Atomic Theory Important People Democritus 300 B C

Atomic Theory

Important People Democritus – 300 B. C. , Aristotle’s student Said that matter can be divided and divided until a particle is reached that can’t be divided any more Particle = “atom”

Important People Aristotle – said Democritus was wrong n Said that matter divides infinitely until nothing is left Robert Boyle & Isaac Newton – first questioned Aristotle’s ideas in the 1700’s

Important People John Dalton – 1 st published atomic theory which stated: n Atoms are solid spheres n All elements are made up of indivisible atoms (we now know that atoms have p+, n 0, e-) n All atoms in same element are identical (we now know about isotopes, which are atoms with different # of n 0)

Important People n Atoms of different elements are different n Atoms of different elements combine in whole # ratios 1 C + 2 O = CO 2 2 H + 1 O = H 20 n Chemical reactions occur when atoms separate, rearrange, and rejoin

Important People JJ Thomson – atoms are solid with + and – areas n Plum Pudding Theory – solid + material with chunks of - material at random

Important People James Rutherford – atoms consist mostly of space with a dense, central + area = NUCLEUS n Nuclear Atom Theory – proved by gold foil experiment

Gold Foil Experiment Lead barrier with hole + Gold foil ++++++++++++ Lead box with hole + Photosensitive screen with hole *Positive charges went straight through – proves that atoms are mostly space *Fewer positive charges reflected off – proves atoms have positive particles

Gold Foil Experiment n Most particles went straight through, proving that atoms are mostly space n Some had slightly bent path as they went through n Some reflected directly back, which meant they must have hit something + and dense to be repelled n Dense + area = nucleus

Important People Niels Bohr – Danish; worked for Thomson and Rutherford n Planetary Theory of Atoms – electrons orbit around the nucleus like planets around the sun

Important People Max Plank – German n Quantum Mechanical Model: e- do not orbit, but move randomly in ecloud James Chadwick – discovered neutron in 1936; nucleus is too heavy to be just protons; a neutron has mass but no charge

Today Warm-Up (10 min) – Significant scientist quiz – You may use your notes – You may use the folks at your table – You only have 10 mins! n Notes – Modern atomic theory n Assignments: – Finish timeline & webquest – Atoms, Ions, & Isotopes practice n Exit Ticket – Kahoot review n

Modern Atomic Theory n Atoms are building blocks of matter n Atoms are the smallest particle retain all characteristics of the element n Atoms have 2 regions = nucleus and electron cloud n Nucleus is very dense; contains protons and neutrons n Electron cloud is mostly space with randomly moving e-

Electron Cloud n Has energy levels n Levels are numbered from nucleus outward n Various n #e- = 2 n 2 levels hold different # of e(n = # of energy level)

Modern Atomic Theory Protons (p+) n mass = 1 amu (atomic mass unit) n Atomic n No # = # of protons 2 atomic #s are the same, so we can identify the element by the # of protons

Modern Atomic Theory Neutrons (n 0) n mass = 1 amu (atomic mass unit) n Mass # - atomic # = # of neutrons (n 0) Electrons (e-) n No n In relative mass neutral atoms, #p+ = #en Discovered using cathode ray tube
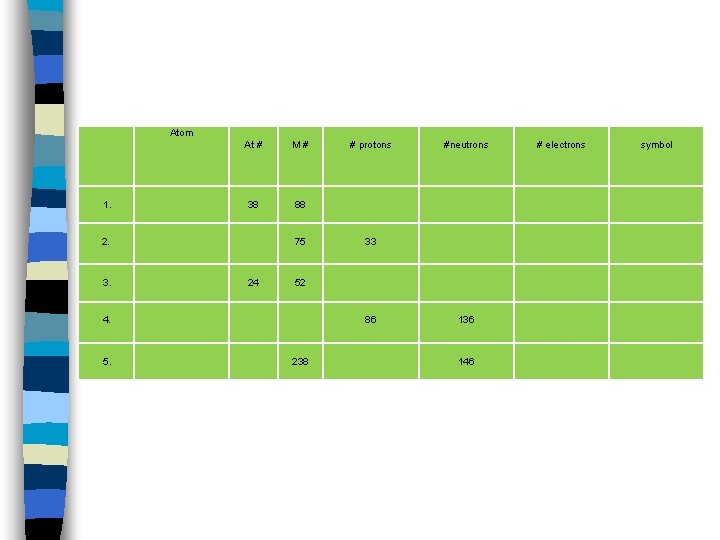
Atom 1. At # M# 38 88 2. 3. 75 24 #neutrons 33 52 4. 5. # protons 86 238 136 146 # electrons symbol

Ions * change in the number of electrons! More or less e- than p+ = charge n Cation lost n Anion has + charge because e- are has – charge because e- are gained

Modern Atomic Theory n Atoms and ions have different #s of ebut always the same # of p+ n What about neutrons? Isotopes are atoms with varying #s of neutrons n 2 ways to write isotopes: Carbon – 12 OR 12 C # n 0 = 12 -6 = 6 Carbon – 14 OR 14 C # n 0 = 14 -6 = 8 6 6

Isotopes n How many neutrons in isotopes: H H protium deutrium 235 92 U? n Common 1 2 1 1 H tritium 3 1
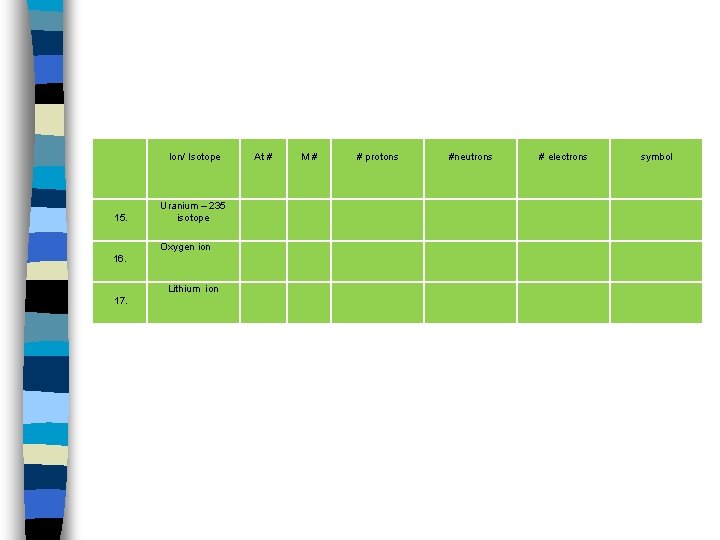
Ion/ Isotope 15. Uranium – 235 isotope Oxygen ion 16. Lithium ion 17. At # M# # protons #neutrons # electrons symbol

For the Remainder of Class Finish webquest (due today) 2. Finish atomic history timeline (due today) 3. Assignment – Atoms, Ions, Isotopes (due today) 1.

Average Atomic Mass Weighted average of atomic masses of naturally occurring isotopes of an element AVG = (% x m) + (% x m)

Average Atomic Mass EX: Copper exists as 2 isotopes Cu @ 69. 17% and Cu @ 30. 83%. Find average atomic mass. 63 29 65 29 (0. 6917 x 63) + (0. 3083 x 65) =

Average Atomic Mass Three isotopes of sulfur occur in nature. Sulfur-31 occurs 4. 05% of the time, while sulfur-32 occurs 90. 75% of the time. Sulfur -33 occurs the remainder of the time. What is the average atomic mass of sulfur?
- Slides: 25