Atomic Theory The modern concept of the atom





















































- Slides: 96

Atomic Theory? The modern concept of the atom was born out of a series of theories and laws that were proposed in the 1700’s and 1800’s. It took many steps and evolutions to arrive at what we know about the atom today; it is worthwhile to look at the history of this evolution.

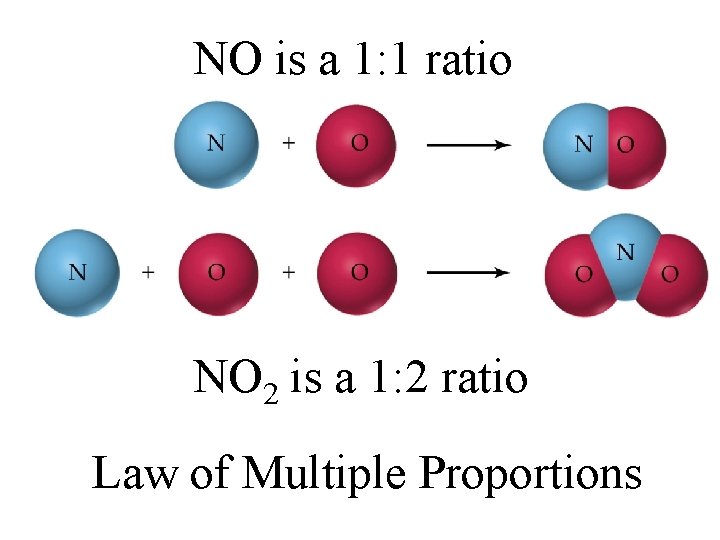
The Early Observations 1)Law of Definite Proportions chemical compounds always contain the same elements in the same proportions 2) Law of Multiple Proportions elements always combine in ratios of small whole numbers 3) Law of Conservation of Mass matter is never created nor destroyed

NO is a 1: 1 ratio NO 2 is a 1: 2 ratio Law of Multiple Proportions

Dalton’s Atomic Theory (1800’s) 1)All matter is composed of small particles called atoms. Still true 2) Atoms of a given elements are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties. True except mass can vary due to isotopes

Dalton’s Atomic Theory 3) Atoms of different elements combine in simple whole-number ratios to form chemical compounds. Definite Composition 4) In chemical reactions, atoms are combined, separated, or rearranged.

Dalton’s Atomic Theory 5) Atoms cannot be divided. Can divide into protons, neutrons, electrons, & quarks Atoms cannot be created or destroyed Conservation of Mass & Energy

Chemical Symbols • Shorthand name for an element • Watch how you write them ! Co = Cobalt CO = Carbon and Oxygen Ni = Nickel NI = Nitrogen and Iodine

Sub-atomic Particles - the important characteristics of these particles are their mass and charge. Names Symbol Charge Mass (amu) proton p+ +1 1. 0 neutron n 0 1. 0 electron e- -1 1/1837 - Tiny

Subatomic Particles electron – neg. charged subatomic particle located outside the nucleus, tiny!!!! proton – subatomic particle of the nucleus that has a positive charge. neutron – electrically neutral subatomic particle located in the nucleus

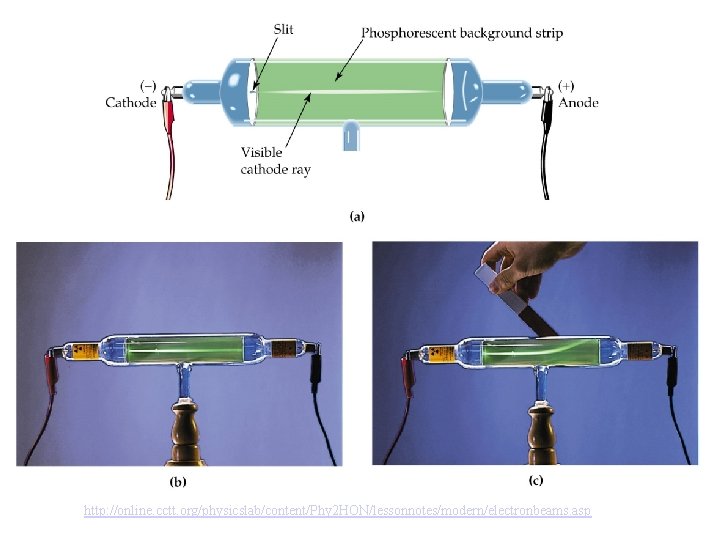
JJ Thomson Exp (electrons) Thomson used a cathode ray tube to discover that light was made of negatively charged particles, electrons. the cathode ray can be “bent” in the presence of a magnet!

http: //online. cctt. org/physicslab/content/Phy 2 HON/lessonnotes/modern/electronbeams. asp

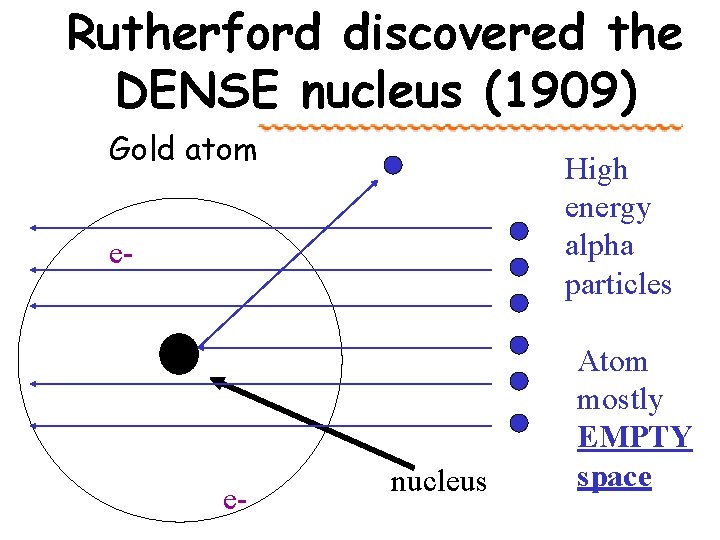
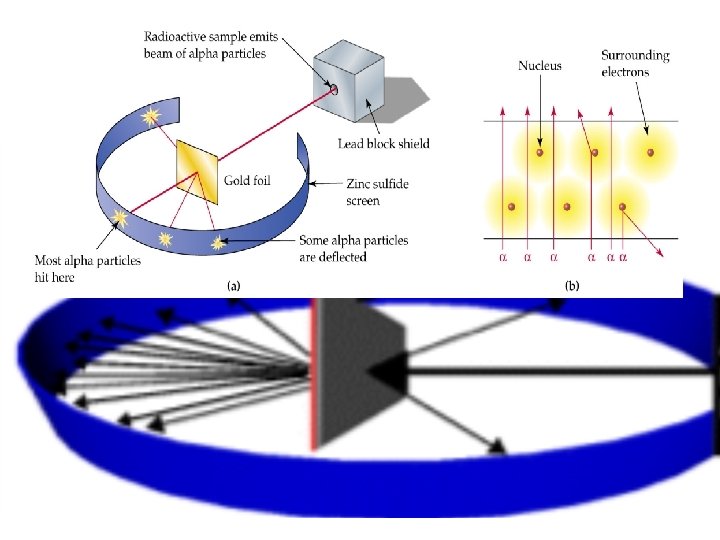
Rutherford discovered the DENSE nucleus (1909) Gold atom High energy alpha particles e- e- nucleus Atom mostly EMPTY space


How does the nucleus stay together? Protons DON’T want to be close – they repel each other There is a STRONG force called the nuclear binding energy to hold all those POSITIVE protons so close together. Nuclear force > Electrostatic (repelling force)

Two regions of the atom Nucleus – positively charged, small, BUT DENSE (most of atom weight) central portion of an atom composed of protons & neutrons. Electron Cloud – negatively charged, takes up most of the atom space, but mostly EMPTY SPACE as electrons are tiny! (cloud area 10, 000 times bigger than nucleus)

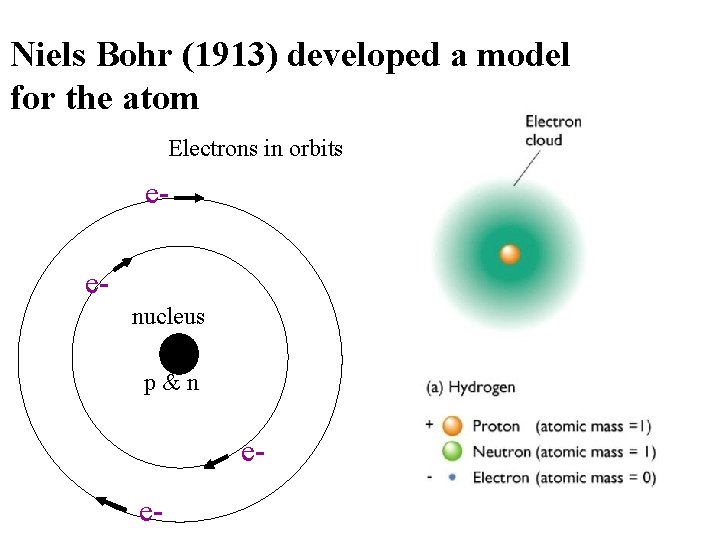
Niels Bohr (1913) developed a model for the atom Electrons in orbits eenucleus p&n ee-

