Atoms and Elements Properties and Changes Scientific Terminology
















- Slides: 17

Atoms and Elements Properties and Changes

Scientific Terminology • Control – an event that is used as a constant and unchanging standard of comparison in scientific experimentation. • Variable - capable of being varied or changed; alterable • Hypothesis - A tentative explanation that accounts for a set of facts and can be tested by further investigation. • Trial - the act of trying, testing, or putting to the proof.

Nature of Matter • Particle Theory of Matter 1. All matter is made up of tiny particles 2. Each pure substance has its own kind of particles 3. Particles attract each other 4. Particles are always moving 5. Particles at a higher temperature move faster on average than particles at a lower temperature

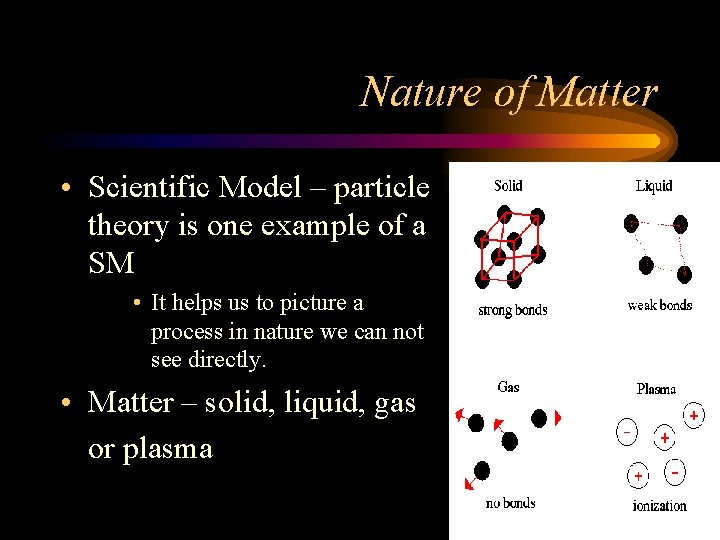
Nature of Matter • Scientific Model – particle theory is one example of a SM • It helps us to picture a process in nature we can not see directly. • Matter – solid, liquid, gas or plasma

Nature of Matter • Physical change – No new substance is being formed

Chemical change – At least one new substance, with new properties, is formed. – If two of the following are true, then a chemical change has probably occurred: – Heat is absorbed or produced – Starting material is used up – New colour appears – Material with new properties forms – Gas bubbles form in a liquid – Grains of solid appear in liquid

Nature of Matter • Physical Properties – observed and measured without forming a new substance – Colour = for gases the colour is often clear – Density = amount of matter that occupies a certain space • Qualitative physical property – Described but not measured

Physical Vs. Chemical Change • Physical change in a substance doesn't change what the substance is. • Chemical change where there is a chemical reaction, a new substance is formed and energy is either given off or absorbed.

Physical Vs. Chemical Properties • Physical properties can be observed or measured without changing its chemical composition. – Examples of physical properties include color and density. • Chemical properties may only be observed by changing the chemical identity of a substance. – In other words, the only way to observe a chemical property is by performing a chemical reaction. Examples of chemical properties include reactivity and flammability.

Physical Vs. Chemical Properties

Mixtures • Mixtures make up most of the matter in the world • Solutions – Solvent – substance that does the dissolving – Solute – substance that dissolves – Water + Salt = Chemical change? • Properties have changed however they can still be separated

Meet the Elements • Element symbols – Based on a lot of common names and used to simplify the process • Einsteinium, polonium, uranium – Format created in 1817 by Berzrelius for two reasons: • Provided standard symbols for all known elements • Showed how to create symbols for any new elements that might be discovered • P. 182 Table 5. 2

Electrons, Protons and Neutrons • Bohr Rutherford Diagram • Protons – place a P in the nucleus, # is the same as atomic number • Electrons – place dots in shells, # is the same as atomic number. Shells fill 2, 8, 8 in terms of # of electrons in each. Pair up electrons. • Neutrons – place an N in the nucleus, # is rounded atomic mass minus atomic number.

Chemical Formulas • Chemical Formula = uses symbols and numbers to represent the composition of a pure substance – Definite proportions • Molecule = smallest independent unit of a pure substance – Electrolysis separates water molecules as the added energy rips the molecules apart

Different kinds of elements • Metals = conduct electricity and heat, hammered into sheets (malleability), stretched into wires (ductility), shiny lustre, and are solids at room temperature. • Non-metals = Some are gases at room temp, not shiny, not stretched into wires, poor electricity and heat conductors • Metalloids = in-between a metal and a non-metal

Families of Metals • Characteristics – Group 1 alkali metals • React rapidly with water to produce hydrogen – Group 17 halogens • Most reactive non-metals – Chlorine & Bromine can cause many serious problems • Na (very reactive) + Cl (very reactive) = Na. Cl (table salt) – Group 18 noble gases • Chemically calm • Calm = noble in historic times

Isotopes, Ions and Electron Dot Diagrams • Isotope – alternative form of an element – Some are stable (normal), some are radioactive • Ions – atoms that have a charge – The charge can be positive or negative • Electron Dot Diagram – Puts electrons around the element name. The only electrons used are the ones in the outer ring.