Atoms and the Periodic Table The Periodic Table

Atoms and the Periodic Table • The Periodic Table

Dmitri Mendeleev • Dmitri Mendeleev (1869, Russian) –Organized elements by increasing atomic mass. One proton and electron are added to each element as you go across the table. Predicted the existence of undiscovered elements.

Henry Mosely • Henry Mosely (1913, British) –Organized elements by increasing atomic number. –Fixed problems in Mendeleev’s arrangement.
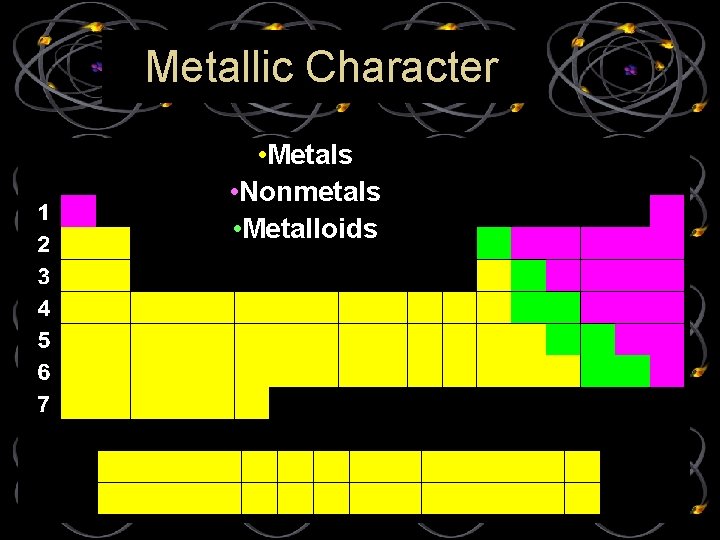
Metallic Character • Metals • Nonmetals • Metalloids

Metallic Character • Metals- good conductors of heat and electricity, Malleable (most metals can be hammered into thin sheets) • Nonmetals- gases that are poor conductors of heat and electricity at room temperature, Brittle (breaks easily) • Metalloids- Conduct heat and electricity better than nonmetals but not as well as metals, Can be shiny or dull , Solids
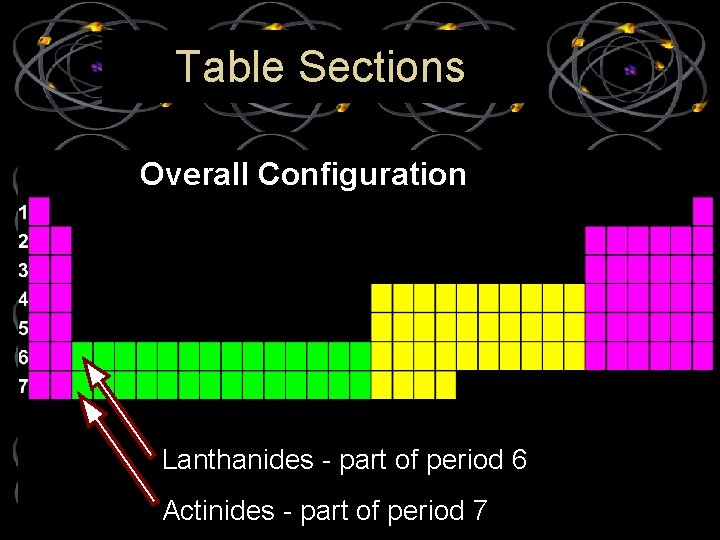
Table Sections Overall Configuration Lanthanides - part of period 6 Actinides - part of period 7
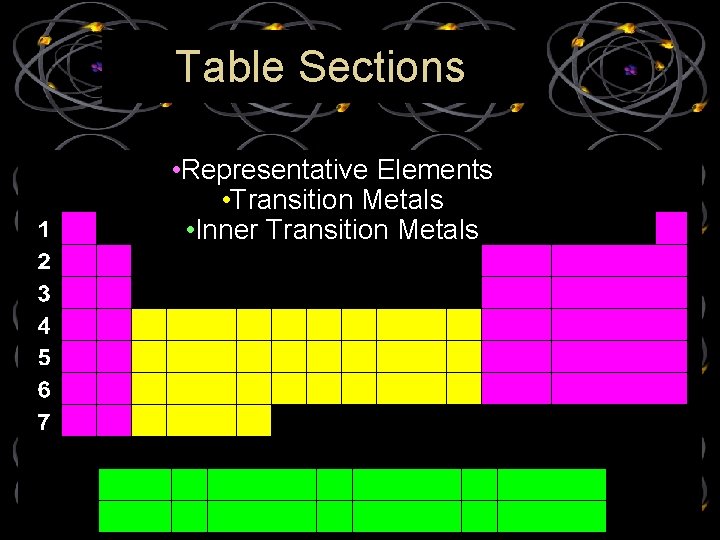
Table Sections • Representative Elements • Transition Metals • Inner Transition Metals
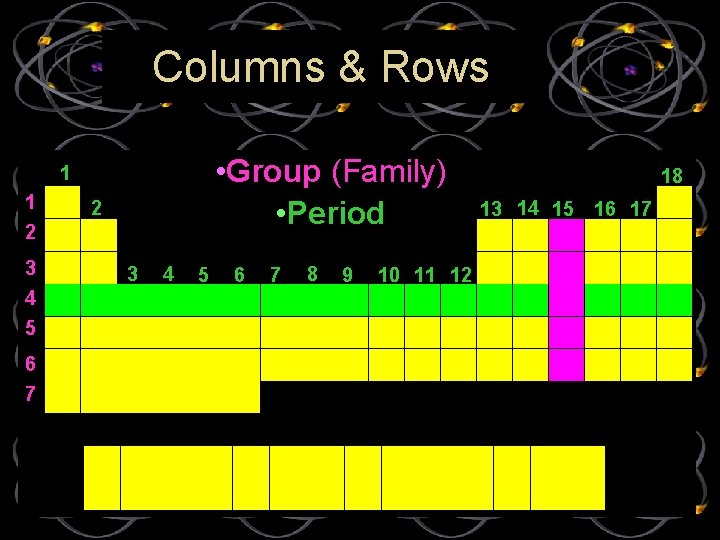
Columns & Rows • Group (Family) • Period 1 1 2 3 4 5 6 7 8 9 10 11 12 18 13 14 15 16 17

Terms • Periodic Law –Properties of elements repeat periodically when the elements are arranged by increasing atomic number. –Moving from left to right in a row of the periodic table, metallic properties decrease.

