Atoms 1 All substances are made from atoms

Atoms 1. All substances are made from atoms. 2. There about 100 different types of atoms 3. An element is a substance which contains only one type of atom
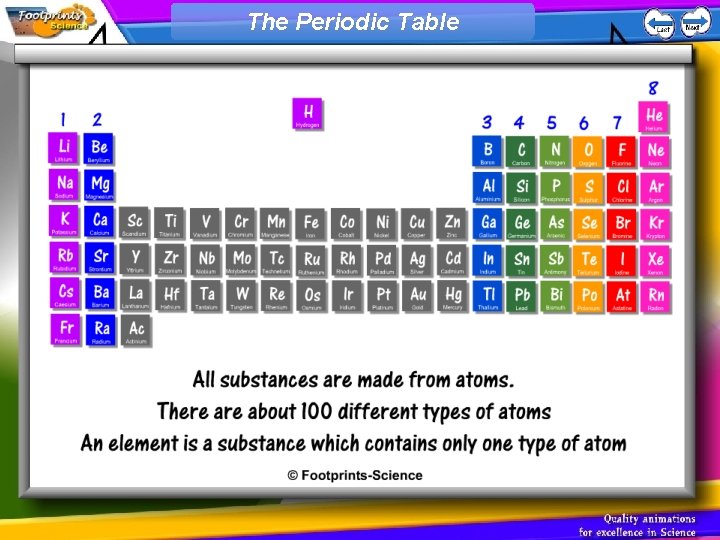
The Periodic Table
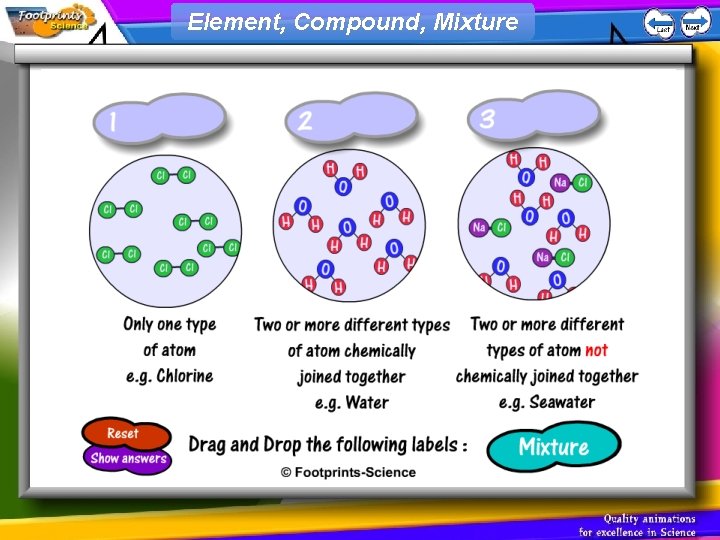
Element, Compound, Mixture

