Atoms and Molecules What is an atom Atoms


















- Slides: 23

Atoms and Molecules

What is an atom? • Atoms are the basic building blocks of matter that make up everyday objects (smallest unit of matter) • A desk, the air, even you are made up of atoms!

Can we see it? • We cannot see atoms with just our eyes or even with glasses • We need a very high powered microscope to see an atom

What is an atom? • The word “atom” means indivisible, meaning it cannot be broken down any further

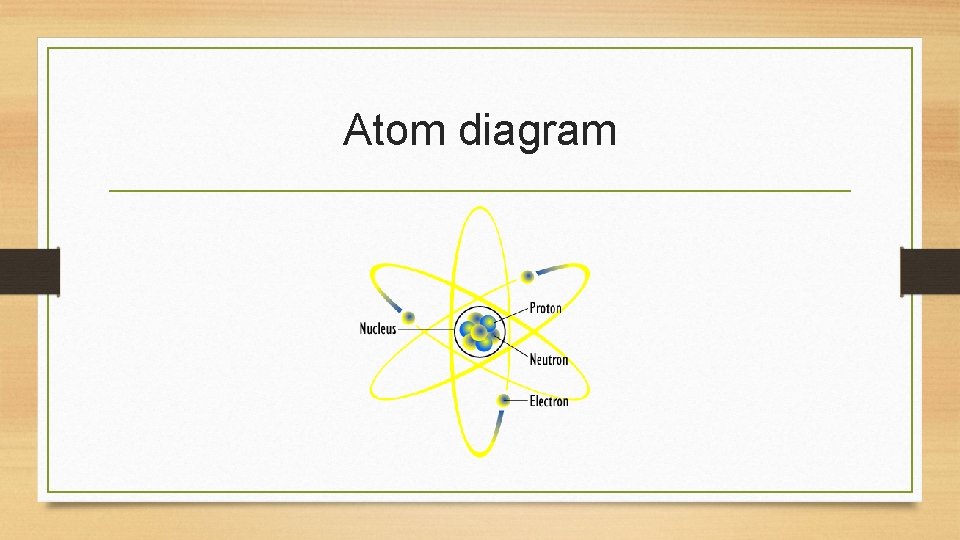
Structure of an atom • Atoms are made up of protons, neutrons, and electrons • Protons are positive (+) • Neutrons are neutral (0) • Electrons are negative (-) • In terms of mass, protons and neutrons are nearly equal • 1 proton = 1 neutron • 1836 electrons = 1 proton (electrons are extra, extra small)

Structure of an atom • The nucleus is the central or middle part of atoms • The nucleus is made up of protons and neutrons • Question: If the nucleus is always made up of protons and neutrons, what charge does the nucleus always hold?

Structures of an atom • Electrons are outside of the nucleus, orbiting in their electron shells or orbitals • Electrons move faster than the speed of light!!! • In 8 th Grade Science, you need to know that there always the same number of electrons as protons • So…. An atom has a neutral charge (protons and electrons cancel each other)

Atom diagram

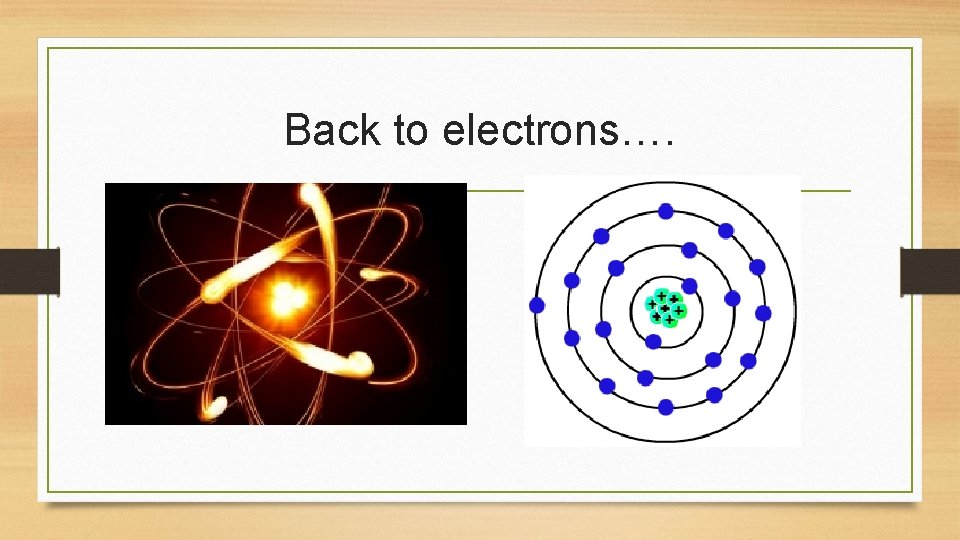
Back to electrons….

