ATOMS 1 1228202 Major atoms in biology n

ATOMS 1 12/28/202

Major atoms in biology n. C HONSP n# of atoms in outer shell –H, –C –N, P –O, S 2 1 4 5 6 12/28/202
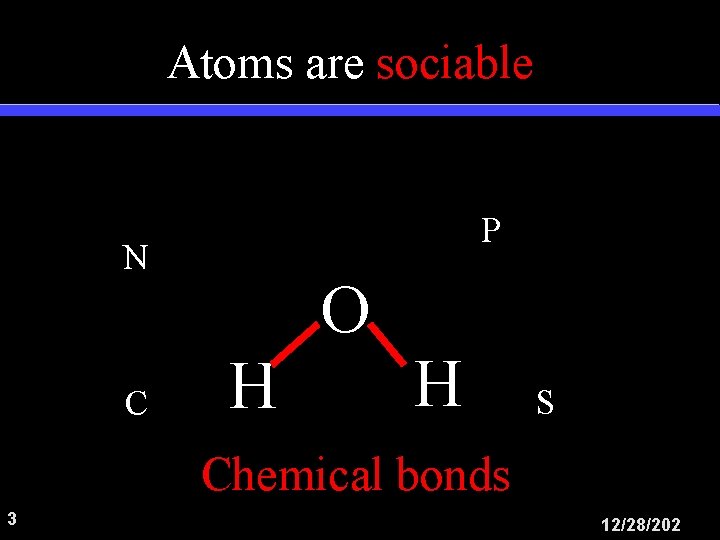
Atoms are sociable P N C O H H S Chemical bonds 3 12/28/202

Chemical bonds. . . Covalent Noncovalent 4 Strong Puny but important 12/28/202
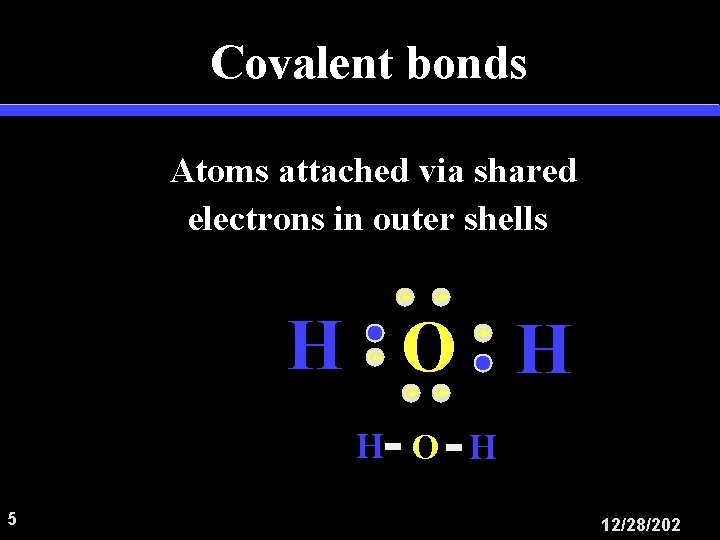
Covalent bonds Atoms attached via shared electrons in outer shells C C H O H 5 12/28/202

Covalent bonds - e. g. CH 3, ethane • stable (strong) 6 12/28/202
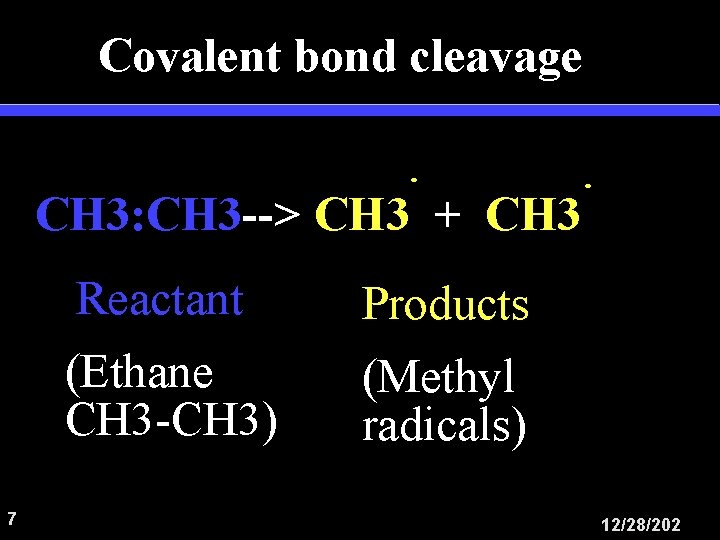
Covalent bond cleavage CH 3: CH 3 --> CH 3 + CH 3 7 Reactant Products (Ethane CH 3 -CH 3) (Methyl radicals) 12/28/202

