Atomic Theory Building blocks of matter Democritus 400

Atomic Theory Building blocks of matter

Democritus – 400 B. C. o All matter is made up of tiny particles called “atomos” o Atoms are indestructible and vary in shape and size Matter: anything that has mass and takes up space

John Dalton - 1808 Atomic Theory 1. All matter is made up of atoms. 2. Atoms of the same element are alike. 3. Atoms of different elements have different masses. 4. Atoms of different elements can join together to form compounds. 5. Atoms cannot be subdivided.

Dalton’s Atom Model o Atom is a solid ball

J. J. Thomson - 1897 Discovered the electron Negatively charged subatomic particle Solid ball with negative particles all throughout it Called the “plum pudding model”

Hantaro Nagaoka - 1904 Atom is like the solar system A huge sphere with tiny negative charged electrons revolving around it. “Saturian model”

Ernest Rutherford - 1911 Atoms are not solid Atoms are mostly empty space Discovered the proton Gold foil experiment http: //www. youtube. com/watch? v=5 p. Zj 0 u_XMbc

Neils Bohr - 1913 Electrons move in orbits or energy levels

James Chadwick - 1932 Discovered the neutron (the proton not alone in the nucleus)
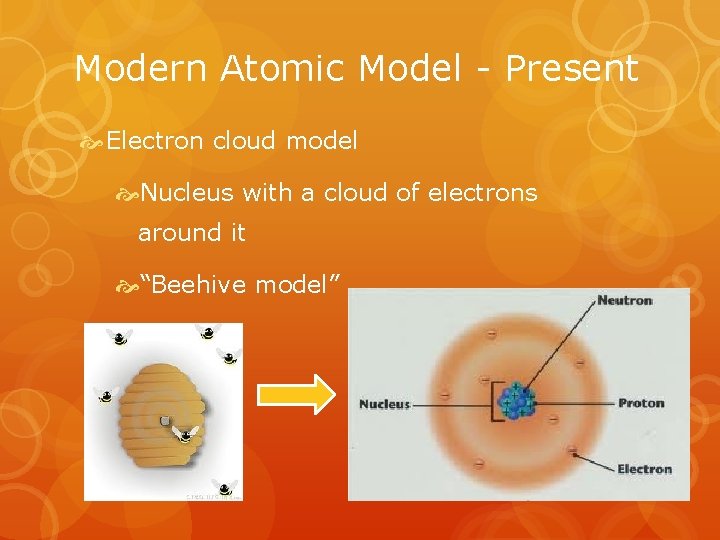
Modern Atomic Model - Present Electron cloud model Nucleus with a cloud of electrons around it “Beehive model”

Bill Nye – “The Atom” http: //www. youtube. com/watch? v=cn. XV 7 Ph 3 WPk&feature =related
- Slides: 11