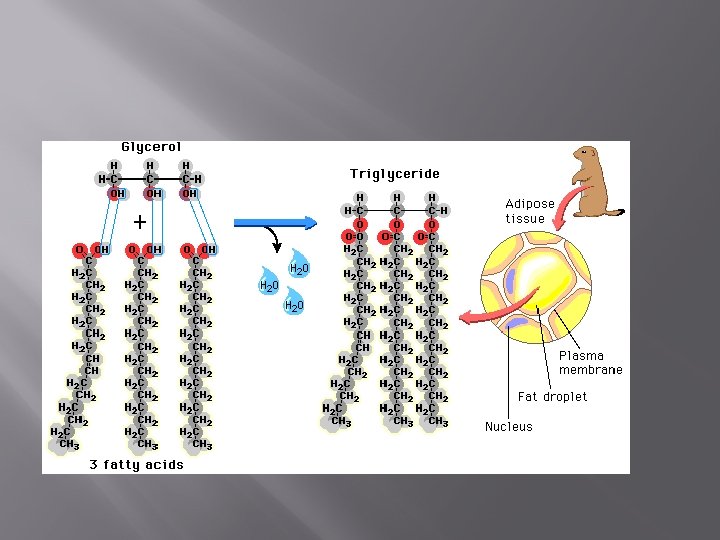
Lipids-Triglycerides � Fats � Made of glycerol and 3 fatty acids tails � Connect via dehydration synthesis to make triglycerides � Triglycerides are fat molecules… clogging arteries…

__________ PHOSPHOLIPIDS (lipid tails + glycerol/phosphate head) Polar head Non-polar tails
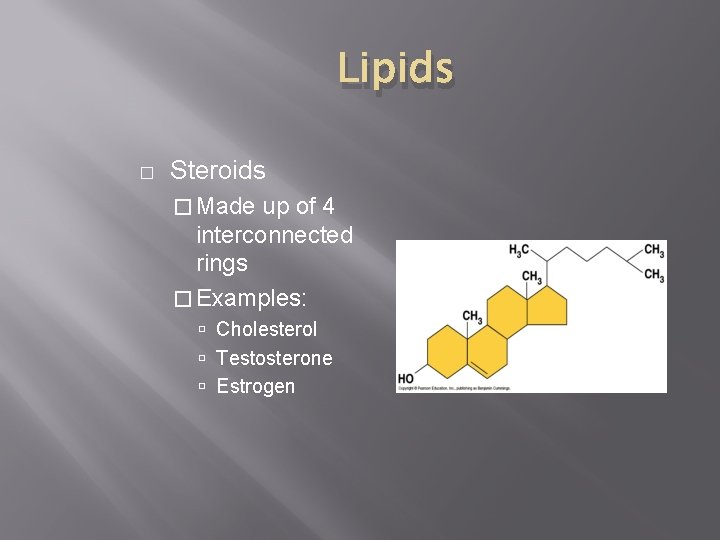
Lipids � Steroids � Made up of 4 interconnected rings � Examples: Cholesterol Testosterone Estrogen

Proteins � � � Most of a cell is made up of proteins Instrumental in almost everything organisms do Structure: � All proteins are created from unique combinations of 20 different amino acids � C, H, O, N (S) � Major Functions: � Structure � Defense (antibodies) � Enzymes- we will discuss these later!