Covalent bond cleavage Reactant Products CH 3: CH 3 --> CH 3 + CH 3 E = +83 kcal/mol E is the difference between the energy of reactants and products 8 12/28/202

Covalent bond strengths 0 -H 110 C-H 99 C-C 83 C=C 146 High bond strength confers stability 9 12/28/202
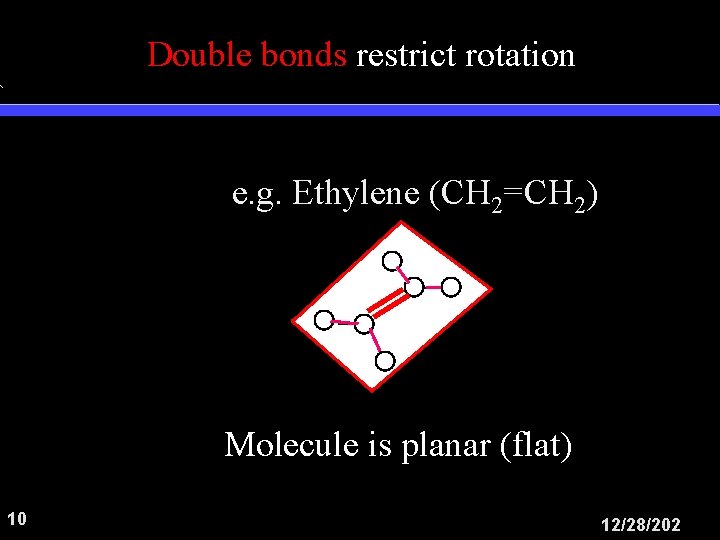
Double bonds restrict rotation e. g. Ethylene (CH 2=CH 2) H H Molecule is planar (flat) 10 12/28/202
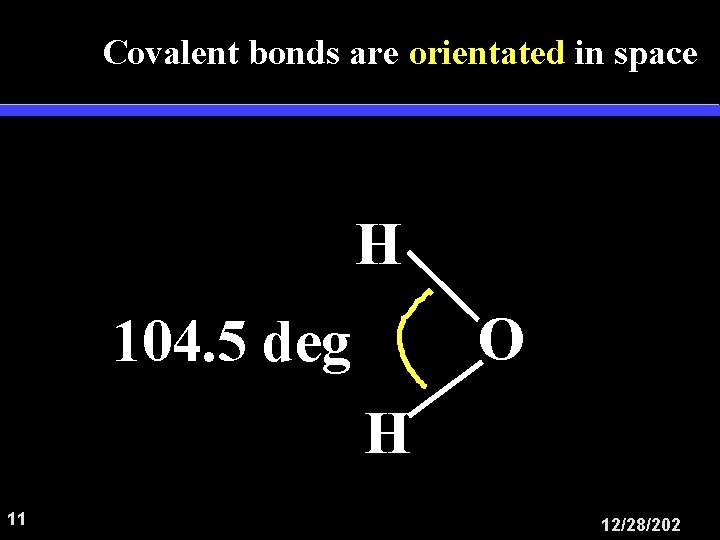
Covalent bonds are orientated in space H O 104. 5 deg H 11 12/28/202

Covalent Non-covalent Bond strength (kcal/mol) 50 -195 1 -5 • 4 kinds. . . 12 12/28/202