Review of the Progression of the Atomic Model • Dalton - everything is made up of atoms • JJ Thompson- atoms have negatively charged electrons • Rutherford- atoms have a nucleus that hold the positively charged mass, but are mostly taken up by negatively charged space • Bohr- Electrons surround the nucleus in distinct orbitals

Quarks • Discovered in the late 1960’s & early 1970’s • 3 families of quarks, each family contains 2 quarks • 1 st Family is Up and Down & 3 of them join to form a proton or a neutron Proton = Up + Down Up charge is +2/3 + -1/3 Down charge is – 1/3 = +1 Neutron = Up + Down 2/3 + -1/3 = 0 charge Charm & Strange – only exist at high energies Top & Bottom only exist at very high energies

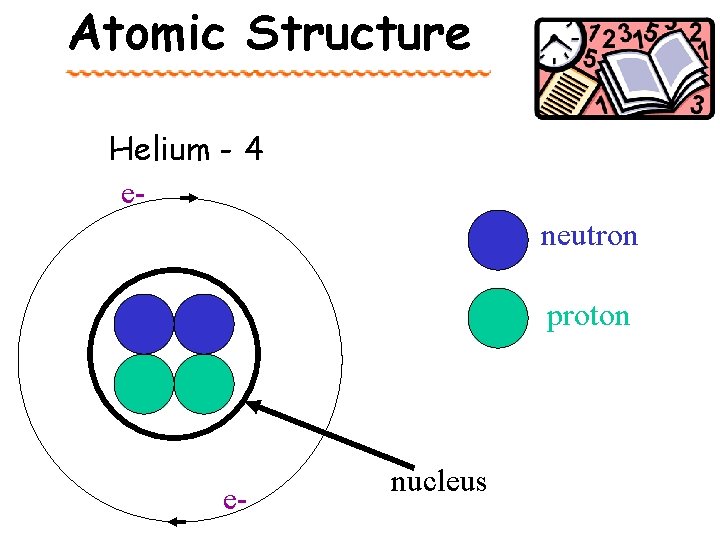
Atomic Structure Helium - 4 eneutron proton e- nucleus

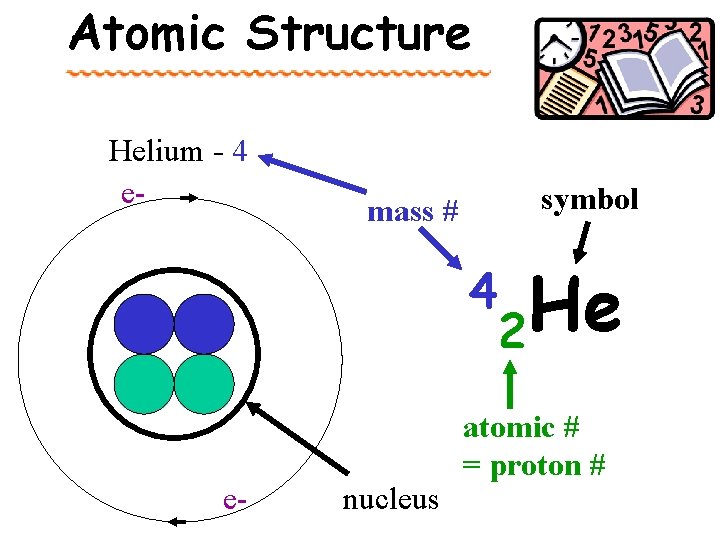
Atomic Structure Helium - 4 e- symbol mass # 4 e- nucleus He 2 atomic # = proton #

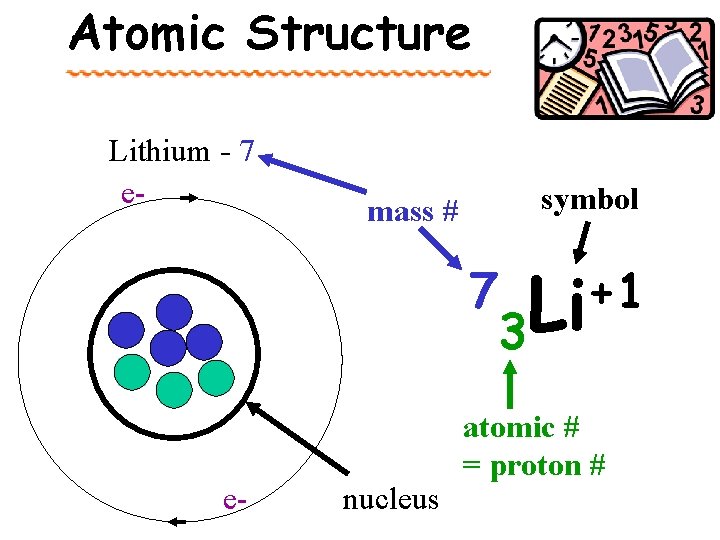
Atomic Structure Lithium - 7 e- symbol mass # 7 e- nucleus 3 +1 Li atomic # = proton #

The Chemists’ Shorthand: Atomic Symbols Mass number 39 K 19 Atomic number = Proton # Element Symbol

Definitions atomic number – the number of protons in the nucleus of an atom. Found on periodic table (PT) mass number – the total number of protons & neutrons in a nucleus. Calculated = p + n average atomic mass – the weighted avg. of the masses of the naturally occurring isotopes of an element. Found on periodic table

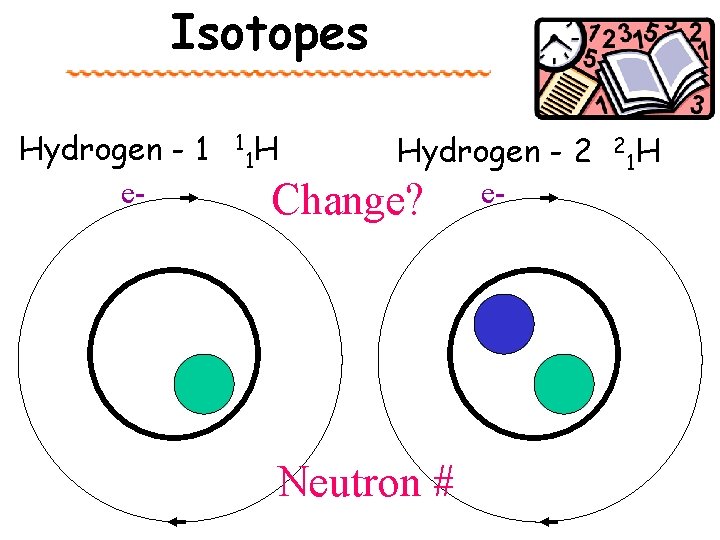
Isotopes Hydrogen - 1 e- 1 1 H Hydrogen - 2 Change? e- Neutron # 2 1 H

Changes or Not? Proton # Atomic # NEVER Changes Neutron # Changes Electron # 1 proton = H 2 protons = He Creates Isotopes Neutral p = e Changes Negative = add electrons to atomic # (anion) Positive = subtract electrons from atomic # (cation) Best equation to always use: Electron # = Proton # – charge

Isotope Practice Sodium-23 23 11 Na 11 protons, __ 11 electrons, __ 12 neutrons __ Copper-64 64 29 Cu 29 protons, 29 __ __ electrons, __ 35 neutrons Silver-108 47 Ag 47 protons, __ 47 electrons, 61 __ __ neutrons

Charge Practice 23 11 Na 1+ 11 protons, __ 10 electrons, __ 12 neutrons __ 37 17 Cl 1 - 17 protons, 18 __ __ electrons, __ 20 neutrons 88 2+ Sr 38 38 protons, __ 36 electrons, 50 __ __ neutrons

Avg. Atomic Mass? Average Atomic Mass is… - weighted average of the atomic of the naturally occurring isotopes. Isotopes % Mass Number abundance Wt. Avg. Mass x % Carbon-12 12 98. 90 % 12 x. 989 = 11. 868 Carbon-13 13 1. 10 % 13 x. 011 = 0. 143 Carbon-14 14 trace Avg. Atomic Mass 12. 011 (11. 868 +. 143)



What is a Mole? A mole is… - SI unit for amount of substance - Avogadro’s Number -> 6. 022 x 1023 particles/1 mole - this is the # of atoms contained in exactly 12 g (1 mole) of carbon. An amount – like 12 eggs in 1 dozen !

