ATOMIC THEORY REVIEW VOCABULARY AND ATOMIC STRUCTURE UNIT

ATOMIC THEORY REVIEW, VOCABULARY, AND ATOMIC STRUCTURE

UNIT THREE – REMINDER OF WHAT WE GET TO COVER! • • Atomic Theory – Progression from Dalton to Heisenberg Atoms, Ions, and Sub-atomic Structure Periodic Table – History, Trends, Organisation Bonding, Naming, Structures

ASSESSMENT FOR THE UNIT? • Current thoughts: • Some of you love tests, some of you HATE! tests • Some of you love projects, some of you HATE! Projects • I am willing to consider both for this unit • People who want a project MUST have an idea approved by me within the NEXT TWO classes. Small groups are a possible option. • I reserve the right to cancel this idea if a better one presents itself! • Other suggestions? • This is your chance to give feedback and suggestions!

REVIEW OF LAST LESSON – ATOMIC THEORY FROM DALTON TO HEISENBERG • DALTON • Billiard Ball model, first described in 1803 • Atoms are tiny indivisible spheres, the hydrogen atom is the smallest known unit • Moved Democritus’ idea from hypothesis to an evidence based theory • Experiments with gasses • Law of Definite Proportions & Law of Partial Pressures • Atoms combine in whole number ratios ONLY!

REVIEW OF LAST LESSON – ATOMIC THEORY FROM DALTON TO HEISENBERG • Thomson • Plum Pudding model, described in 1904 • Introduced the idea of sub-atomic particles • Discovered the corpuscle (later renamed the electron (e-) ) • Evidence of a particle 1/1000 th the mass of Hydrogen • Cathode ray tube experiment – Deflected a stream of e-

REVIEW OF LAST LESSON – ATOMIC THEORY FROM DALTON TO HEISENBERG • Rutherford model or planetary model, 1911 • Discovered that atoms are mostly empty space, with a small dense nucleus that was positively charged • Directed the Marsden-Geiger Gold foil experiment

REVIEW OF LAST LESSON – ATOMIC THEORY FROM DALTON TO HEISENBERG • Bohr model or Rutherford-Bohr model, 1913 • Added a quantum physical interpretation to the Rutherford model of the atom. Introduced the idea of QUANTISED e- states (discrete, fixed orbits) • Used the Rydberg formula to explain the spectral emission and absorption of Hydrogen Electrons orbit the nucleus in orbits that have a set size & energy • The energy of the orbit is related to its size, lowest energy in the smallest orbit • Radiation is absorbed or emitted when an electron moves from one orbit to another

REVIEW OF LAST LESSON – ATOMIC THEORY FROM DALTON TO HEISENBERG • Schrӧdinger • Quantum mechanical model, 1926 • Orbitals would create a standing wave, therefore electrons do not have a set path • Electrons exist in orbitals, a space where electrons can be predicted to be found

REVIEW OF LAST LESSON – ATOMIC THEORY FROM DALTON TO HEISENBERG • Heisenberg • Electron cloud model, 1927 • Developed the Uncertainty Principle • It is not possible to know both the position & momentum of a particle • Electrons can only be described through probability distributions

REVIEW OF LAST LESSON – ATOMIC THEORY FROM DALTON TO HEISENBERG • Chadwick (Jimmy Neutron) • Added the neutron to the model of the atom to FINALLY! explain atomic mass, 1932 • Measured the results of showering elements with alpha particles, first successful in creating nuclear fission in 1919. • Ultimately his work led to the creation of the atomic bomb (fission of Uranium-235)

KEY UNIT VOCABULARY • Divide the following words into these categories: • I know this! • I think I know this (Maybe? ) • Wut even? !?