Terms • Valence Electrons –e- in the outermost energy level • First Ionization Energy –energy required to remove an e- from a neutral atom
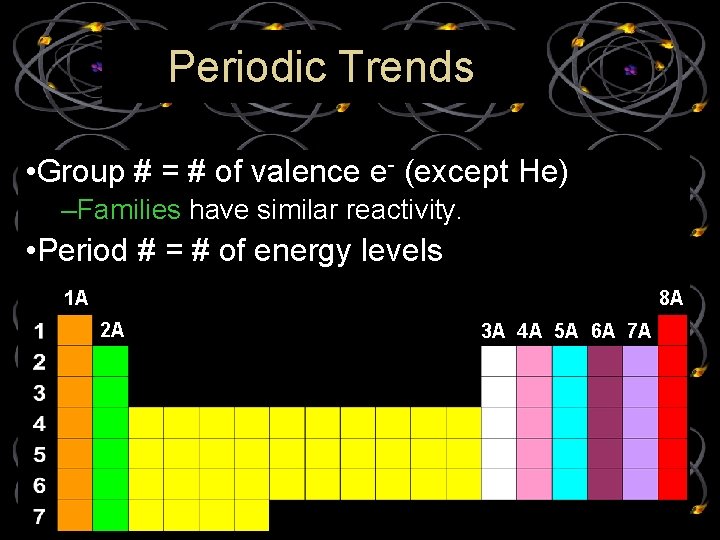
Periodic Trends • Group # = # of valence e- (except He) –Families have similar reactivity. • Period # = # of energy levels 1 A 8 A 2 A 3 A 4 A 5 A 6 A 7 A
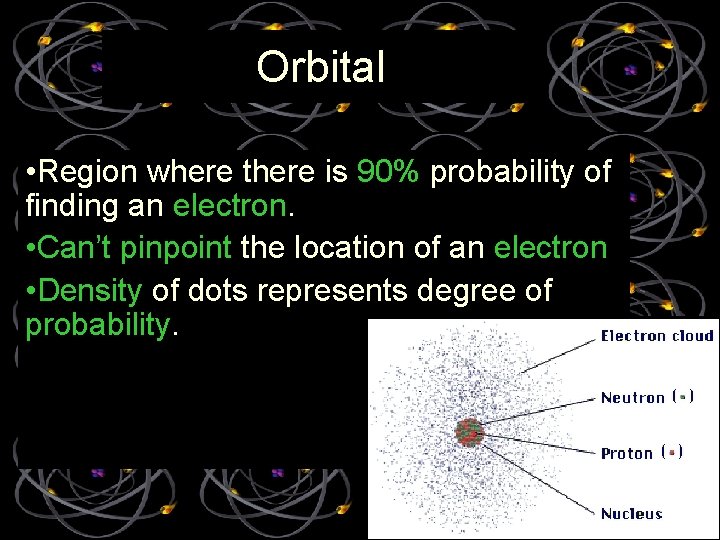
Orbital • Region where there is 90% probability of finding an electron. • Can’t pinpoint the location of an electron • Density of dots represents degree of probability.
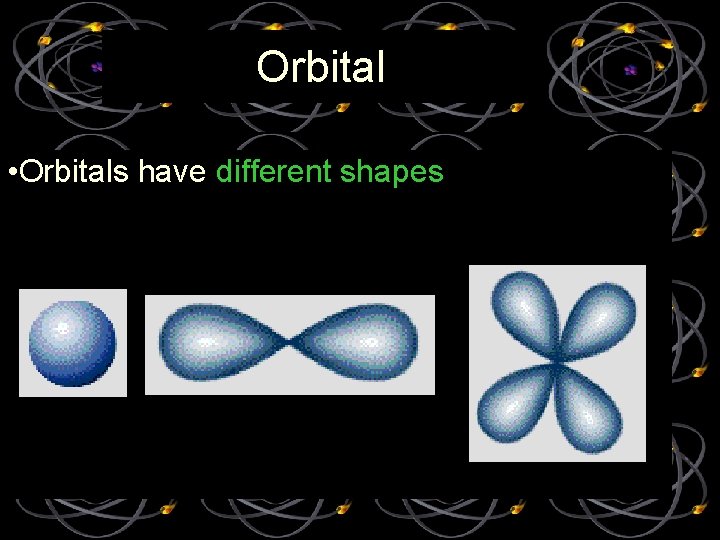
Orbital • Orbitals have different shapes
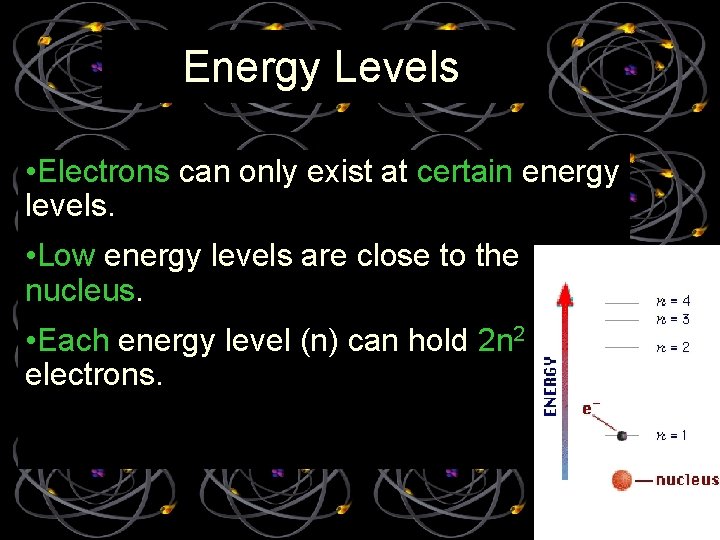
Energy Levels • Electrons can only exist at certain energy levels. • Low energy levels are close to the nucleus. • Each energy level (n) can hold 2 n 2 electrons.