Structure of an Atom 1. Atoms have a small central nucleus containing protons and neutrons 2. Electrons orbit the nucleus 3. Protons, neutrons and electrons have different masses and charges
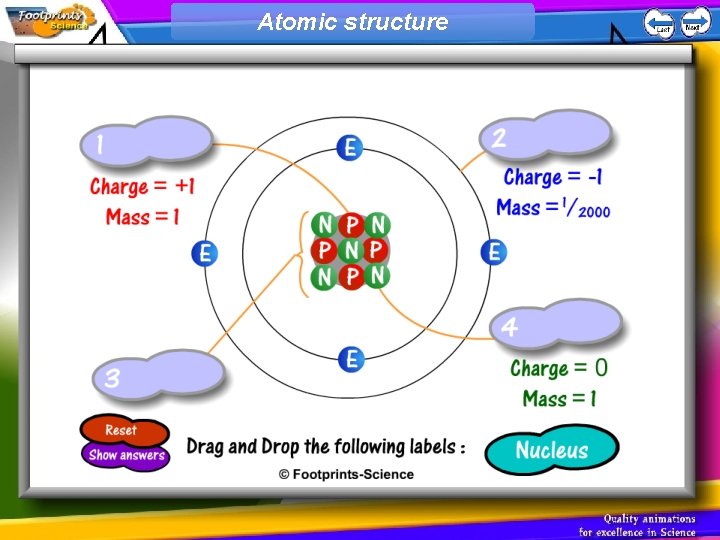
Atomic structure
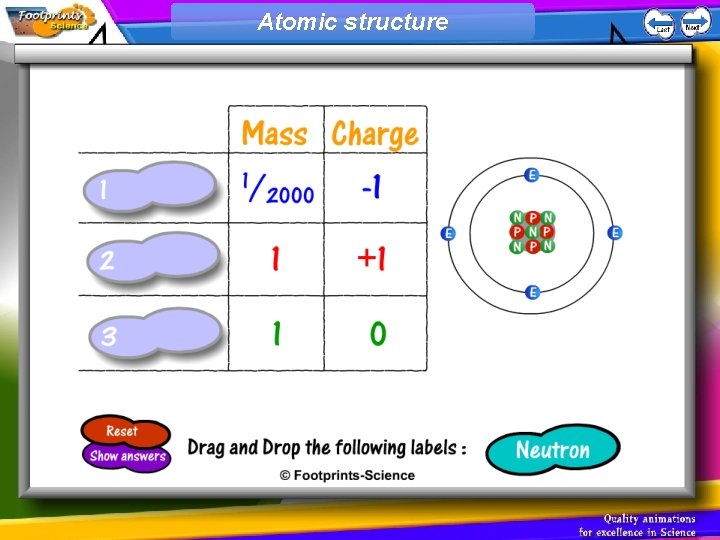
Atomic structure

Atomic number 1. The atomic number is the number of protons in the nucleus 2. In an atom, the number of electrons always equals the number of protons 3. Atoms do not have an overall electrical charge

Atomic number

Mass number 1. The mass number is the total number of protons and neutrons in an atom 2. The number of neutrons can be calculated by subtracting the atomic number from the mass number

