Atomic Theory Review Basic Atomic Structure A Look

Atomic Theory Review Basic Atomic Structure: A Look Inside the Atom Watch the Video Linked above Before starting these notes.

Atomic Theory & Bonding What is an Atom? • smallest particle of an element that still has the properties of that element • An atom = proton(s) + electron(s) + neutron(s) (PEN) • Fun Fact: 50 million atoms, lined up end-to-end = 1 cm

An ELEMENT is made up of one type of atom • The element, oxygen, is O • IONS are atoms with a charge. • The ion of oxygen is O 2 - • Atoms can join together to form MOLECULES. • The oxygen molecules are O 2 • COMPOUNDS are made up of several (2+)atoms • Hydrogen and oxygen are atoms/elements • H 2 O is a compound

Structure of an Atom: The nucleus is at the centre of an atom • composed of protons (positive charge) and neutrons (neutral charge) Electrons (negative charge) exist in the space surrounding the nucleus

Subatomic Particles: • Atoms are made up of smaller particles known as sub-atomic particles. Name Symbol Charge Location Atomic Mass Proton p 1+ nucleus 1 AMU Neutron n 0 nucleus 1 AMU Electron e 1– area surrounding the nucleus 1/1836 (0)

Families of the Periodic Table: • Columns of elements are called groups, or families • All elements in a family have… • similar properties • bond with other elements in similar ways • have the same number of valence electrons • Family names (on the periodic table!): • Group 1 = alkali metals • Group 2 = alkaline earth metals • Group 17 = the halogens • Group 18 = noble gases • Periods are horizontal rows on the periodic table
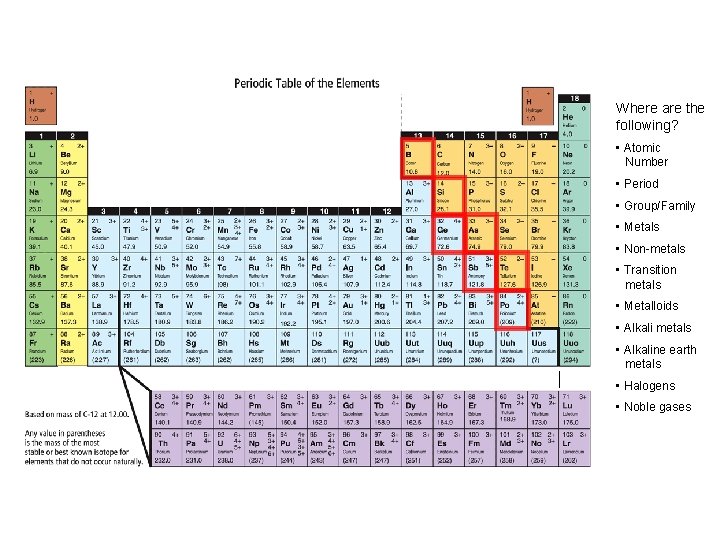
INCREASING REACTIVITY Where are the following? • Atomic Number • Period • Group/Family • Metals • Non-metals • Transition metals • Metalloids • Alkali metals • Alkaline earth metals • Halogens • Noble gases

Periodic Table & Ion Formation: • Atoms gain and lose electrons to form ions • Metals lose electrons & become positive ions (cations) • Some metals can have more than one charge (multivalent) • ie. Iron, Fe, loses either 2 (Fe 2+) or 3 (Fe 3+) electrons • Non-metals gain electrons & become negative ions (anions) • Atoms do this in an attempt to have the same number of valence electrons as the nearest noble gas – to become stable
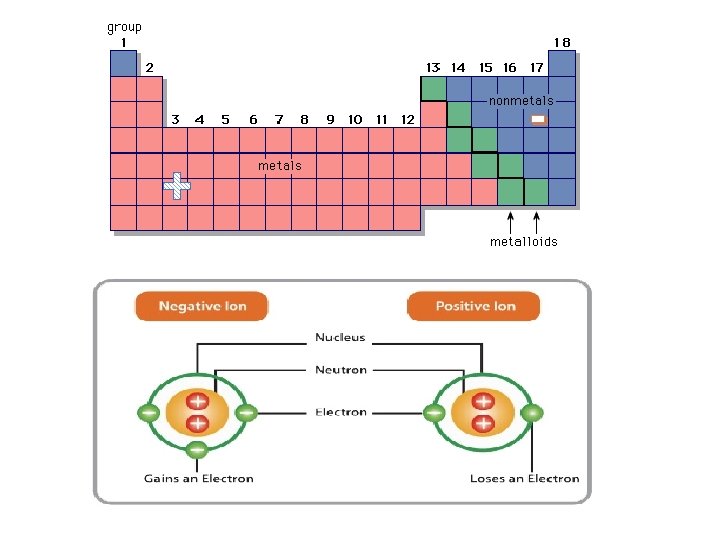