What is a Molar Mass? Molar mass is… - the mass of 1 mole of substance For example: 1 mole of carbon has a mass of 12. 01 g 1 mole of silicon has a mass of ____ 28. 09 g 196. 97 g 1 mole of gold has a mass of _____

Units and the Periodic Table Masses reported in the Periodic Table can be used two ways: - a special unit just for atoms called an atomic mass unit (amu). - one mole of substance has a mass in grams exactly equal reported in the Periodic Table (g/mol).

To think about • If you had 1 dozen chicken eggs (12) and compared them to 1 dozen ostrich eggs (12 also of those), they weigh different amounts. • SAME with the mole. • 1 mole (6. 022 x 1023 atoms) of Carbon weighs less then 1 mole (6. 022 x 1023 atoms) of gold (gold atom is bigger)

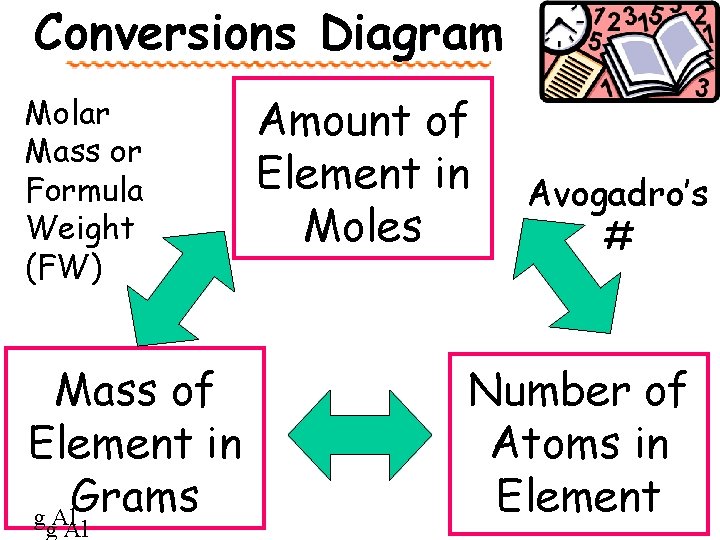
Conversions Diagram Molar Mass or Formula Weight (FW) Mass of Element in Grams g Al Amount of Element in Moles Avogadro’s # Number of Atoms in Element

Conversion Practice 1. Moles in 107. 92 g Al? 1 mole Al __107. 92 g Al________ 26. 982 g Al 4. 00 mol Al -> ___ 2. Grams in 1. 51 moles Cu? 1. 510 moles Cu___63. 546 g Cu __ 1 mole Cu 95. 95 -> _____ grams Cu

Conversion Practice 3. Grams in 0. 5 moles Ag? -> 53. 9 grams Ag 107. 868 g Ag 0. 5 moles Ag_____ 1 mole Ag 4. Moles in 124 g Mg? 1 mole Mg 124 g Mg___ _______ 24. 305 g Mg -> 5. 1 mole Mg

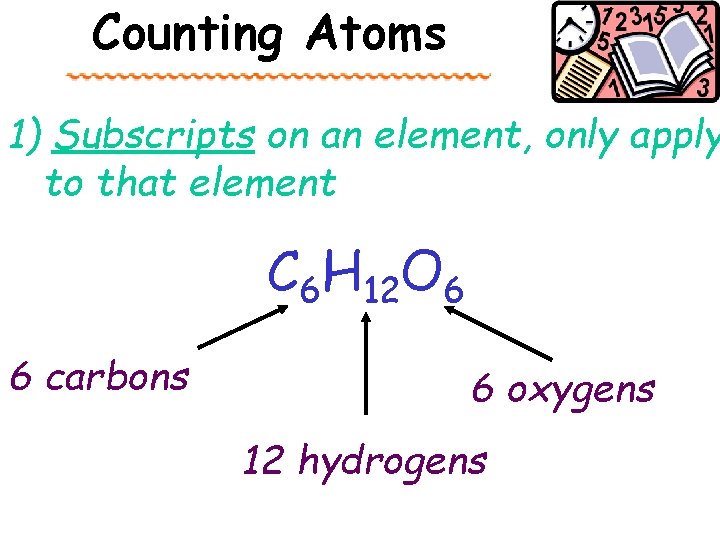
Counting Atoms 1) Subscripts on an element, only apply to that element C 6 H 12 O 6 6 carbons 6 oxygens 12 hydrogens

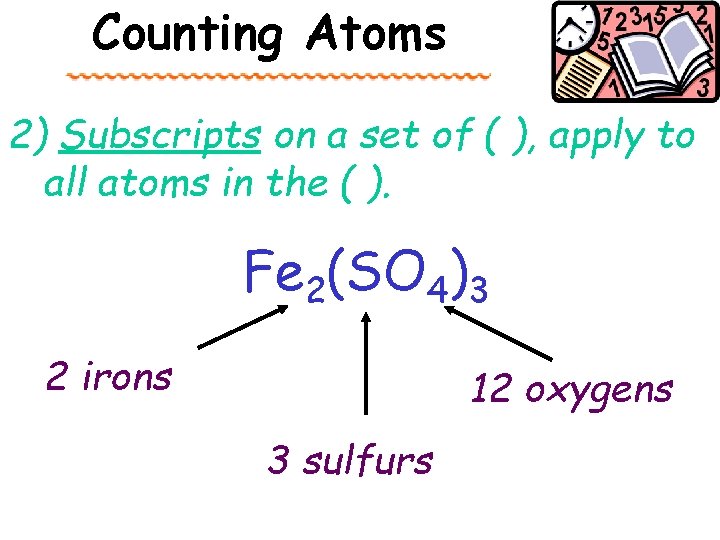
Counting Atoms 2) Subscripts on a set of ( ), apply to all atoms in the ( ). Fe 2(SO 4)3 2 irons 12 oxygens 3 sulfurs

For parenthesis – you multiply • Just in case you didn’t get 12 oxygen, you multiply the number on the outside of the parenthesis with the numbers inside. Fe 2(SO 4)3 Means there are 3 (SO 4) groups SO 4 3 S and 12 O
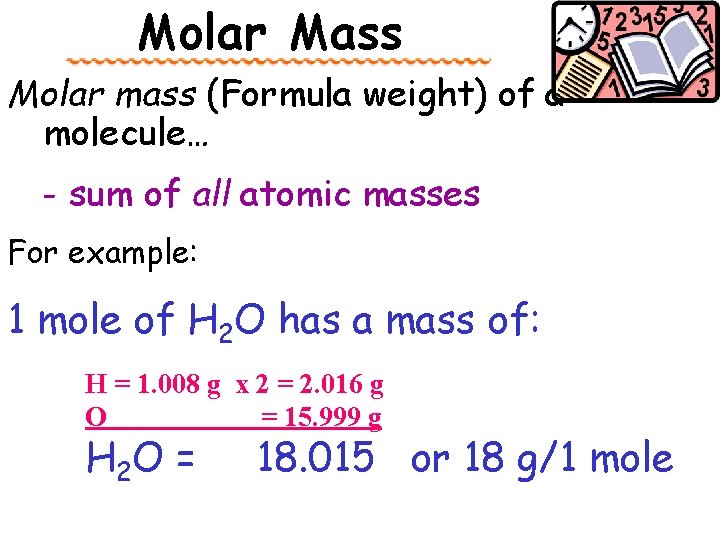
Molar Mass Molar mass (Formula weight) of a molecule… - sum of all atomic masses For example: 1 mole of H 2 O has a mass of: H = 1. 008 g x 2 = 2. 016 g O = 15. 999 g H 2 O = 18. 015 or 18 g/1 mole

Molar Mass 1 mole of Na. Cl has a mass of Na = 23 g Cl = 35. 5 g Na. Cl = 58. 5 g / 1 mole of Si. O 2 has a mass of Si = 28. 1 g O = 16 g x 2 = 32 g Si. O 2 = 60. 1 g / 1 mole

Molar Mass Practice Calc. Molar Mass of the following: • H 2 O 2 (hyd. peroxide) -> 34. 02 g/mol • CH 3 OH (methanol) -> 32. 05 g/mol • Mg(OH)2 (Magnesium hydroxide) -> 58. 305 g/mol • Fe 2(SO 4)3 (Iron III sulfate) -> 399. 885 g/mol