Mass number
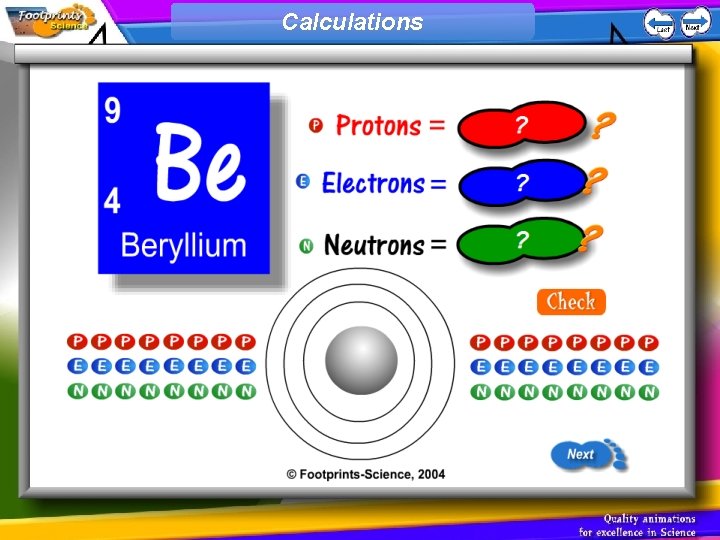
Calculations

Isotopes 1. Isotopes are atoms of the same element that have different numbers of neutrons (e. g. carbon-12 and carbon 14) 2. Isotopes have the same atomic number but different mass numbers
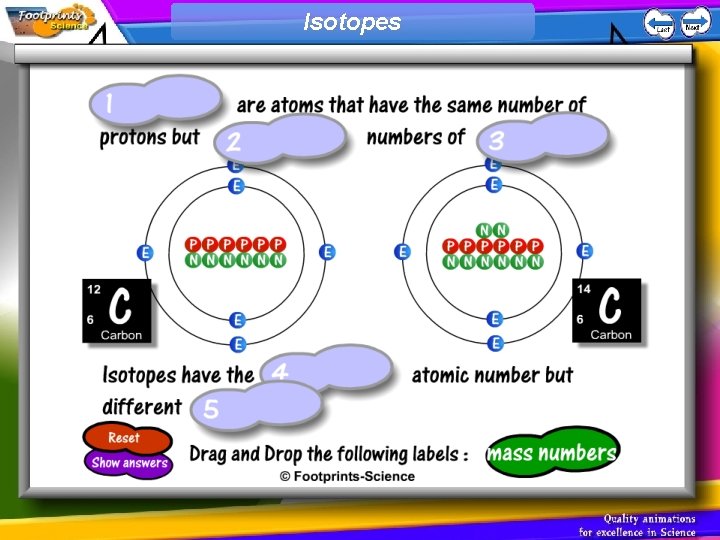
Isotopes

Electrons 1. Each electron in an atom is at a particular energy level (or shell) 2. Electrons fill the lowest energy levels first (nearest to the nucleus) 3. The first shell can take a maximum of 2 electrons, the second shell can take 8, and the third shell can take 8
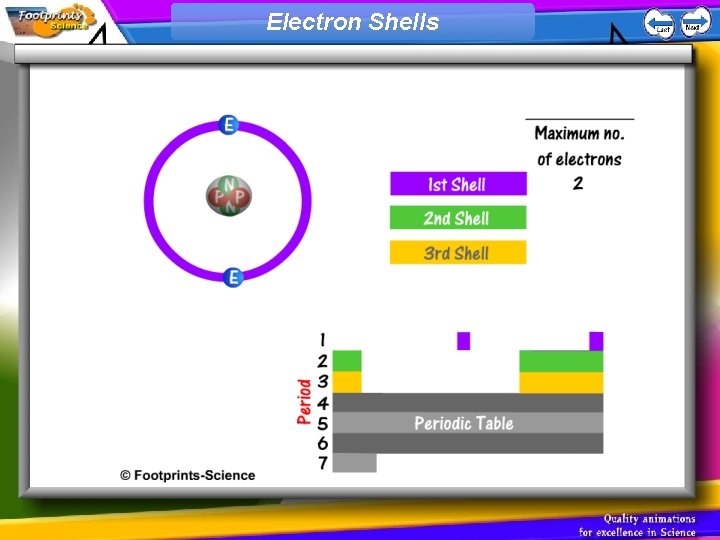
Electron Shells
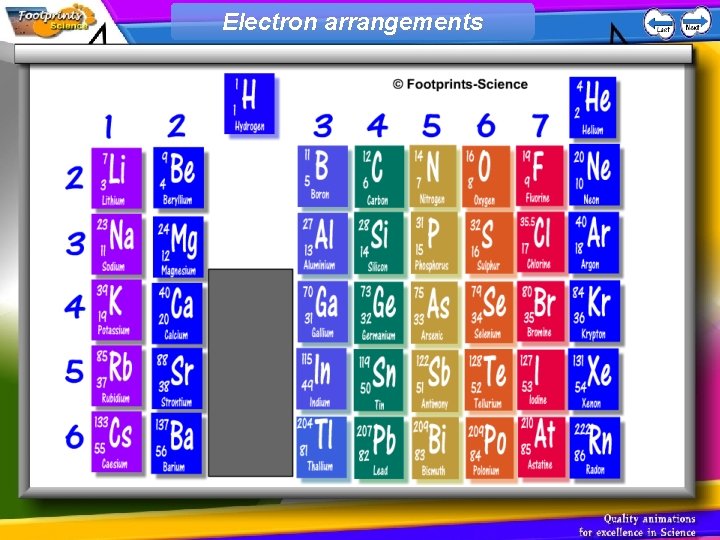
Electron arrangements
Wordsearch
Quiz
- Slides: 18