Numbers to Remember : Atomic number = # of protons = # of electrons in atoms In ATOMS: # of protons = # of electrons in every atom AND ONLY IN ATOMS!!! (not ions) *this keeps the atom neutral since protons are (+)ive and electrons are (-)ive Positive Ions Negative Ions Metals generally form positive Ions Non-Metals generally form negative Ions They have only a few electrons in their valence so they tend to lose electrons to be “happy” They have almost a full valence so they tent to gain electrons to be “happy”
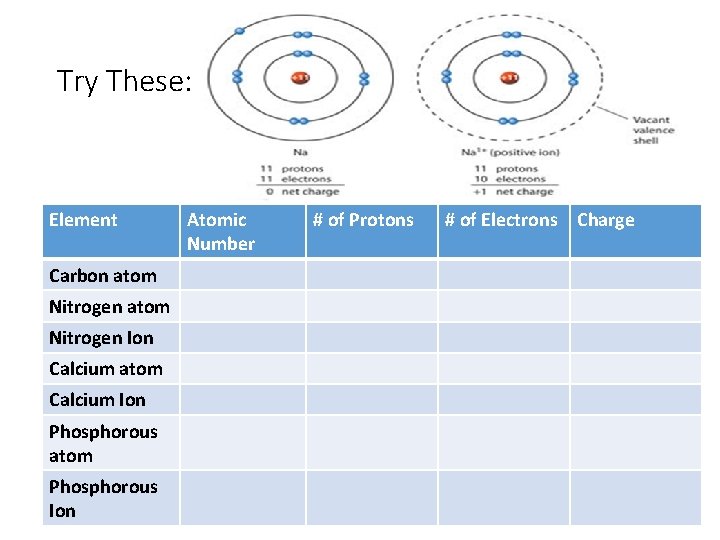
Try These: Element Carbon atom Nitrogen Ion Calcium atom Calcium Ion Phosphorous atom Phosphorous Ion Atomic Number # of Protons # of Electrons Charge
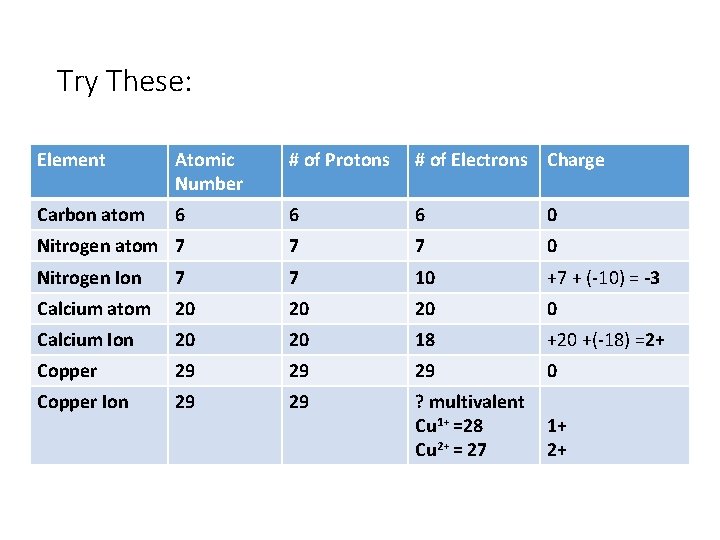
Try These: Element Atomic Number # of Protons # of Electrons Charge Carbon atom 6 6 6 0 Nitrogen atom 7 7 7 0 Nitrogen Ion 7 7 10 +7 + (-10) = -3 Calcium atom 20 20 20 0 Calcium Ion 20 20 18 +20 +(-18) =2+ Copper 29 29 29 0 Copper Ion 29 29 ? multivalent Cu 1+ =28 Cu 2+ = 27 1+ 2+

