Atoms and Elements Atoms and Elements Lesson Objectives

Atoms and Elements

Atoms and Elements Lesson Objectives � All: Use models and diagrams to explain the difference between atoms and elements � Most: describe the difference between an atom, element and compound � Some: Start using chemical symbols for common elements e. g. C for Carbon and O for oxygen.

Last week we learned that all substances are made of atoms are tiny particle that join together to make up different elements. Atoms are absolutely tiny. You would need to use a really expensive microscope to be able to see one…

Above to the left is a drawing of an atom. To the right is the first ever actual photograph showing the inside of an atom. Hopefully you can see that there is a part in the very middle, this is called the nucleus and it is like the brain of the atom. On the outside there is a shell, a bit like a coat the atom wears. Inside of this there is some space

Hopefully you can from the images on the previous slide that inside the atom there is a part in the very middle, this is called the nucleus and it is like the brain of the atom. On the outside there is a shell, a bit like a coat the atom wears. Inside of this there is some space where electrons fizz around You are probably thinking “Why do I need to know this? ”

is a substance that is made up of just one type of atom. Copper is an element made up of copper atoms only. Carbon is an element made up of Carbon atoms only. Helium is an element made up of helium atoms only.

What happens if something has more than 1 type of atom in it?

is a substance that has more than one type of atom Water is known as H 2 O This means that within each water mollecule there is 2 Hydrogen Atoms and 1 Oxggen atom See if you can draw a water molecule using simple coloured circles to represent the mollecules?
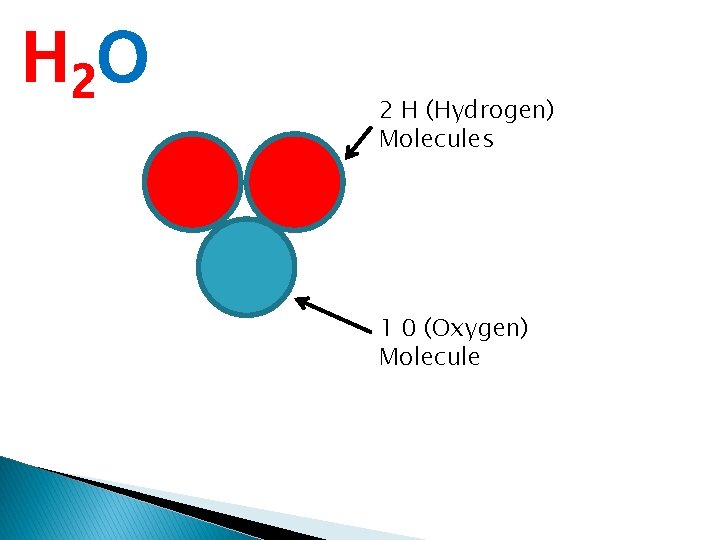
H 2 O 2 H (Hydrogen) Molecules 1 0 (Oxygen) Molecule

Now think about these atoms Oxygen O Blue Hydrogen H Red Carbon C Green Sulphur S Yellow Using coloured circles see if you can draw molecules of the following COMPOUNDS…

CO 2 Carbon dioxide

SO 2 Sulphur dioxide

CH 4 Methane

H 2 SO 4 Sulphuric acid

Carbon dioxide= CO 2 Now draw this compound answer the following questions 1. How many carbon atoms are there? 2. How many oxygen atoms are there? 3. How many atoms are there altogether? 4. How many elements are there?
- Slides: 15