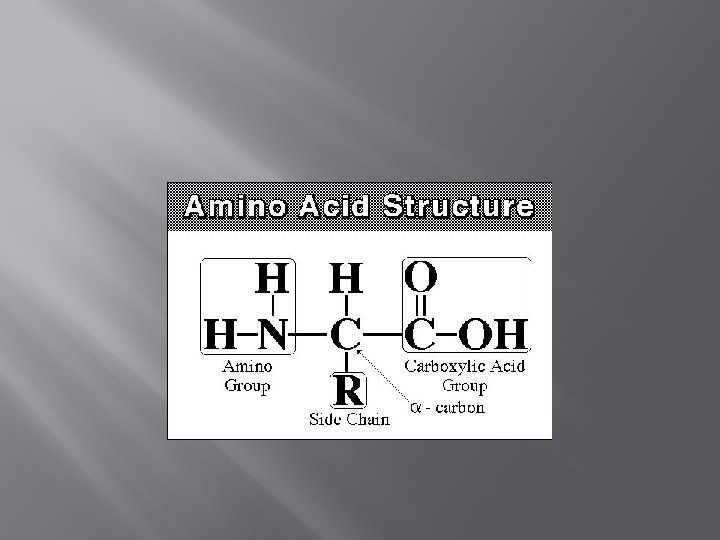
Proteins � Amino Acids � Amino acids are the monomers of proteins � Organic molecules possessing both carboxyl and amino groups ( get it AMINO…. ACID) � 20 types of AAs that make up 1000 s of different proteins � AAs are linked together by peptide bonds to make polypeptides
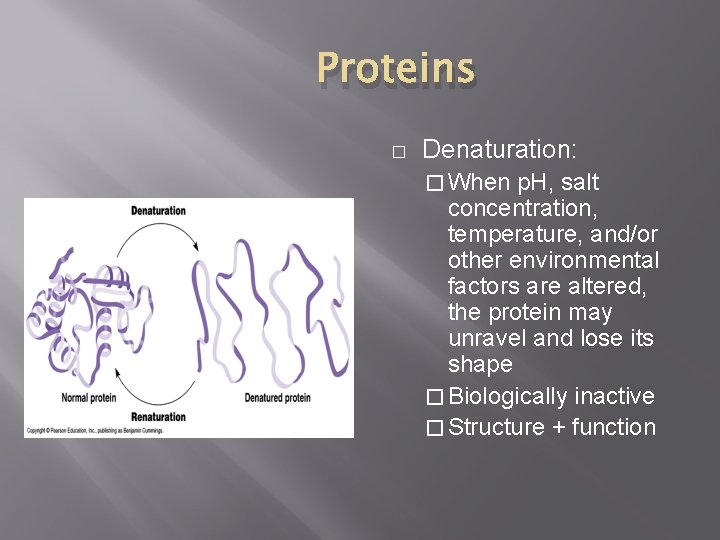
Proteins � Denaturation: � When p. H, salt concentration, temperature, and/or other environmental factors are altered, the protein may unravel and lose its shape � Biologically inactive � Structure + function
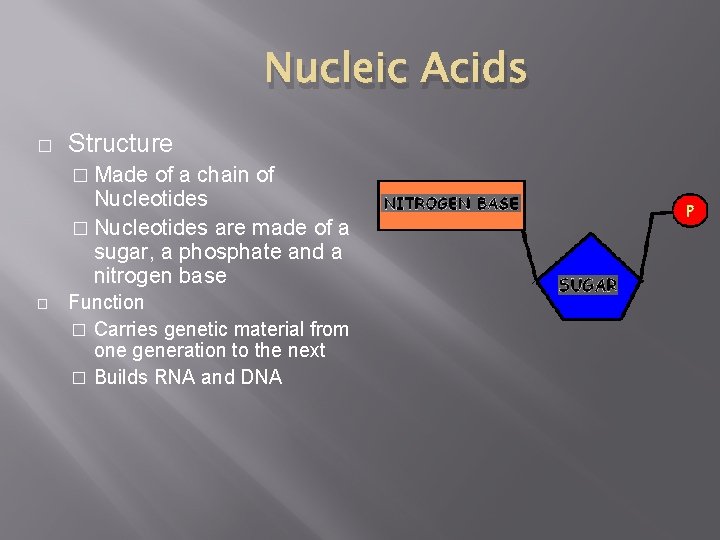
Nucleic Acids � Structure � Made of a chain of Nucleotides � Nucleotides are made of a sugar, a phosphate and a nitrogen base � Function � Carries genetic material from one generation to the next � Builds RNA and DNA
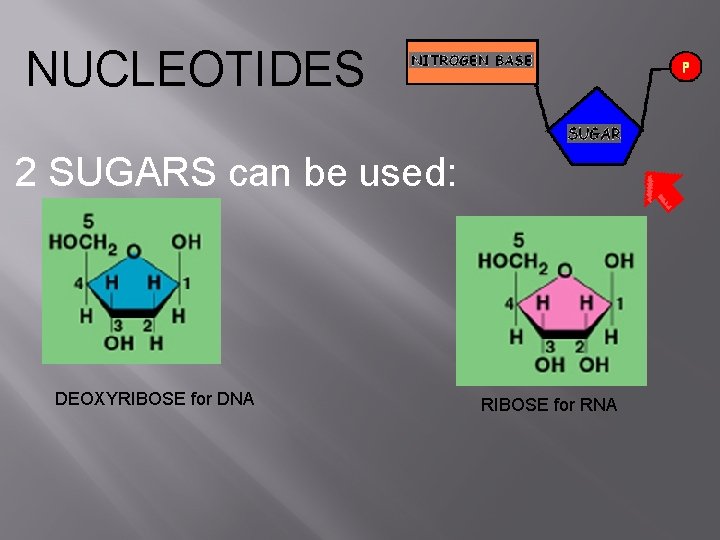
NUCLEOTIDES 2 SUGARS can be used: DEOXYRIBOSE for DNA RIBOSE for RNA
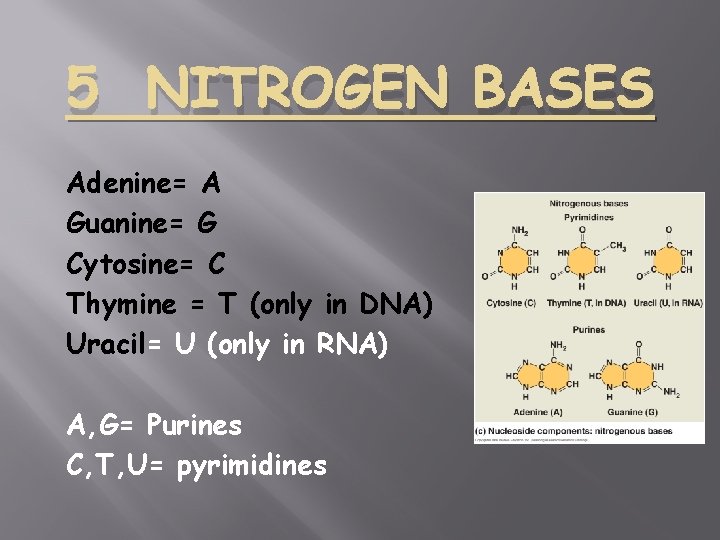
5 NITROGEN BASES Adenine= A Guanine= G Cytosine= C Thymine = T (only in DNA) Uracil= U (only in RNA) A, G= Purines C, T, U= pyrimidines