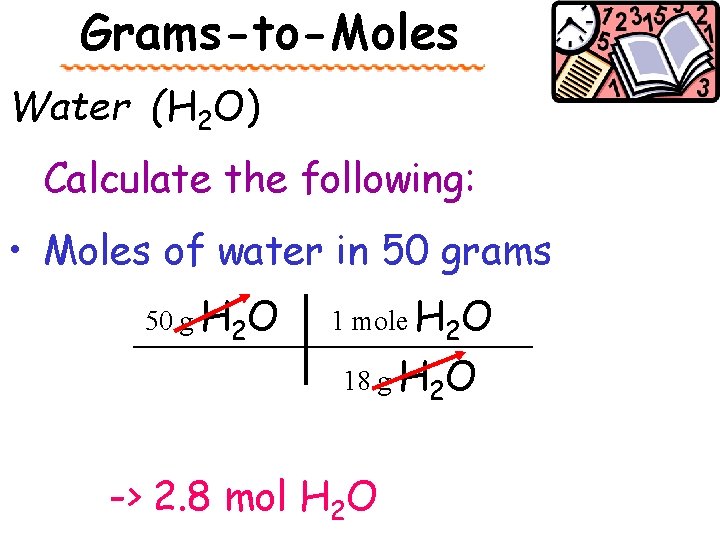
Grams-to-Moles Water (H 2 O) Calculate the following: • Moles of water in 50 grams 50 g H 2 O 1 mole H 2 O __________ 18 g H 2 O -> 2. 8 mol H 2 O

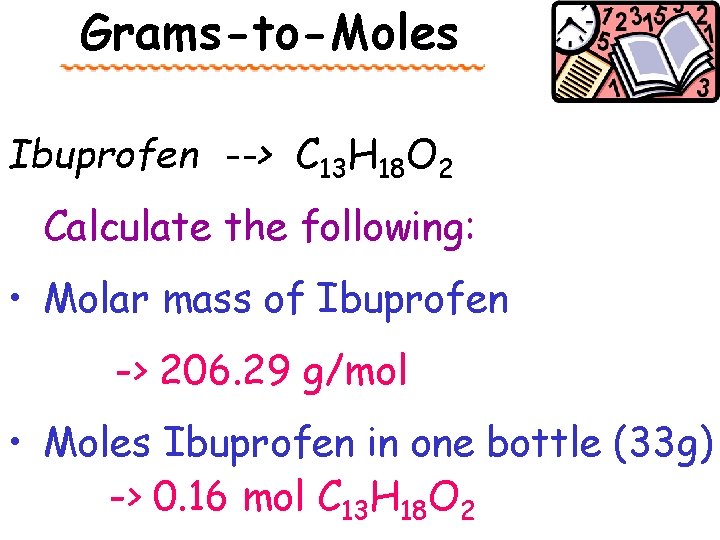
Grams-to-Moles Ibuprofen --> C 13 H 18 O 2 Calculate the following: • Molar mass of Ibuprofen -> 206. 29 g/mol • Moles Ibuprofen in one bottle (33 g) -> 0. 16 mol C 13 H 18 O 2

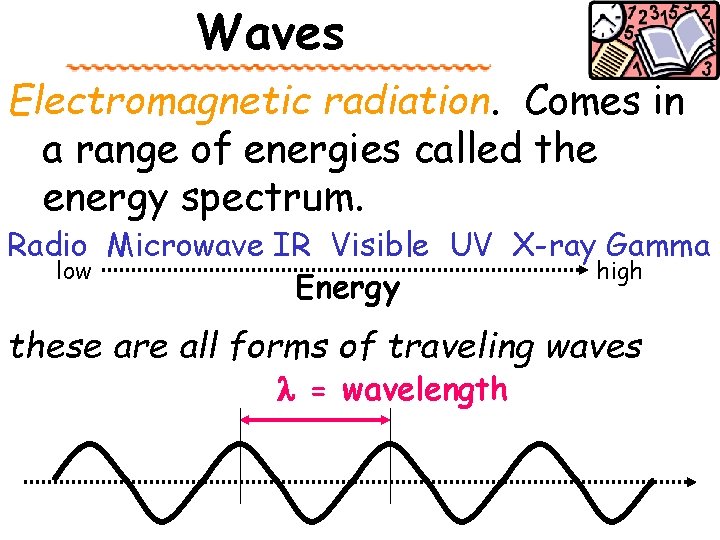
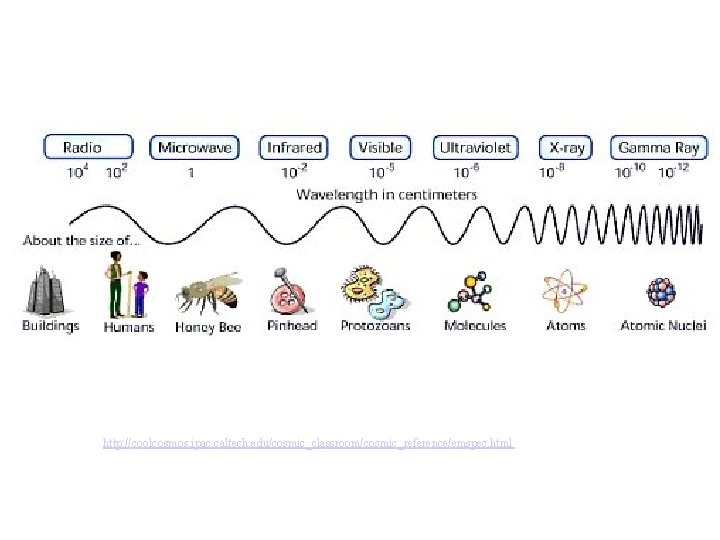
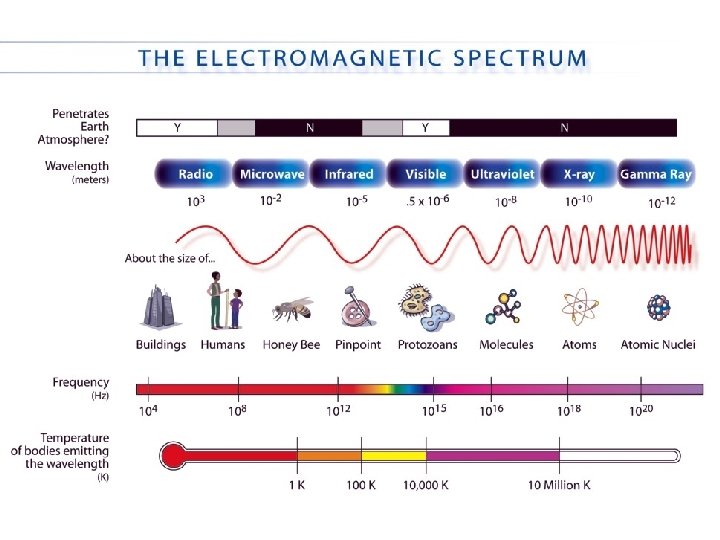
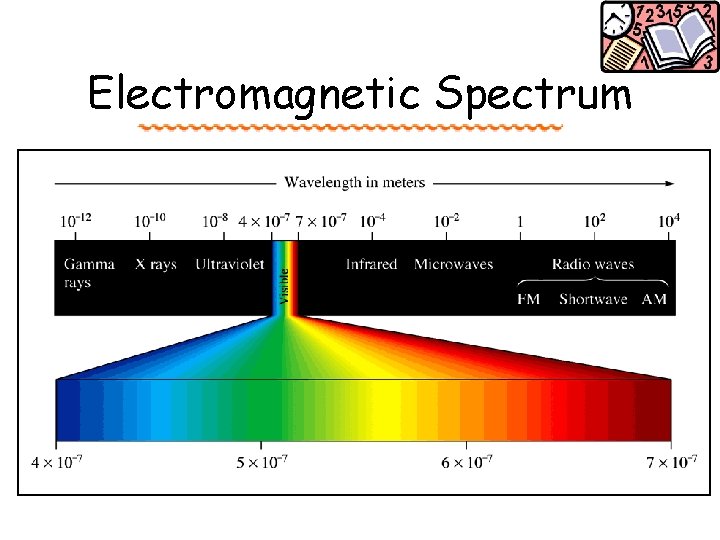
Waves Electromagnetic radiation. Comes in a range of energies called the energy spectrum. Radio Microwave IR Visible UV X-ray Gamma low high Energy these are all forms of traveling waves l = wavelength

Waves are characterized by their frequency (n) wavelength _______(l) and their _______ which is the number of times a like point (e. g. “a crest”) passes a stationary point in a given amount of time. nm Units -> wavelength: usually ___ frequency: s-1 or hertz (Hz)

Waves Wavelength (l) and frequency (n) are Speed of light related by the ______(c). c = 3. 00 x 108 m/s c=lxn inverse relation! note that this is an ______ http: //www. colorado. edu/physics/2000/waves_particles/wavpart 4. html low frequency high long wavelength short

http: //coolcosmos. ipac. caltech. edu/cosmic_classroom/cosmic_reference/emspec. html