Non-covalent bonds • Hydrogen bonds • ionic bonds • van der Waal's interactions • hydrophobic interactions 13 12/28/202
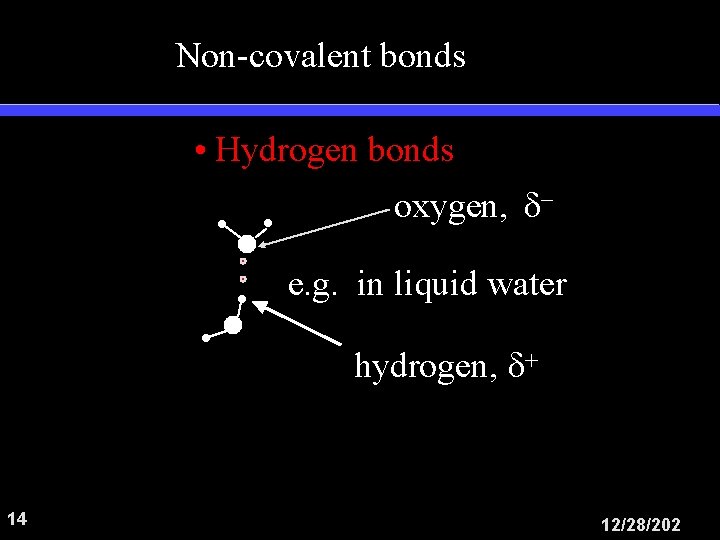
Non-covalent bonds • Hydrogen bonds oxygen, e. g. in liquid water hydrogen, 14 12/28/202

Hydrogen bond In liquid water Each molecule may be H-bonded to 4 others. Bonds are transient 15 12/28/202

Typical Hydrogen bonds H O or H N 16 12/28/202

Non-covalent bonds (cont. ) Ionic bonds Result of attraction between + and - charges of ionized atoms + e. g. Na Cl 17 12/28/202
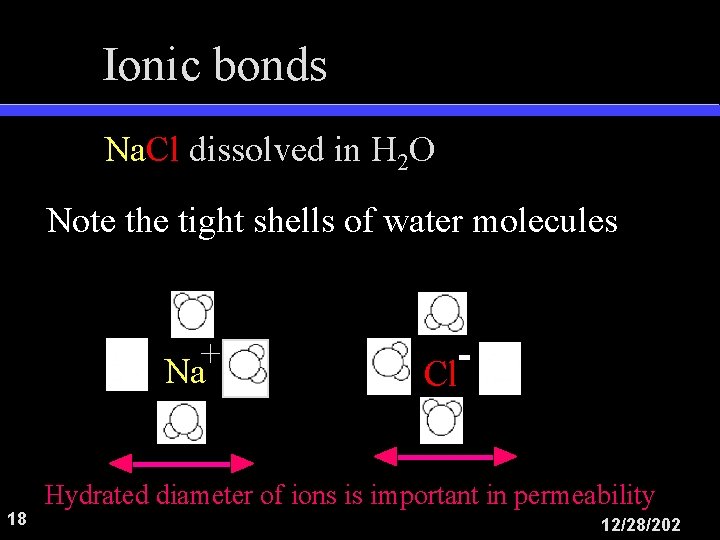
Ionic bonds Na. Cl dissolved in H 2 O Note the tight shells of water molecules + Na 18 Cl Hydrated diameter of ions is important in permeability 12/28/202

Non-covalent interactions (cont. ) Van der Waals interactions Due to electron fluxes any atom can become a transient dipole 19 12/28/202

Non-covalent interactions (cont. ) Van der Waals interactions 2 atoms in van der Waals contact 20 12/28/202
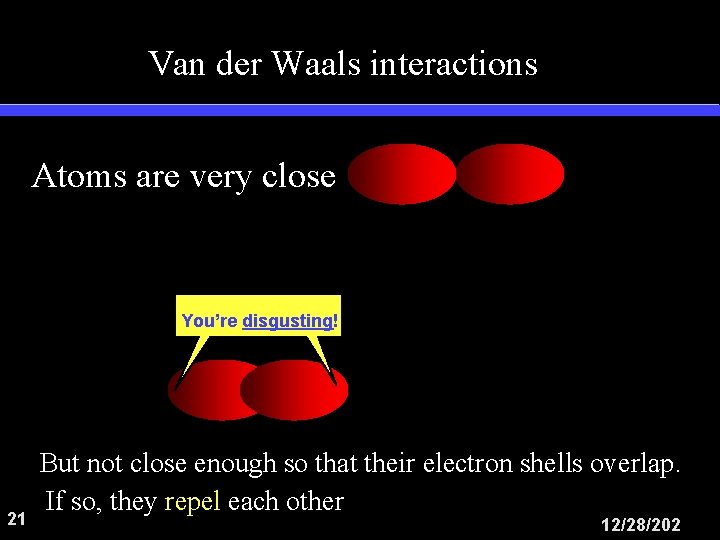
Van der Waals interactions Atoms are very close You’re disgusting! 21 But not close enough so that their electron shells overlap. If so, they repel each other 12/28/202