RNA vs. DNA � RNA � Sugar is ribose � Contains Uracil not Thymine � Single stranded � DNA � Sugar is Deoxyribose � Contains A, T, C, G � Double stranded
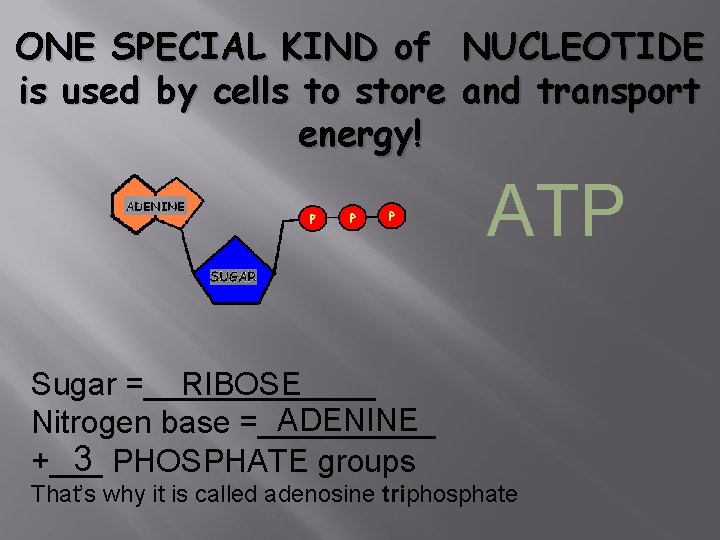
ONE SPECIAL KIND of is used by cells to store energy! NUCLEOTIDE and transport ATP Sugar =_______ RIBOSE ADENINE Nitrogen base =_____ 3 PHOSPHATE groups +___ That’s why it is called adenosine triphosphate
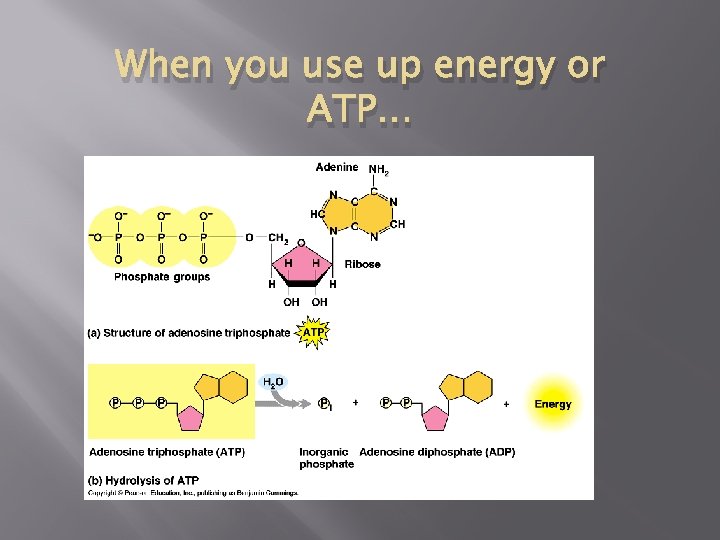
When you use up energy or ATP…