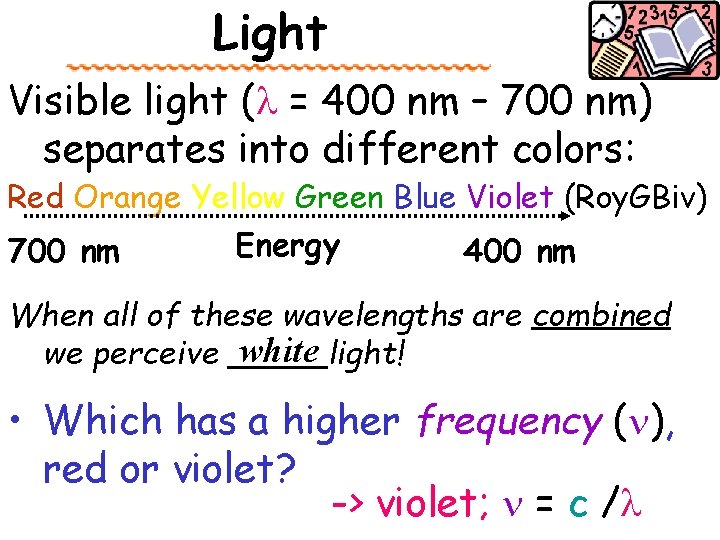
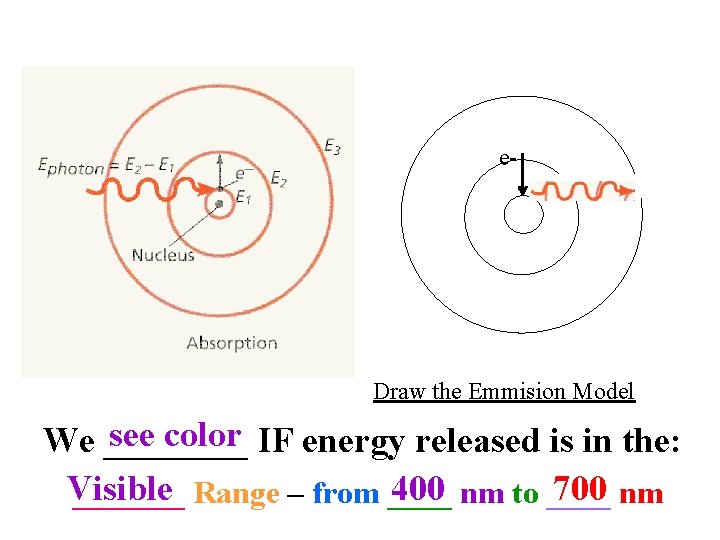
Light Visible light (l = 400 nm – 700 nm) separates into different colors: Red Orange Yellow Green Blue Violet (Roy. GBiv) Energy 700 nm 400 nm When all of these wavelengths are combined white we perceive _____light! • Which has a higher frequency (n), red or violet? -> violet; n = c /l

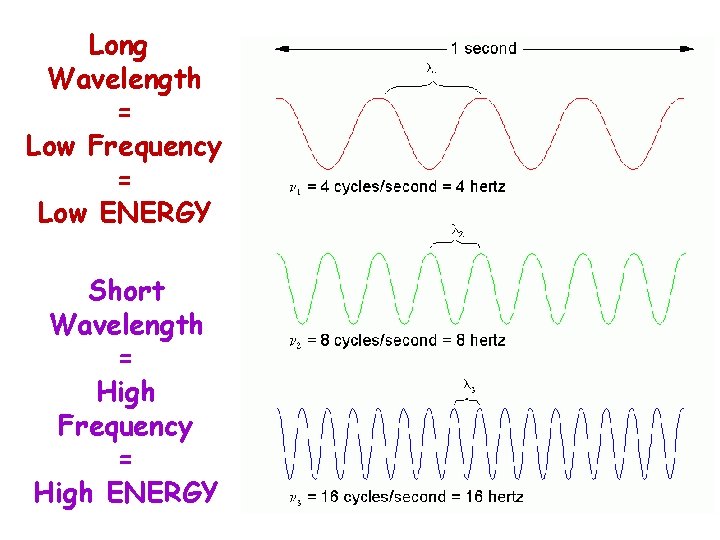
Long Wavelength = Low Frequency = Low ENERGY Short Wavelength = High Frequency = High ENERGY

Electromagnetic Spectrum

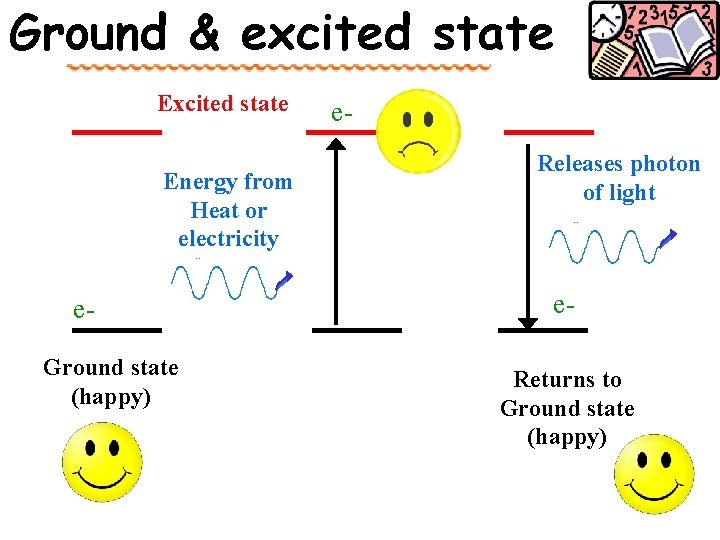
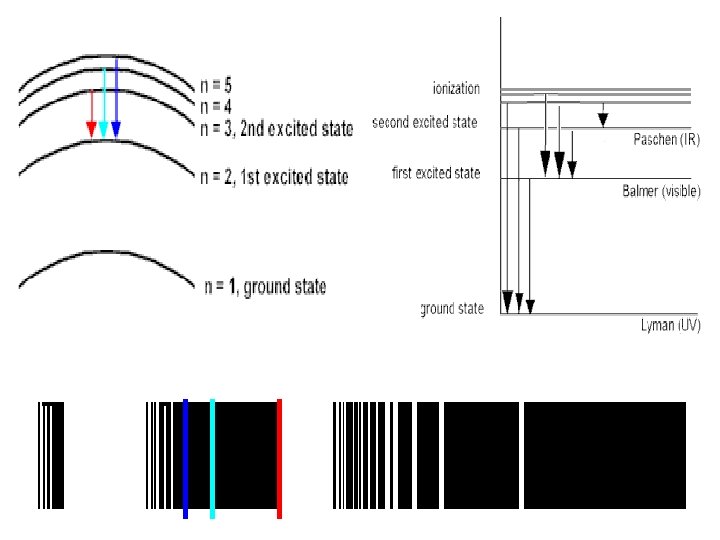
Atom Line-Emission Spectrum Ground state Lowest energy state of an atom. _______ Lazy We all want to be _____ Higher potential Excited state - ______ energy than ground state. Takes ____ Energy !! When an excited atom returns to gives off energy ground state, it _______ in form of electromagnetic radiation

Waves and Particles Understanding how electromagnetic radiation interacts with atoms has helped developed electrons are many theories about how the ____ organized and behave in atoms. Namely, how e- are arranged in shells. Electrons want to be in the lowest energy can be excited level (ground), but ________ to a higher state. releasing They return to ground level by ____ the energy.

Ground & excited state Energy from Heat or electricity e. Ground state (happy) e. Releases photon of light e. Returns to Ground state (happy)

e- Draw the Emmision Model see color IF energy released is in the: We ____ Visible 400 nm to ____ 700 nm _______ Range – from ____

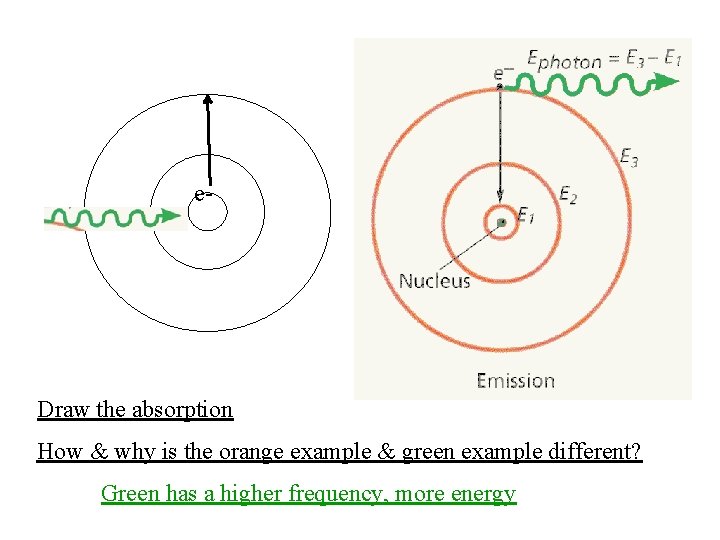
e- Draw the absorption How & why is the orange example & green example different? Green has a higher frequency, more energy

Light observe colored light when: We _______ only produces light 1) A source of light ______ of one (or a narrow range) of energies (e. g. , “neon” signs, flames, lasers). 2) removed One or more colors (energies) are _____ from white light by some substance that absorbs them (ex. Cobalt glass absorbs a specific wavelength and allows others to go through) White light Absorbs red White - red = green


Heisenberg Uncertainty Principle: cannot know the speed and position We ______ of the electron at the same time. If something is moving then its location is changing, and if something is not moving zero ! it’s speed is _____ Electron density distribution surrounding the nucleus in a H atom. There is a higher probability of finding an electron closer to the nucleus ! ____________

Quantum Number Each set of quantum numbers then describes: behavior for the • a pattern of ____ electron. • a region of space were the electron density can be found (electron _______).