Our next Attraction Hydrophobia Starring a cast of thousands of greasy characters Hydrocarbons 22 12/28/202
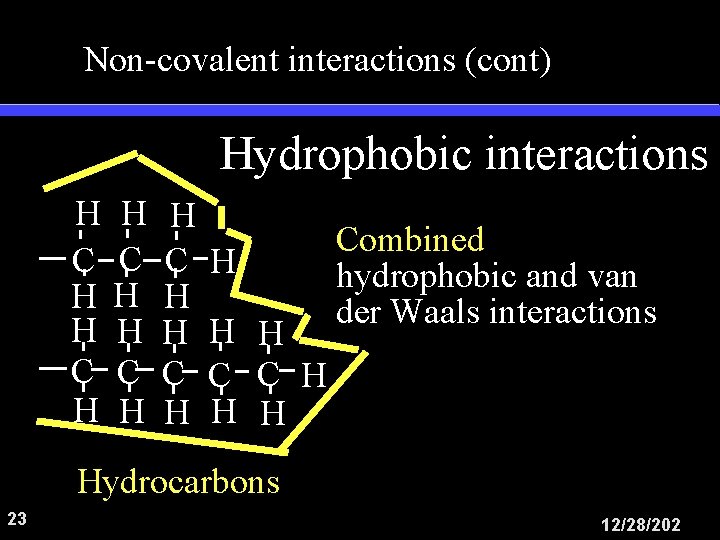
Non-covalent interactions (cont) Hydrophobic interactions H C H H C H H H H C C H H H Combined hydrophobic and van der Waals interactions Hydrocarbons 23 12/28/202
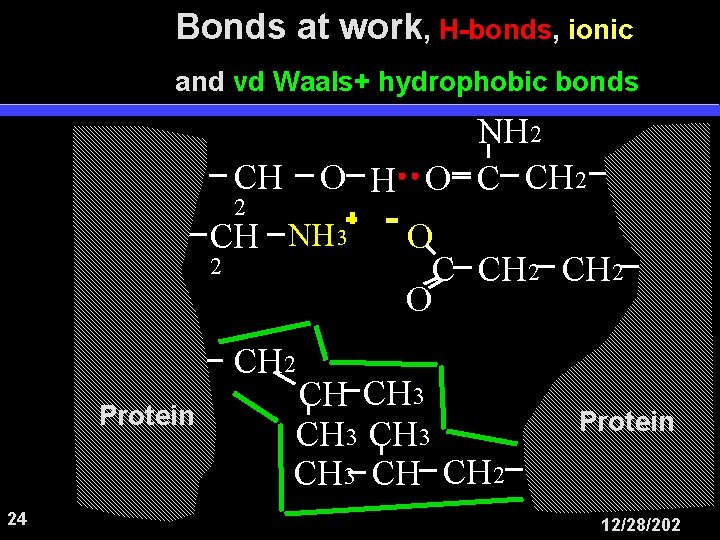
Bonds at work, H-bonds, ionic and vd Waals+ hydrophobic bonds NH 2 O H O C CH 2 CH NH 3 2 O C CH 2 O CH 2 Protein 24 CH CH 3 CH CH 2 Protein 12/28/202
Trivia Time How many water molecules in a teaspoonful? A one, a two, a three. . . As many as the teaspoonfuls of water present in the Atlantic Ocean 25 12/28/202

An everyday protein molecule, hemoglobin All your hemoglobin alpha chains are identical with 574 amino acids chained together in precise sequence In the past second you have just assembled 400 million of them 26 12/28/202

27 12/28/202
- Slides: 27