Atomic Theory Democritus 460 370 B C There

Atomic Theory

Democritus 460 – 370 B. C. • There are various basic elements from which all matter is made • Everything is composed of small atoms moving in a void • Some atoms are round, pointy, oily, have hooks, etc. to account for their properties • Ideas lacked experimental evidence • Ideas rejected by leading philosophers because void = no existence

First Concept of an Atom

Lavoisier • The “Father of Modern Chemistry. ” • In 1774, he showed/developed the Law of Conservation of Mass: Matter cannot be created nor destroyed, only changed in form.

John Dalton 1766 -1844 • Introduced his ideas in 1803 • Each element is composed of extremely small particles called atoms • All the atoms of a given element are identical, but they differ from those of any other element • Atoms are neither created nor destroyed in any chemical reaction (Repeating Lavoisier) • A given compound always has the same relative numbers and kinds of atoms
Dalton’s Model

J. J. Thomson 1856 -1940 • Cathode Ray tubes were developed in 1879 by William Crookes and fine tuned over the next 20 years. • Discovered electron 1897 – Cathode Ray Experiment • Plum Pudding model 1904 • All sources (elements) could produce cathode rays so electrons in all atoms. • All atoms are neutral so there also had to be a positive charge in the atoms • Electrons in a soup of positive charges • Discovered isotopes in 1913
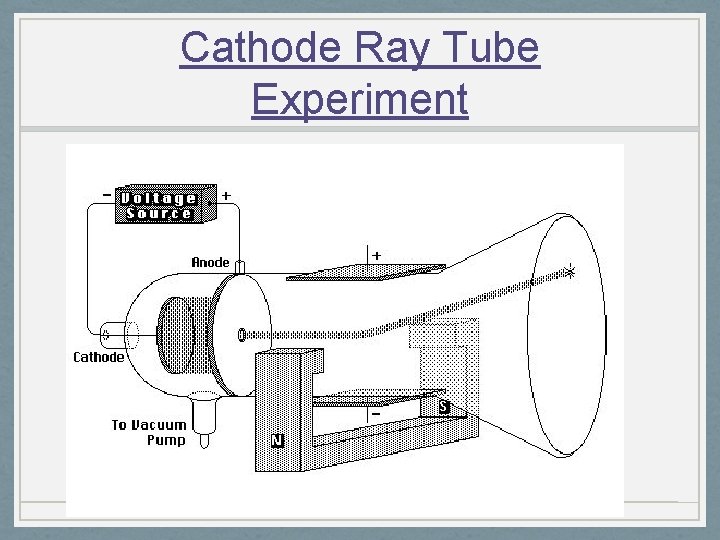
Cathode Ray Tube Experiment

Plum Pudding Model

In 1909 Robert Millikan measured the charge on an electron in his Oil Drop Experiment. • Using the charge to mass ratio he concluded that: • the mass of an electron is 1840 times smaller than that of the Proton • the charge on an electron is -1.

Ernest Rutherford (That’s Sir to you) 1871 -1937 • Becquerel discovered radioactivity around 1895 but Rutherford separated and defined the three types. • Figured out alpha particles were helium nucleii using atomic spectra. • Worked with Gieger to develop an Alpha particle counter.

Ernest Rutherford 1871 -1937 • Nucleus Theory 1911 • Alpha particle gold foil experiment • Most particles went straight through so an atom is mostly empty space • Some particles were deflected so the nucleus (center of the atom) must have a positive charge • An atom’s mass is mostly in the nucleus • Electrons in fixed orbit

Rutherford to Chadwick • Rutherford was convinced the nucleus, because of it’s mass, contained another particle other than protons. • He thought the other particle was a “neutral couplet” • Chadwick discovered the neutron by bombarding atoms with alpha particles which caused a release of neutrons from the nucleus.
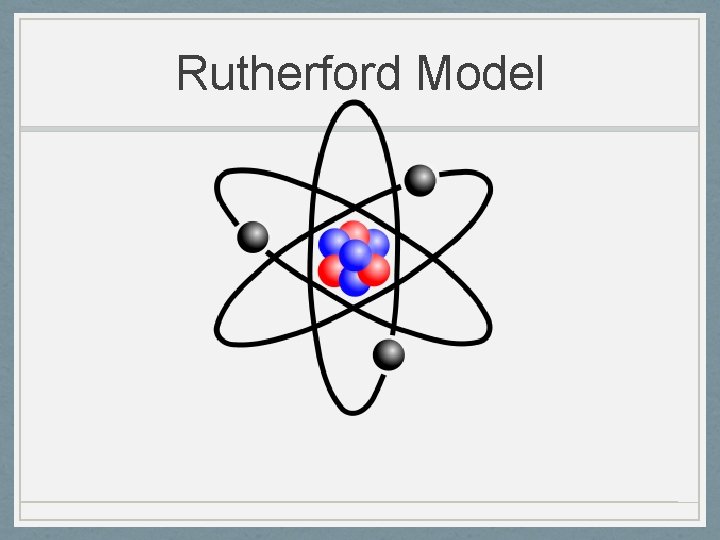
Rutherford Model