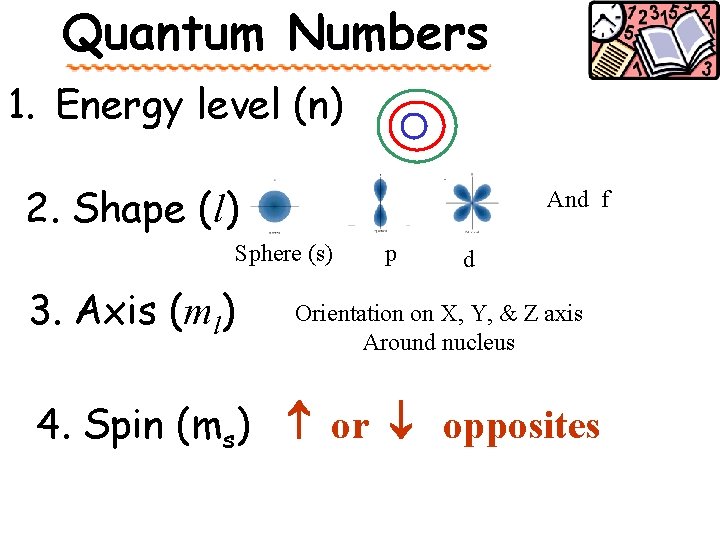
Quantum Numbers orbital The Chemist way of describing ____ & Predicting where an electron should be use 4 character addresses for each electron n, l, m, spin,

There are four quantum numbers : n, l, m, spin 1. Principal Q# (n) main energy level (or shell) • average distance from nucleus • the larger the n, the further away from table the nucleus (find this on Periodic ______ # 1 – 7) Total # of orbitals in a squared shell = n ____ Sublevels to follow ! 1 2 3 4

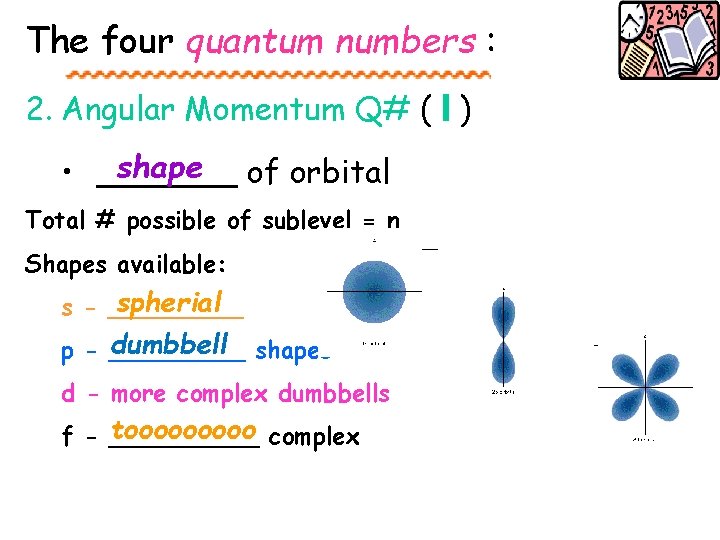
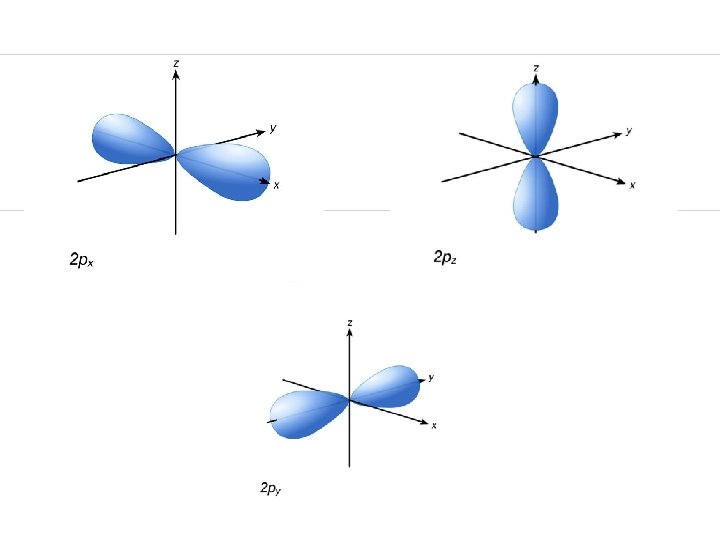
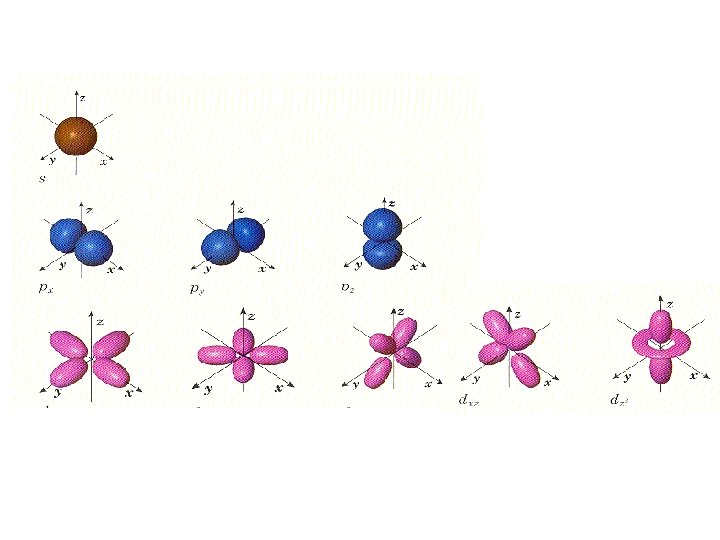
The four quantum numbers : 2. Angular Momentum Q# ( l ) shape of orbital • _______ Total # possible of sublevel = n Shapes available: spherial s - _____ dumbbell shapes p - _____ d - more complex dumbbells tooooo complex f - _____

The four quantum numbers : 3. Magnetic Q# (ml) • orientation of orbital shape around nucleus This is like on the axis of a 3 -dimensional graph _______



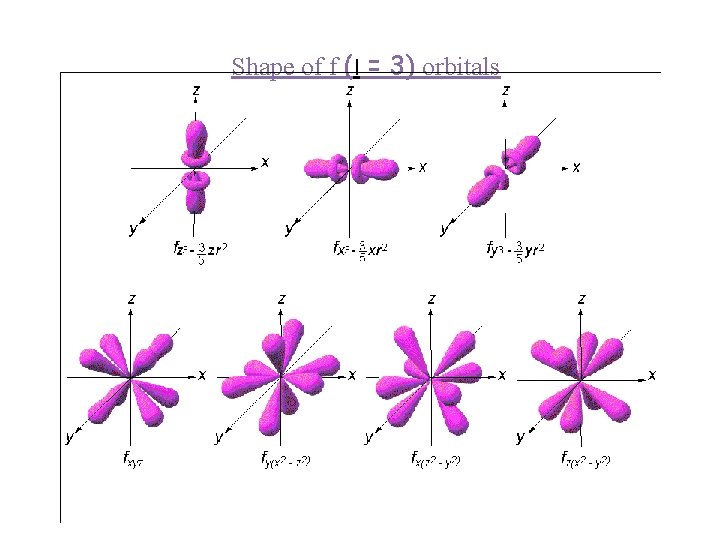
Shape of f (l = 3) orbitals


The four quantum numbers : 4. Spin electron spin Q# (ms). • ms can only equal +½ or -½. ↑ ↑ or A single orbital can hold a maximum OPPOSITE of two electrons in _____ spins

Quantum Numbers 1. Energy level (n) 2. Shape (l) And f Sphere (s) 3. Axis (ml) p d Orientation on X, Y, & Z axis Around nucleus 4. Spin (ms) or opposites