• Electrons allow for chemical bonding to occur (how compounds/chemical reactions generally occur) • Valence electrons are the electrons that allow chemical bonding to occur • These electrons are found on the furthest orbital/shell from the nucleus

How many valence electrons do we have here (in the element Sodium, or salt)?

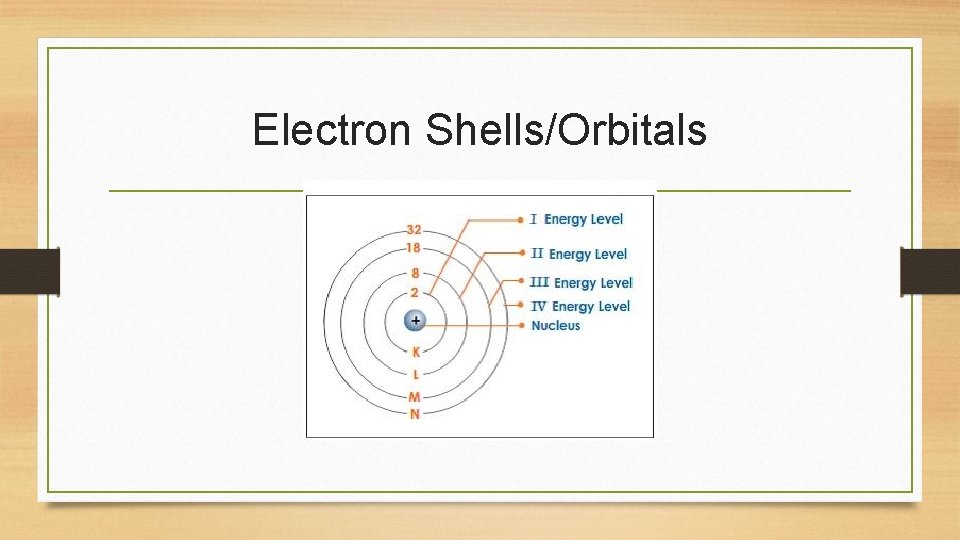
Electron Shells/Orbitals • Electron shells can only hold so many electrons • • Shell 1 - 2 electrons Shell 2 - 8 electrons Shell 3 - 18 electrons Shell 4 - 32 electrons • Atoms always keep trying to fill their shells!!!

