Atomic Theory Models Bohr 1913 Subatomic Particles Mass
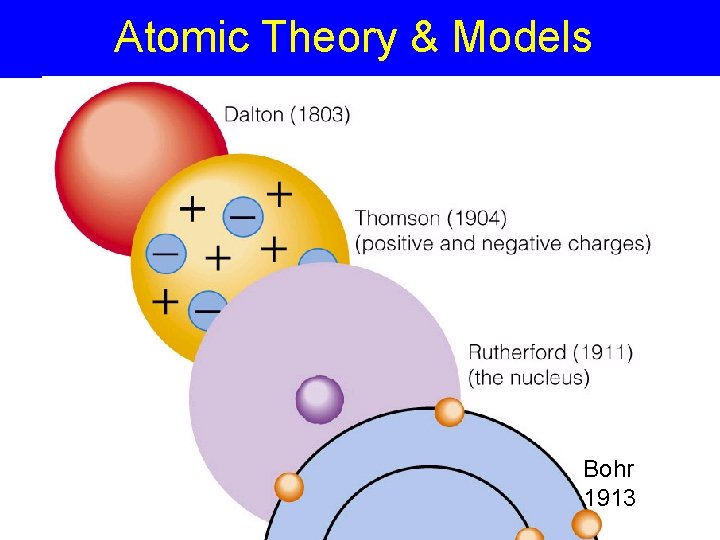
Atomic Theory & Models Bohr 1913

Subatomic Particles Mass Particle Symbol (amu) Proton p+1 1 amu Location Inside Nucleus Neutron n 0 1 amu Inside Nucleus Electron e-1 0 amu Outside Nucleus (Mass of e-1 actually 1/1836 amu) ________________________________ amu = atomic mass unit 1 amu = 1/12 th the mass of a Carbon-12 atom

Atomic Theory & Models Atom – Greek “atomos” = indivisible - smallest particle of an element John Dalton’s Model – 1803 - Atoms are solid spheres

Radioactive Particles Alpha, Beta, and Gamma (first 3 Greek letters). Alpha particles are helium nuclei (2 p+, 2 n 0). Particle Alpha Beta Gamma Description positive, mass = 4 amu neg. , very small; less than 1 amu energy ray; no mass; no charge

Atomic Theory & Models J. J. Thomson – - Atoms have particles - Used Cathode ray tubes (demo) http: //www. chem. uiuc. edu/clcwebsite/cathode. html -1897 discovered electron (= particles in cathode ray). - Revised atom model = “plum pudding model” with raisins of electrons + Electron ray e-

Ernest Rutherford 1911 Gold Foil Experiment http: //micro. magnet. fsu. edu/electromag/java/rut herford/ http: //www. mhhe. com/physsci/chemistry/essenti alchemistry/flash/ruther 14. swf

Rutherford’s Model of the Atom Ernest Rutherford 1911 Gold Foil Experiment Rutherford Model - discovery of dense nucleus with a positive (+) charge +

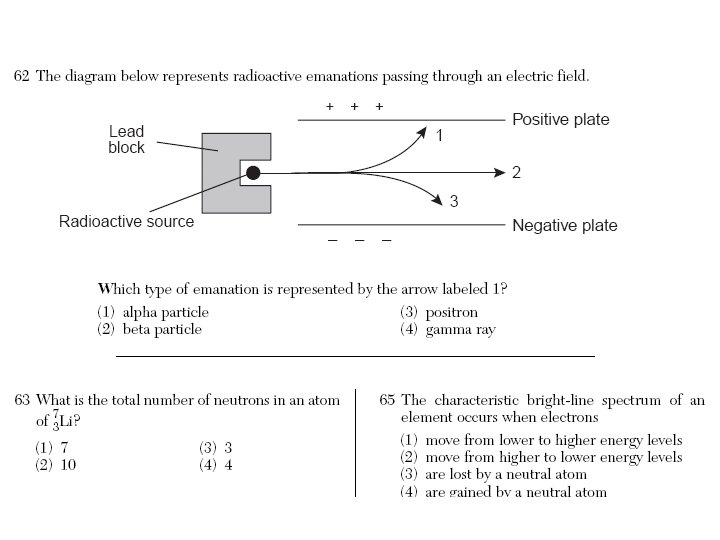
Rutherford’s Experiment - 1911 Observation 1) Most of alpha (+) particles pass thru undeflected. 2) Some alpha (+) particles bounced back; repelled. Conclusion 1) Most of atom is open space. 2) Dense nucleus (most of mass of atom) with a positive charge (same charge at alpha particles).

Bohr Model of Atom - 1913 Niels Bohr - electrons are found in energy levels, different energy layers or shells Nucleus & Energy levels