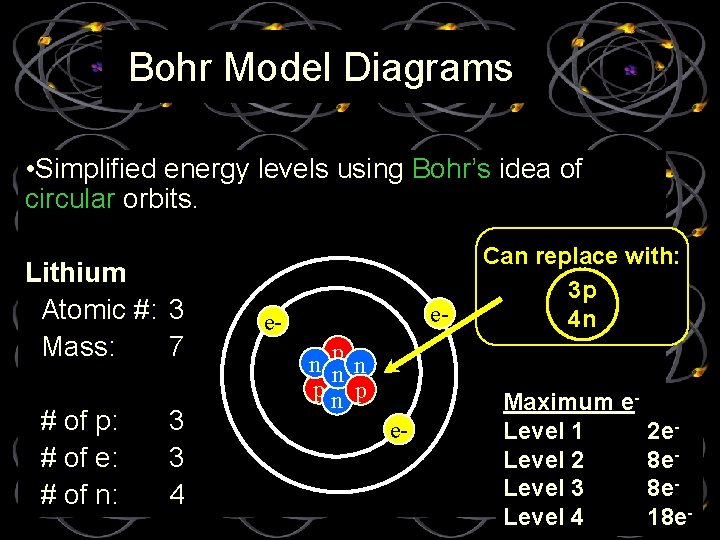
Bohr Model Diagrams • Simplified energy levels using Bohr’s idea of circular orbits. Lithium Atomic #: 3 Mass: 7 # of p: # of e: # of n: 3 3 4 e- en np n pn p e- Can replace with: 3 p 4 n Maximum e. Level 1 Level 2 Level 3 Level 4 2 e 8 e 8 e 18 e-
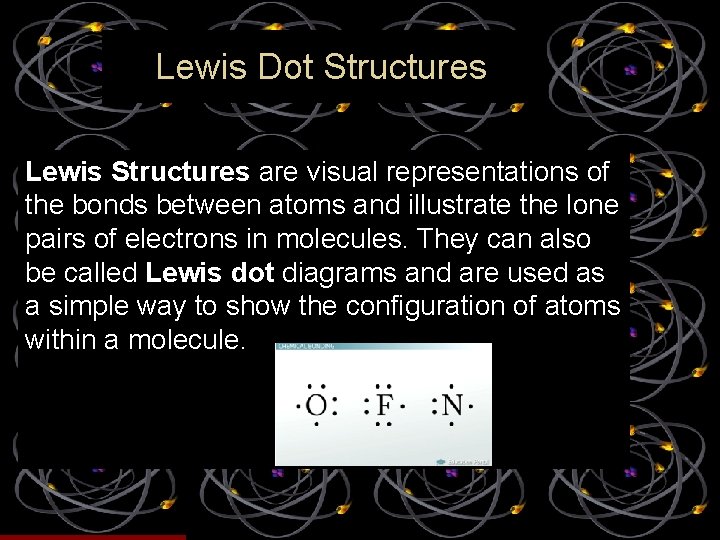
Lewis Dot Structures Lewis Structures are visual representations of the bonds between atoms and illustrate the lone pairs of electrons in molecules. They can also be called Lewis dot diagrams and are used as a simple way to show the configuration of atoms within a molecule.
- Slides: 16