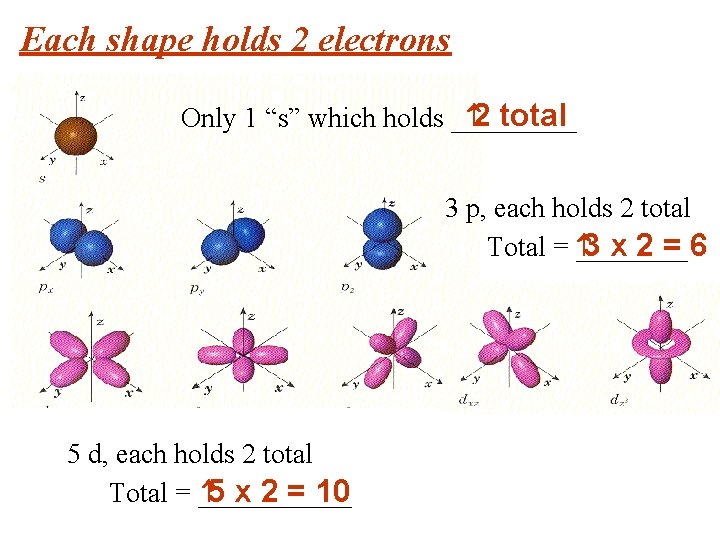
Each shape holds 2 electrons 2 total Only 1 “s” which holds _____ 3 p, each holds 2 total 3 x 2 = 6 Total = ____ 5 d, each holds 2 total 5 x 2 = 10 Total = ______

Subshells and Electrons The maximum number of electrons that can be placed in a subshell is determined by the number orbitals in that subshell. Subshell Orbitals Max. e- s 1 ↑ __ 2 ↑ __ ↑ ↑ ↑__ __ ↑ __ 5 __ 7_______ ↑ ↑ ↑ f 3 ↑ __ ↑__ ↑ ↑ ↑ d ↑ p 6 10 14

Quantum Number n 1 2 3 4 Subshells s s p d f # orbitals 1 1 3 5 7 Possible quantum number combinations Total # Orbitals n 2 12 = 1 # e- per main level 2 n 2 2(12) = 2 22 = 4 2(22) = 8 32 = 9 2(32) = 18 42 = 16 2(42) = 32

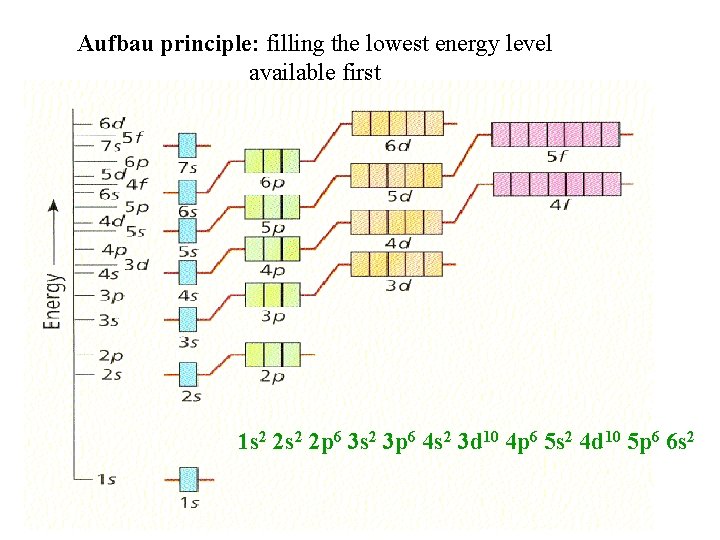
Electron Configuration: Arrangement of electrons in an atom Rules, rules, always rules: Aufbau principle: lowest possible energy Everyone wants to be in the ____ level - we don’t want to work. lowest to _____. highest So always fill from _______ Strange order need to follow with changes!! - 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6 5 s 2 4 d 10 5 p 6 6 s 2 Though you will write it in order of numbers The periodic table will help figure out order!

Aufbau principle: filling the lowest energy level available first 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6 5 s 2 4 d 10 5 p 6 6 s 2

Orbital filling table

Electron configuration seating chart model for filling sub shells and orbitals

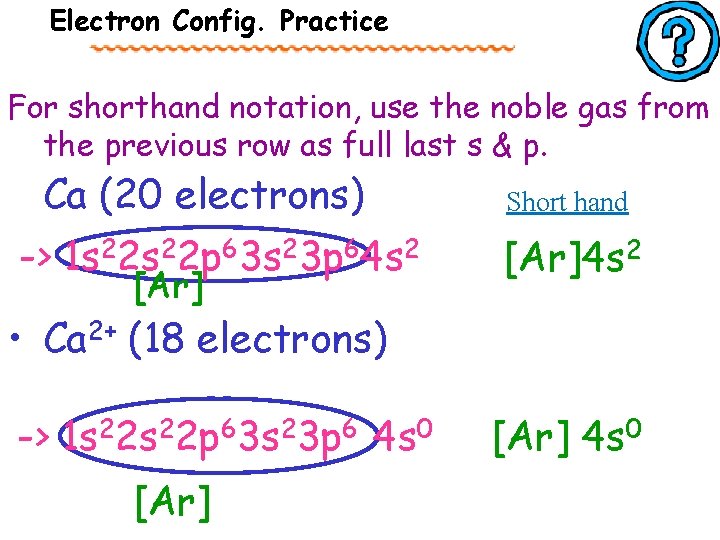
Electron Config. Practice For the following elements, write out their electron configuration and noble gas notation (short hand). Remove from s first • Ca (20 electrons) -> 1 s 22 p 63 s 23 p 64 s 2 • Ca 2+ (18 electrons) -> 1 s 22 p 63 s 23 p 64 s 0

Electron Config. Practice For shorthand notation, use the noble gas from the previous row as full last s & p. Ca (20 electrons) -> 1 s 22 p 63 s 23 p 64 s 2 [Ar] Short hand [Ar]4 s 2 • Ca 2+ (18 electrons) -> 1 s 22 p 63 s 23 p 6 4 s 0 [Ar] 4 s 0

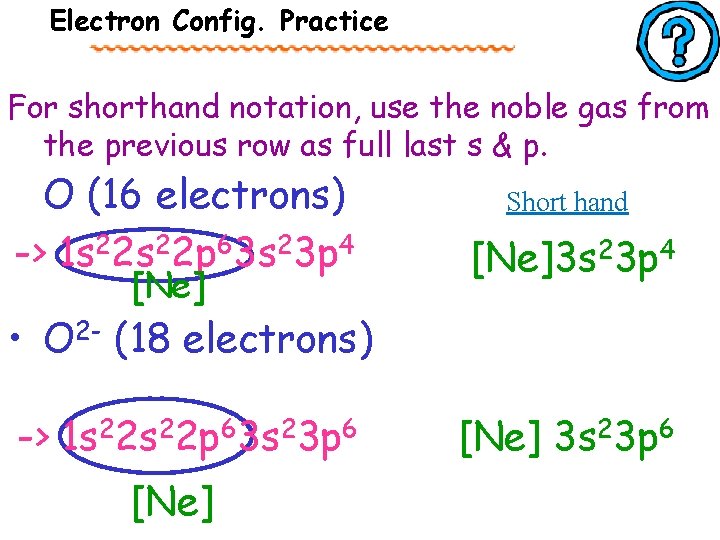
Electron Config. Practice For the following elements, write out their electron configuration and noble gas notation (short hand). • O (16 electrons) -> 1 s 22 p 63 s 23 p 4 • O 2 - (18 electrons) -> 1 s 22 p 63 s 23 p 6

Electron Config. Practice For shorthand notation, use the noble gas from the previous row as full last s & p. O (16 electrons) -> 1 s 22 p 63 s 23 p 4 [Ne] Short hand [Ne]3 s 23 p 4 • O 2 - (18 electrons) -> 1 s 22 p 63 s 23 p 6 [Ne] 3 s 23 p 6

Electron Config. Practice For the following elements, write out their electron configuration and noble gas notation (short hand). • Fe (26 electrons) -> 1 s 22 p 63 s 23 p 64 s 23 d 6 • Fe 2+ (24 electrons) -> 1 s 22 p 63 s 23 p 64 s 03 d 6 • Fe 2+ (23 electrons) -> 1 s 22 p 63 s 23 p 64 s 03 d 5

Electron Config. Practice Remove from s 1 st • Fe (26 electrons) 23 d 6 2 2 6 2 6 [Ar]4 s -> 1 s 2 s 2 p 3 s 3 p 4 s 3 d [Ar] 03 d 6 [Ar] 4 s • Fe 2+ (24 electrons) -> 1 s 22 p 63 s 23 p 6 4 s 03 d 6 [Ar] • Fe 2+ (24 electrons) [Ar] 4 s 03 d 5 -> 1 s 22 p 63 s 23 p 6 4 s 03 d 5

Pauli Exclusion Principle The Pauli Exclusion Principle states that no two electrons can have the same set of four quantum numbers [n, l, ms] in an atom. Another way to say this is that no orbital can contain more than two electrons and their spins must be opposed ( ).