Niels Bohr 1885 -1962 • Planetary Model 1913 • Nucleus surrounded by orbiting electrons at different energy levels • Electrons have definite orbits • These orbits are at different distances from the nucleus. • These orbits, or energy levels, are not evenly spaced. • Utilized Planck’s Quantum Energy theory • Worked on the Manhattan Project (US atomic bomb)
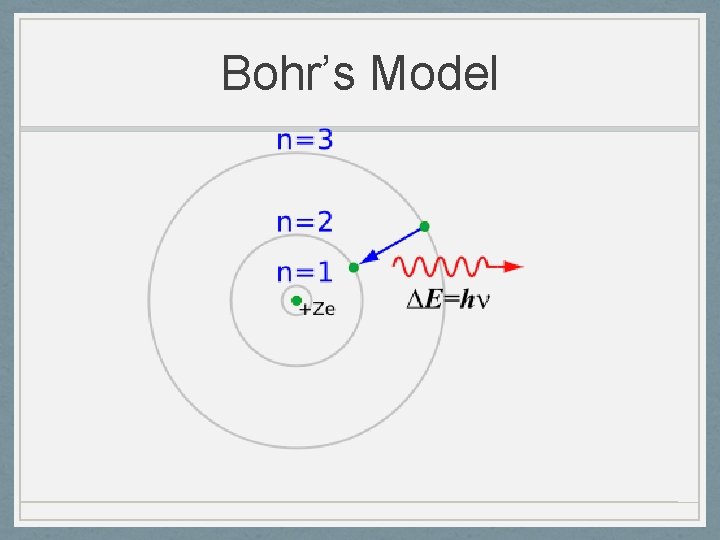
Bohr’s Model
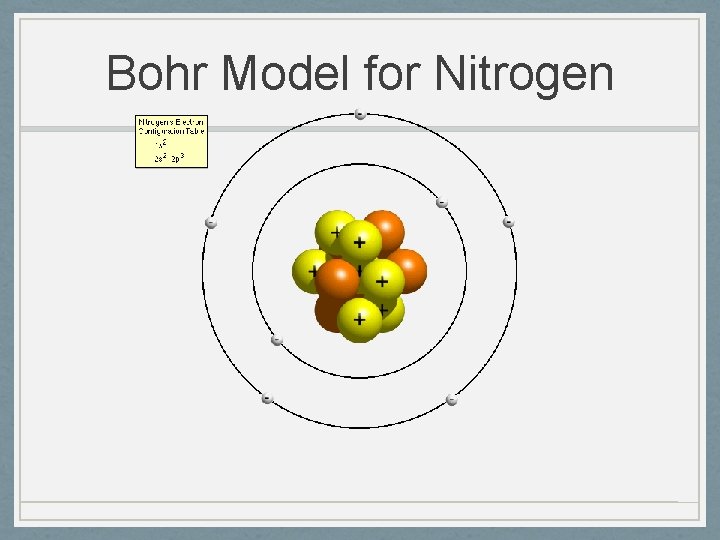
Bohr Model for Nitrogen

Ernst Schrödinger 1887 -1961 Werner Heisenberg 1901 -1976 • Quantum Mechanical Model 1926 • Electrons are in probability zones called “orbitals”, not orbits and the location cannot be pinpointed • Electrons are particles and waves at the same time • Developed quantum numbers based on theories of Einstein and Planck
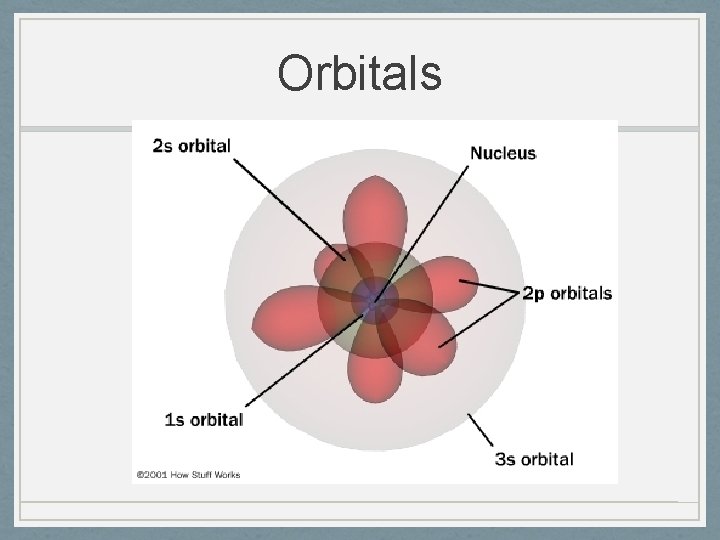
Orbitals

Questions • How did the atomic theory come about? • Was it one person? • Did each person start anew? • Are we sure the current theory is correct? • What technological developments might lead to changes in the current theory? • What would cause one to change this or any theory?
- Slides: 20