You are doing Great! Lets Keep Going Mass Number = The mass number is the ACTUAL mass of 1 atom = #Protons + # Neutrons (since both have mass) Atomic Mass = This is AVERAGE mass of the ISOTOPES of an element • Found on the periodic table and generally has a decimal. • This would be impossible in realty since the mass of an atom comes from protons and neutrons that each weight 1 amu and you can’t have part or a proton or neutron BUT it is also impossible to take a sample of an element and have only 1 atom! That same will contain many atoms and will contain a variety of the isotopes of the atoms of that element! Isotope = Same element (same atomic number / # of protons) = Different mass number because Different # of NEUTRONS
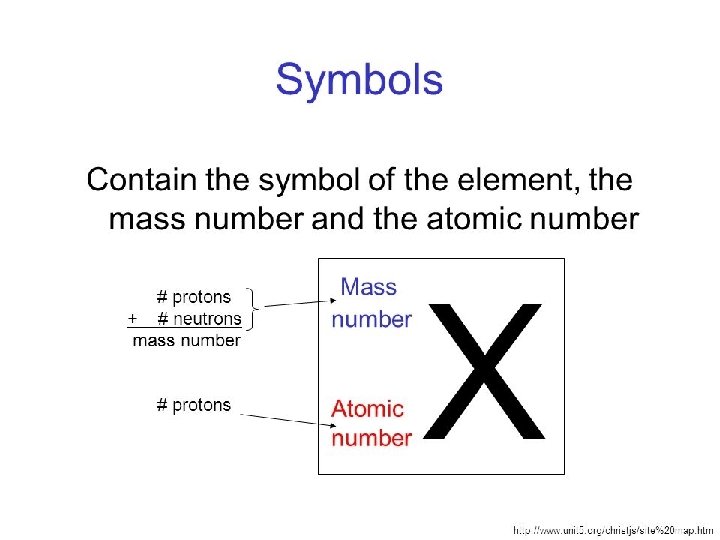
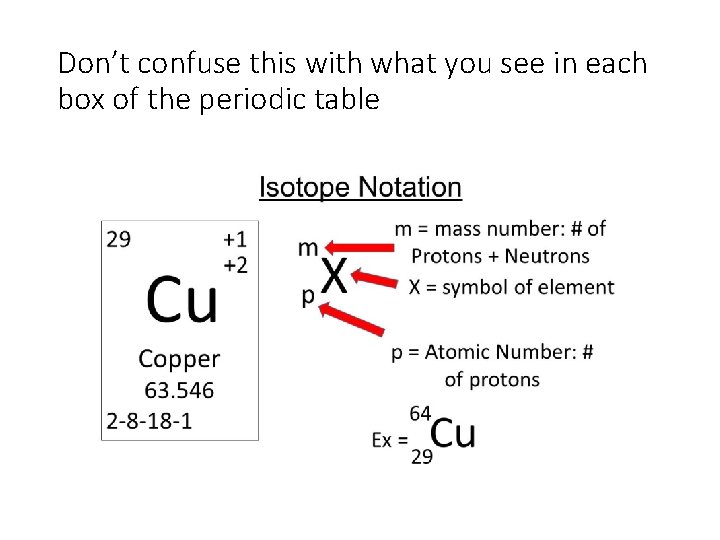
Don’t confuse this with what you see in each box of the periodic table

Practice: • Determine the number of protons neutrons and electrons for the symbol above? • If an element has 15 protons 17 neutrons and 18 electrons, write is isotope notation below?