UNIT VOCABULARY TERMS: • • • Sub-atomic particles Atom Element Molecule Ion Compound Proton Neutron Valence (electron) • • Electron Isotope Atomic Mass Atomic Number Metal Non-metal Metalloid Polarity Extra Words • Quantised • Quark • Lepton • Nucleon • Guage Boson • Higgs Boson Once you have your words sorted, compare with your table or make a group of 4. Focus on explaining words you know, but others don’t. Then work together to discover the meanings of words that are new to everyone

VOCABULARY • Sub-atomic particle - particles smaller than atoms of which there are two kinds: elementary and composite particles • Atom - the smallest component of an element having the chemical properties of the element • Element – one of a class of substances that cannot be separated into simpler substances by chemical means • Molecule – the smallest physical unit of an element or compound, consisting of one or more like atoms in an element and two or more different atoms in a compound • Compound - composed of two or more parts, or ingredients • Proton - a positively charged elementary particle that is a fundamental constituent of all atomic nuclei • Neutron - an elementary particle having no charge • Electron - an elementary particle that is a fundamental constituent of matter, having a negative charge

VOCABULARY • Isotope - any of two or more forms of a chemical element having the same atomic number, but having different atomic weights. • Quark – believed to constitute all the elementary particles classed as baryons and mesons; they are distinguished by their flavors, up, down, strange, charm, bottom, and top, and their colours, red, green, and blue. • Lepton - not subject to the strong force and are believed to be truly elementary and not composed of other subunits • Nucleon - a sub-atomic particle that comprises part of the nucleus • Guage Boson - a boson that mediates the interaction between elementary particles • Higgs Boson - a type of heavy, electrically neutral particle with zero spin

BUILDING ATOMS! • Be careful not split them… • Everyone will need a lap top, phones don’t work well (a tablet might…) • Google search: phet build an atom • It should be the 1 st option • Click on the “play” arrow to load • Select atom • Wait patiently until we are all at the same screen, please move quickly • Remember there’s candy in the NEXT part of the lesson

BUILDING ATOMS! Ensure you are on the ORBIT model AND You must check off the STABLE/UNSTABLE box
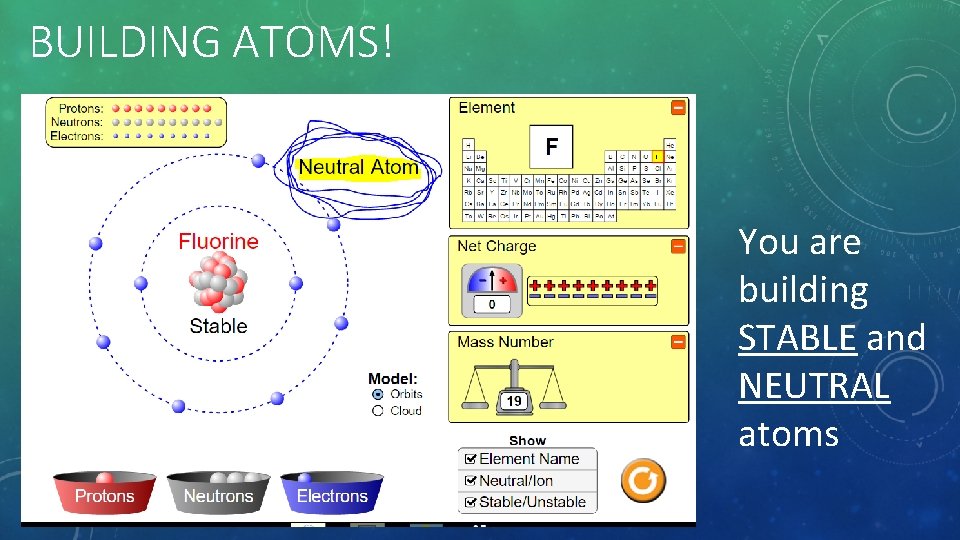
BUILDING ATOMS! You are building STABLE and NEUTRAL atoms

BUILDING ATOMS! Complete the Three atoms on your sheet Experiment with creating plausible ions Use the periodic table, google, etc. Once you have completed this, investigate the first ten elements and look at both orbit mode and cloud mode Apply the vocabulary words we worked on earlier! When everyone is ready we will move on

EDIBLE BOHR MODELS • We have: • BERRY protons, • JU JUBE neutrons and • SKITTLE electrons!!! • We will create: • Lithium and Beryllium • Add to your definitions of atomic mass, isotopes, ion, atomic number and others
- Slides: 19