Modern Charge Cloud Model (1926) The charge-cloud model, also called the quantum-mechanical model: No Fixed Orbit of Electrons - does not attempt to describe the path of each electron in a fixed orbit. Fuzzy Cloud for Electrons = possible positions of electrons in terms of probability.
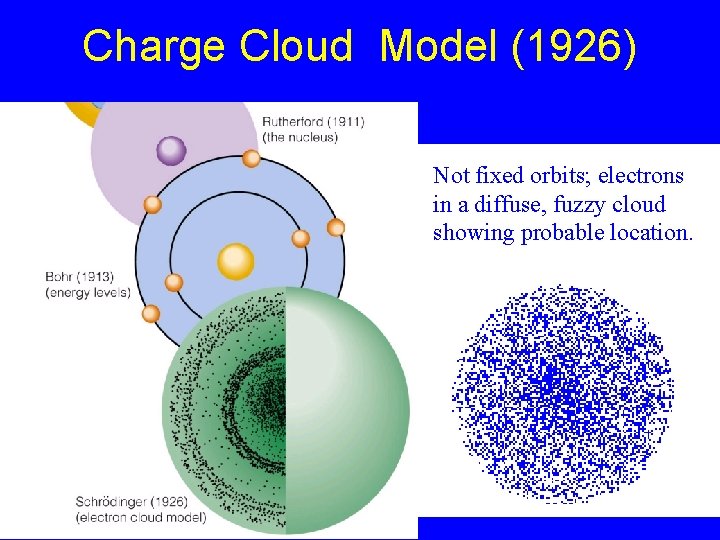
Charge Cloud Model (1926) Not fixed orbits; electrons in a diffuse, fuzzy cloud showing probable location.
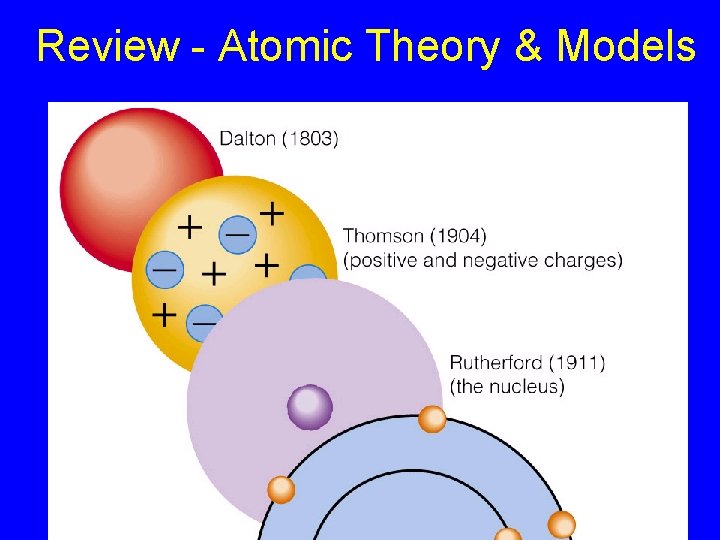
Review - Atomic Theory & Models
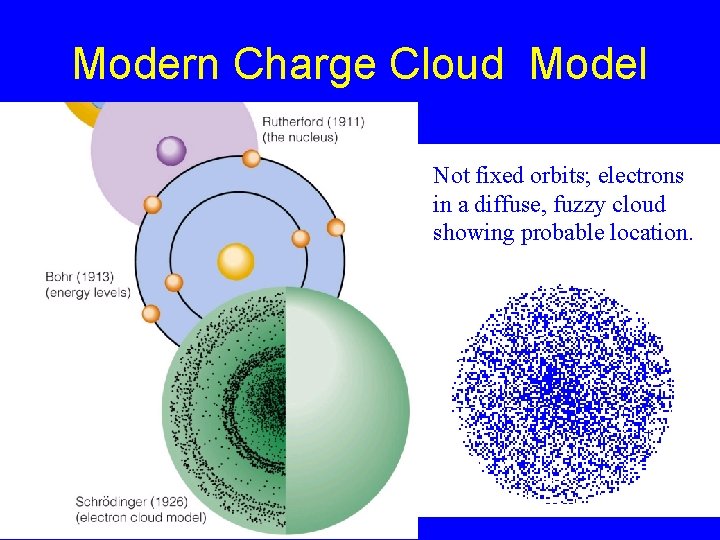
Modern Charge Cloud Model Not fixed orbits; electrons in a diffuse, fuzzy cloud showing probable location.

Find the # of Subatomic Particles by using “MAN” & “APE” Mass Number (= p+ & no) -- minus Atomic Number (= p+ only) = # of Neutrons = no At # = # P+ ~ # e -

Now You Describe these Atoms Use “MAN” & “APE” Atom 19 # Protons # Neutrons # Electrons F 9 ------------------------------39 K 19
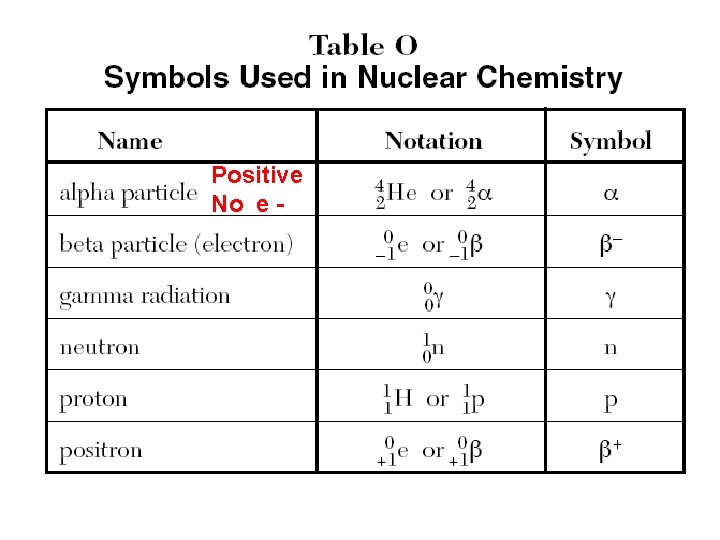
Positive No e -
- Slides: 17