Practice: • Determine the number of protons neutrons and electrons for the symbol above? • Protons = 29 (bottom number is atomic number) • Neutrons = 35 (64 -29 = 35 since top number is mass number) • Electrons = 29 ( no charge was indicated for P = e- ) • If an element has 9 protons 10 neutrons and 10 electrons, write is isotope notation below? -1
If you are confused – Video Links Below • Difference between Mass Number and Atomic Mass • https: //www. youtube. com/watch? v=m 15 DWkk. Ge_0 • Isotopes Explained • https: //www. youtube. com/watch? v=Ebo. We. Wmh 5 Pg • Isotope Notation and Practice • https: //www. youtube. com/watch? v=BYiu 0 k. IWd 30 Otherwise Watch this before Moving on • Valence Electrons and the periodic Table • https: //www. youtube. com/watch? v=y. ADr. Wd. NTWEc

Bohr Diagrams: § Bohr diagrams show many electrons appear in each electron shell around an atom. § Each shell holds a maximum number of electrons (2, 8, 8, 18) § Electrons in the outermost shell are called valence electrons § Except for the transition elements, the last digit of the group # = # of valence electrons Note: Think of the shells as being 3 D like spheres, not 2 D like circles!
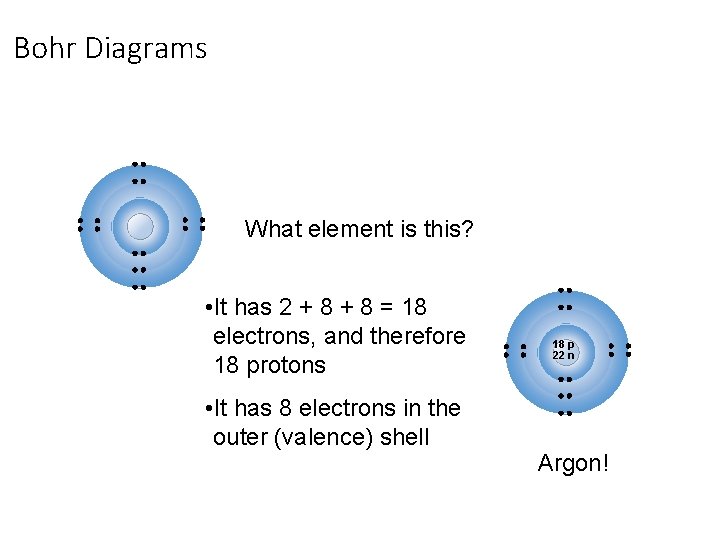
Bohr Diagrams What element is this? • It has 2 + 8 = 18 electrons, and therefore 18 protons • It has 8 electrons in the outer (valence) shell 18 p 22 n Argon!
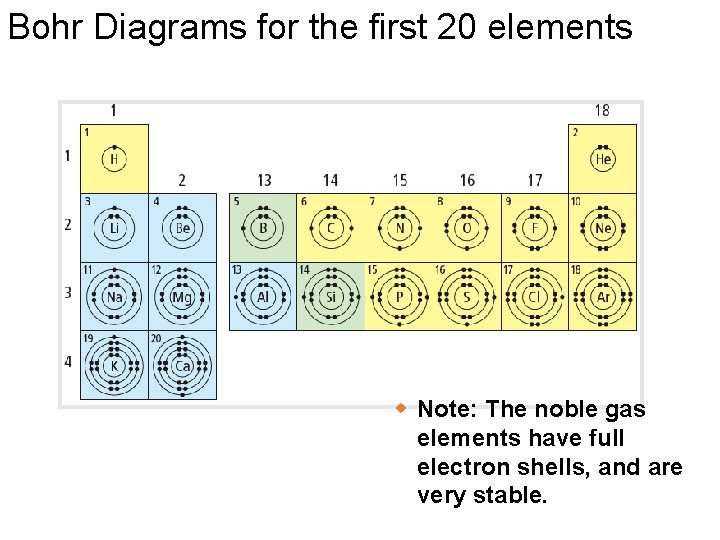
Bohr Diagrams for the first 20 elements w Note: The noble gas elements have full electron shells, and are very stable.