Electron Shells/Orbitals

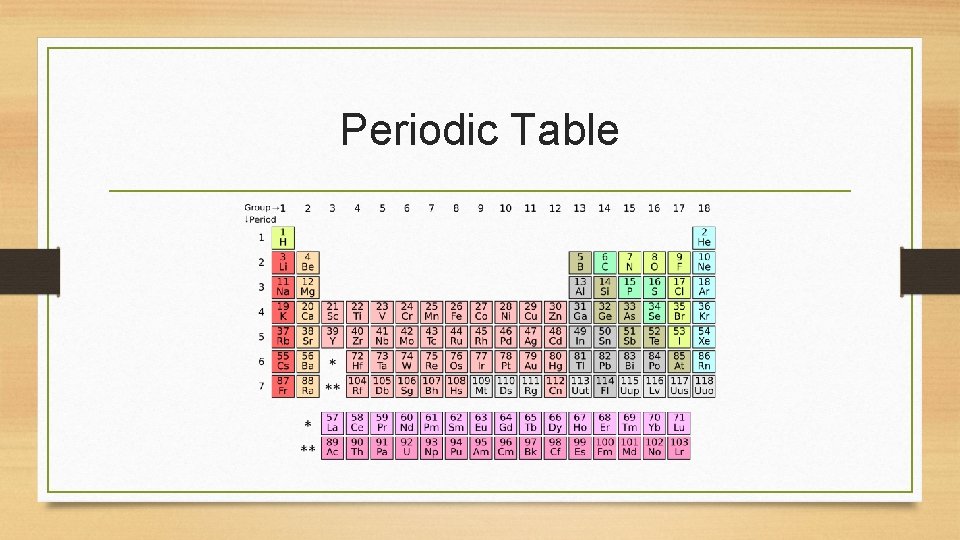
Periodic Table

How is the periodic table arranged? • • We read the periodic table like a book, or a magazine We read left to right (horizontally) Thus, moving from left to right, atomic numbers always increase by 1 Elements that are found in the same period (row/horizontal/left to right) have the same number of electron shells/orbitals • Elements that are found in the same group (vertical/column/up and down) have the same number of valence electrons • The periodic table is very useful for learning about chemistry and elements!!

What is an element? • An element is a substance that is made entirely from one type of atom. • Hydrogen, helium, carbon, and nitrogen are a few to name • There are 118 elements known! Most of these are natural occurring

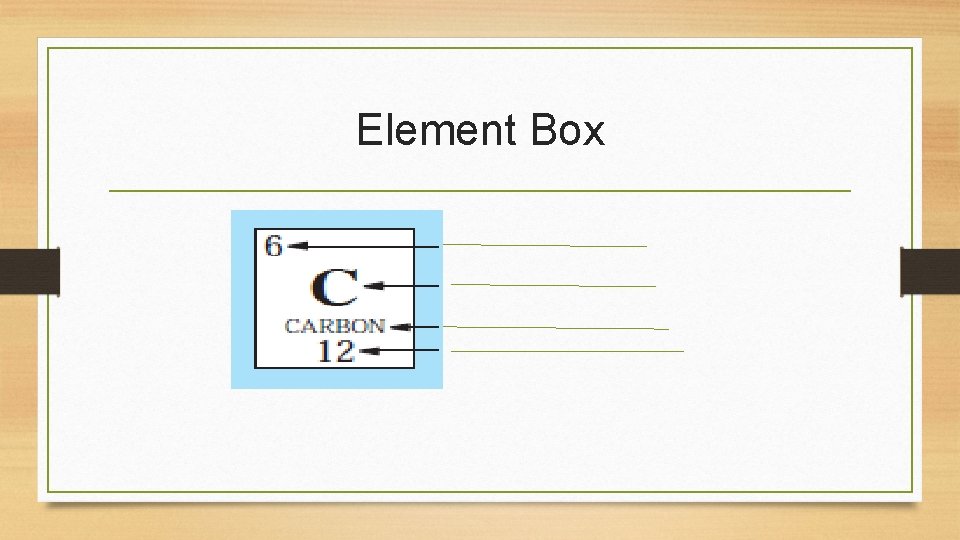
Element Box

• Atomic number equals protons AND electrons • How many protons does C have? • How many electrons does C have? • Always round atomic weight to nearest whole number • How do we find # of neutrons?

Important Formulas • Protons= atomic number • Electrons= atomic number • Protons = Electrons • Atomic mass = protons + neutrons (in nucleus) • Neutrons = atomic mass – atomic number • Example: for Carbon: • Atomic mass (12) – atomic number (6)= 6

Remember one of ATOM’s rules • Atoms are always neutral because there an equal amount of protons and electrons • If we have an atomic number of 16, then we have 16 protons and 16 electrons, therefore our atom is neutral because protons (+) and electrons (-) cancel each other out

Element Box Practice • Draw & fill in a square for the element Neon. • Also, find the number of neutrons that exist in one atom of neon

Element Box Practice • Draw & fill in a square for the element Nickel. • Also, find the number of neutrons that exist in one atom of nickel

Element and Periodic Table Practice • http: //education. jlab. org/elementmath/