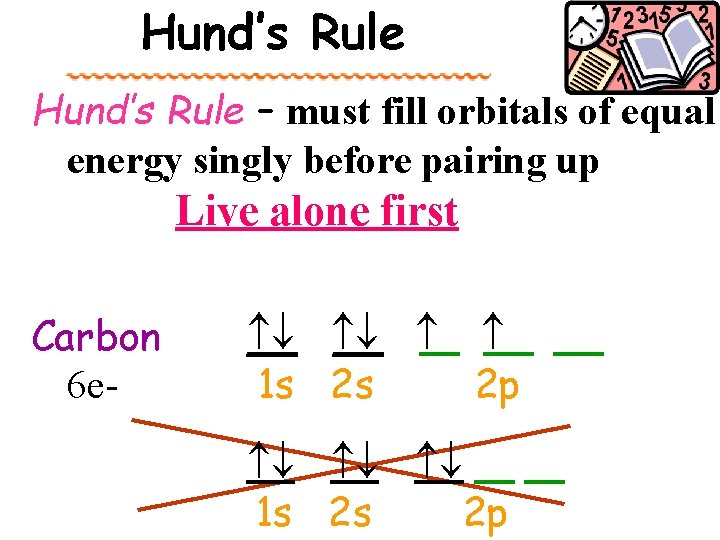
Hund’s Rule – must fill orbitals of equal energy singly before pairing up Live alone first Carbon 6 e- __ __ 1 s 2 s 2 p

Orbital Diagrams Orbital Notation – orbitals are drawn as lines, and electrons as ( ) spins. Hydrogen -- one e- is placed in 1 s orbital 1 s 1 1 s electron configuration: ____ Lithium --two e- in 1 s; one e- in 2 s orbital 1 s 2 s 22 s 1 1 s electron configuration: _____

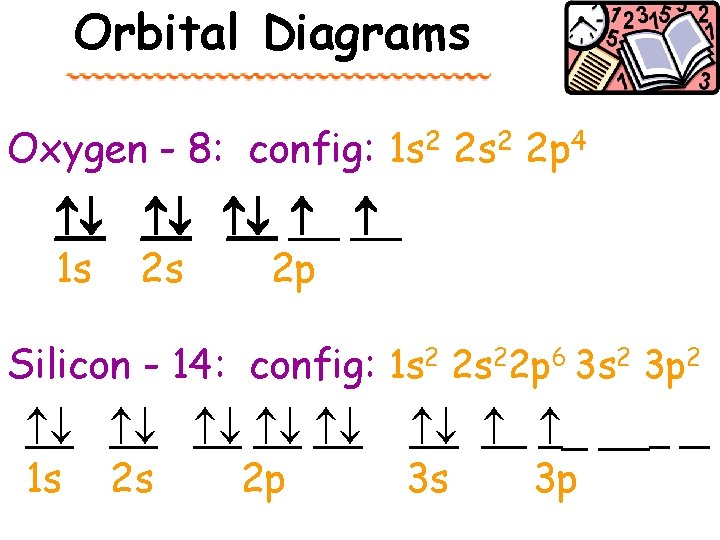
Orbital Diagrams Oxygen - 8: config: 1 s 2 2 p 4 __ __ __ 1 s 2 s 2 p Silicon - 14: config: 1 s 2 2 s 22 p 6 3 s 2 3 p 2 1 s 2 s 2 p _ __ 3 s 3 p

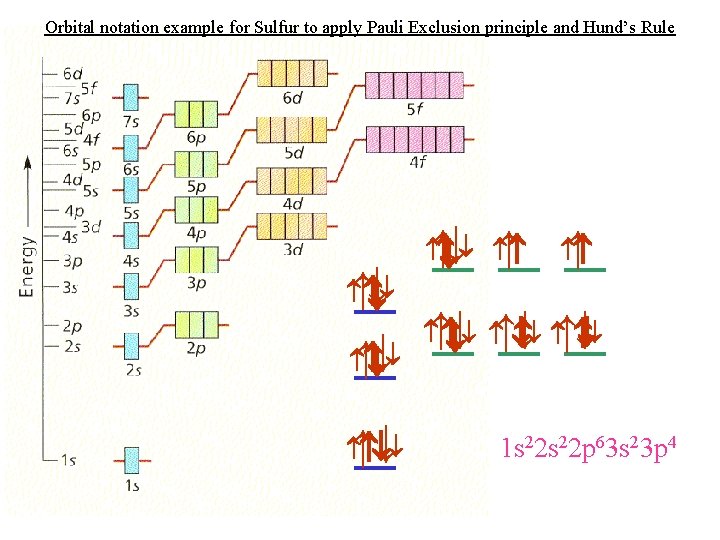
Orbital notation example for Sulfur to apply Pauli Exclusion principle and Hund’s Rule ↑ ↑ ↑ ↑ ↑ ↑ ↑ 1 s 22 p 63 s 23 p 4

Bohr model for Sulfur Neutral Sulfur has 16 protons & 16 electrons Neutrons = 32 – 16 = 16 neutrons 16 p 16 n 1 s 22 p 63 s 23 p 4 2 e 8 e 6 e

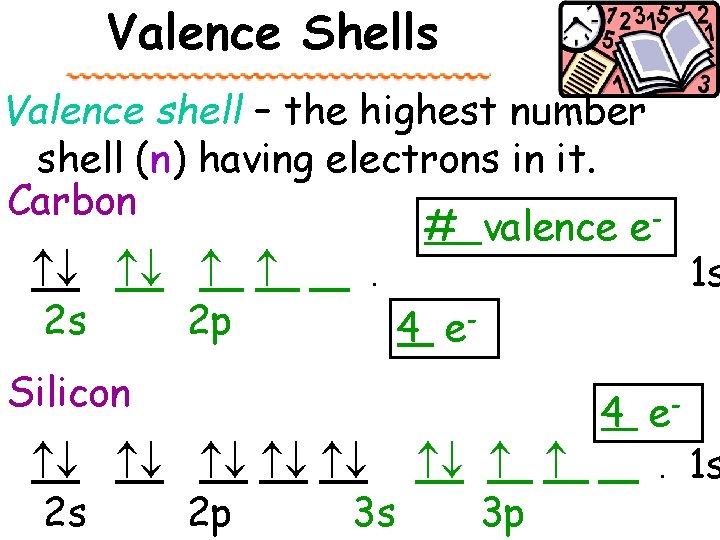
Valence Shells Valence electrons are located in an outermost energy level (n), atom’s ____ the valence shell. In chemistry, most important these are the ________ electrons because they are the ones involved in bonds! The number of electrons in the valence shell is the total number in that shell. the s & p subshells of _____

Valence Shells Valence shell – the highest number shell (n) having electrons in it. Carbon # valence e . 1 s 2 s 2 p 4 e. Silicon 4 e . 1 s 2 s 2 p 3 s 3 p

Valence Shells Circle the valence shell in the following electron configurations: • Cl (#17) 1 s 22 p 6(3 s 23 p 5 ) • Ar (#18) 1 s 22 p 6(3 s 23 p 6 ) • K (#19) 1 s 22 p 63 s 23 p 6(4 s 1 ) • Ca (#20) 1 s 22 p 63 s 23 p 6 (4 s 2 ) • Sc (#21) 1 s 22 p 63 s 23 p 6(4 s 2 )3 d 1

Valence electrons & Being “Happy” LAST s & p • Atom wants to have their _____ orbitals full Octet • Then you have a FULL _______ s has 2 electrons & p has 6, so 2 + 6 = 8 (octet) • Noble gases are all happy as they have full except for He with ____) only 2 octets (_______ • Everyone wants to be like a Noble Gas ! Lose or gain – So they will _______electrons to do it !

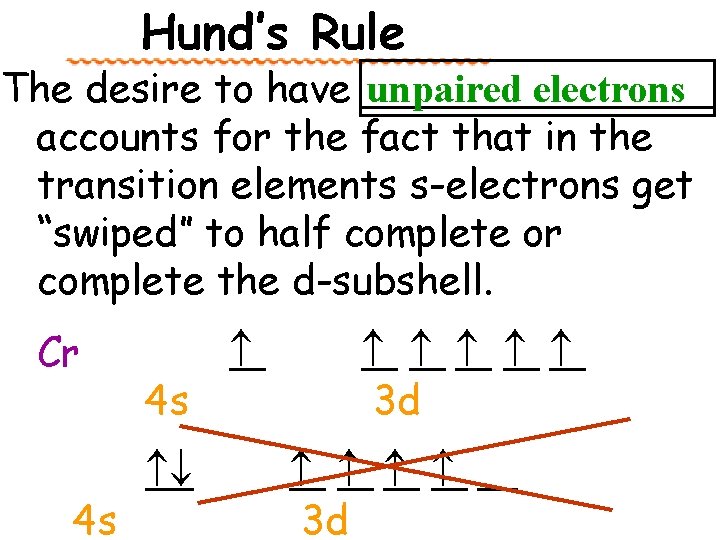
Hund’s Rule The desire to have _______ unpaired electrons accounts for the fact that in the transition elements s-electrons get “swiped” to half complete or complete the d-subshell. Cr 4 s 4 s 3 d