Bonding Types: When two atoms get close together, their valence electrons interact. • Recall that Ions form by the atom gaining or losing electrons to “fill or empty” the valence. • Metal Elements (left) “want” to lose electrons and Non-Metal Elements “want” to gain electrons • So if two Non-Metals meet at a bar? . . . but really, they both want more electrons so they make an agreement to SHARE and then they are both happy.
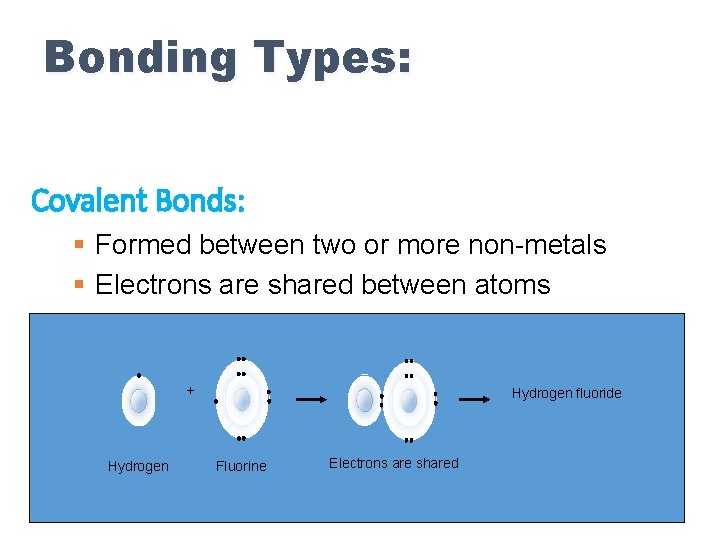
Bonding Types: Covalent Bonds: § Formed between two or more non-metals § Electrons are shared between atoms + Hydrogen fluoride Fluorine Electrons are shared

Bonding Types: When two atoms get close together, their valence electrons interact. • Ok so what if two metals walk into a bar? Well there would be a bar fight between the metals! • Metals “need” to get rid of electrons and there would be no way for them to strike a mutual deal • BUT if a Metal were to meet a Non-Metal at the bar it would be love at first sight. Well not quite, but they would be able to make each other happy since one “wants” to gain and the other “wants” to lose electrons
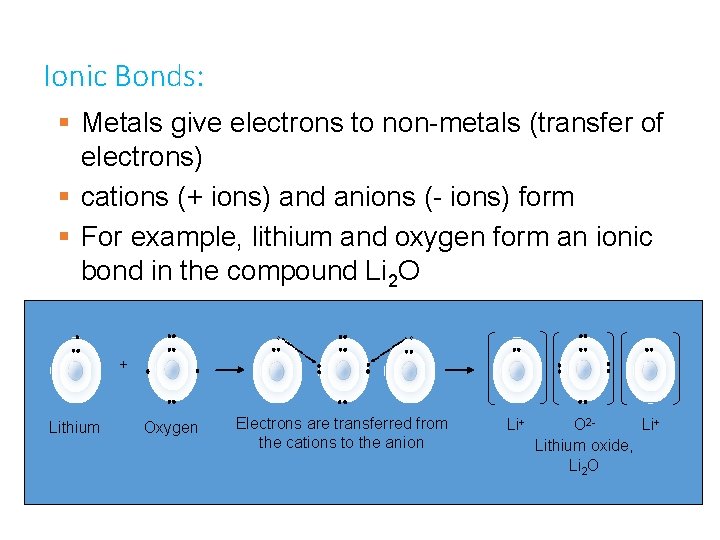
Ionic Bonds: § Metals give electrons to non-metals (transfer of electrons) § cations (+ ions) and anions (- ions) form § For example, lithium and oxygen form an ionic bond in the compound Li 2 O + Lithium Oxygen Electrons are transferred from the cations to the anion Li+ O 2 Li+ Lithium oxide, Li 2 O