ATP ADP Cycle � � � So, ATP has three phosphates but energy is released when ATP turns to ADP by losing a phosphate Hydrolysis Cycles back and forth

Cohesion � � Water sticks to water (hydrogen bonds) Helps plants transport water through vessels

Adhesion � � Water sticks to other molecules (like the side of a glass) Also helps keep water moving up against gravity in plants.

Surface Tension � � Related to cohesion Molecules resist being pulled apart due to strong hydrogen bonds… form a “film”

Density Why can’t you fill a water bottle completely when you want to freeze it? � � What occurs to the structure of water when it freezes? Insulation for life below the surface…

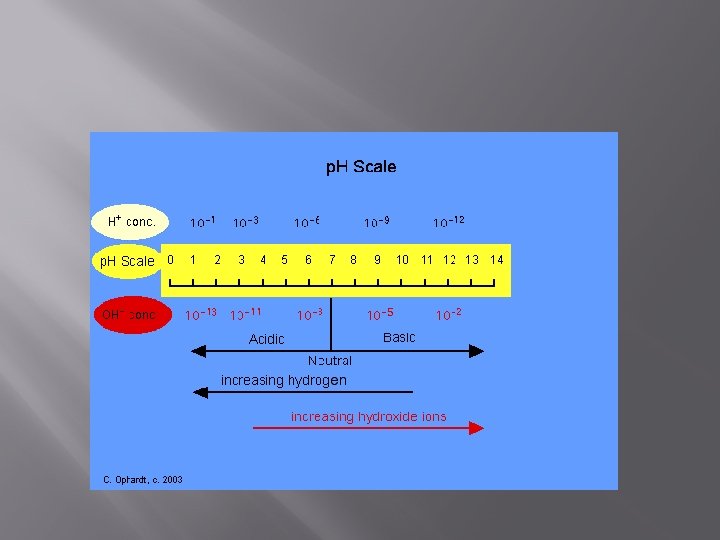
p. H Scale- ever heard of it? � � � An acid is a compound that releases protons (H+) when dissolved in water A base is a compound that adds OH- when dissolves in water. We measure these reactions on the p. H scale

The p. H Scale- ranges from 0 -14 � p. H of 1 means lots of H+ ions and SUPER ACIDIC � p. H of 14 means lots of OH- (or low H+) and SUPER BASIC � Water is neutral with a p. H of 7
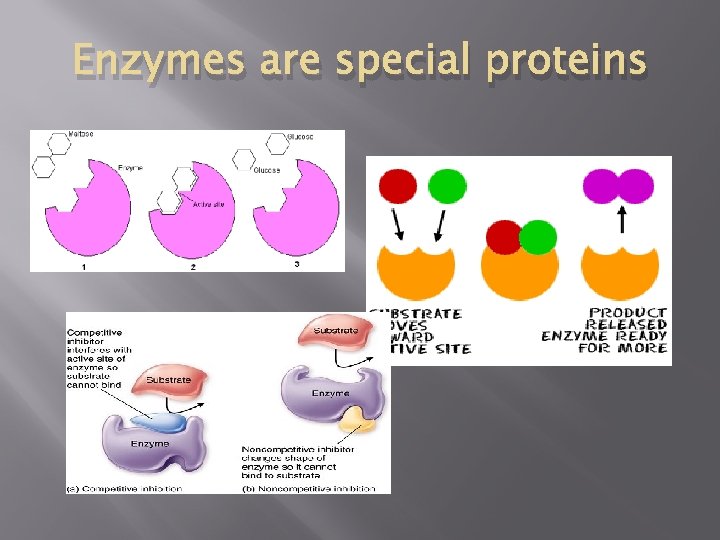
Enzymes are special proteins
- Slides: 42