Lewis Diagrams: • Like simplified Bohr diagrams • Only valence electrons are shown • Dots representing electrons are placed around the element symbols (on 4 sides, imagine a box around the symbol) • Electron dots are placed singularly, until the fifth electron is reached, then they are paired. Example: Nitrogen atom
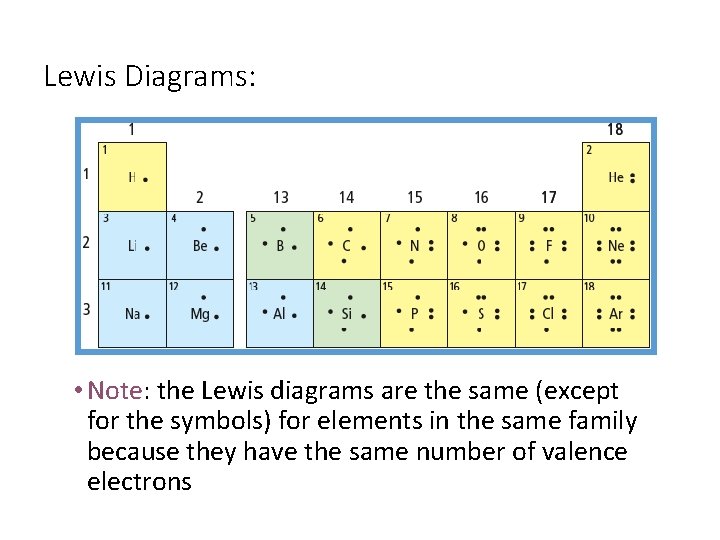
Lewis Diagrams: • Note: the Lewis diagrams are the same (except for the symbols) for elements in the same family because they have the same number of valence electrons

Lewis Diagrams for Ions: • For positive ions: one electron dot is removed from the valence shell for each positive charge of the ion. • For negative ions: one electron dot is added to each valence shell for each negative charge of the ion. • Square brackets and the charge are placed around each ion Example: Nitrogen ion
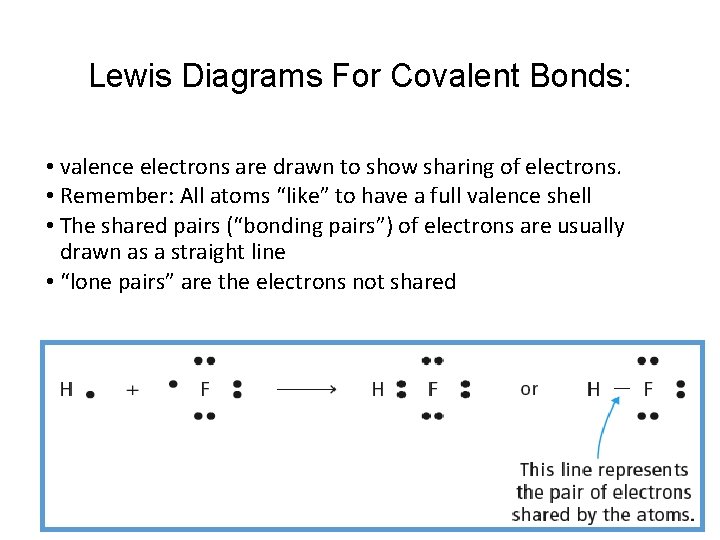
Lewis Diagrams For Covalent Bonds: • valence electrons are drawn to show sharing of electrons. • Remember: All atoms “like” to have a full valence shell • The shared pairs (“bonding pairs”) of electrons are usually drawn as a straight line • “lone pairs” are the electrons not shared

Lewis Diagrams For Ionic Bonds: • • • • • Be • • • • • Cl • • • • 2+ • Be • • • Cl Each beryllium has two Since Be wants to electrons to transfer donate 2 electrons and away, and each Cl– wants to chlorine wants one accept only one, two Cl – ions are necessary more electron • • – • • • • Cl • • • • • Be 2+ • • • • • Cl The ionic compound Beryllium chloride is formed –

Online Quiz via Classroom before next class!
- Slides: 31