Atomic Theory and Structure Nuclear theory Subatomic particles

Atomic Theory and Structure Nuclear theory Subatomic particles Isotopes Ions

History n n Matter seems continuous in the macroscopic world. Leucippus (5 th Century, B. C. ) and Democritus (460 – 370 B. C. ) – Greek philsophers that proposed otherwise. q q Matter is ultimately composed of small, indivisible particles called atomos or atoms. Not widely accepted.

Dalton’s Model of the Atom – Early Atomic Theory, l 808, Broad n Elements are composed of minute, indivisible acceptance n n n particles called atoms. Atoms of the same element are alike in mass and size. Atoms of different elements have different masses and sizes. Chemical compounds are formed by the union of two or more atoms of different elements Atoms combine to form compounds in simple, whole -number ratios. Atoms of two elements may combine in different ratios to form more than one compound

Modifications to Dalton’s Theory n n Atoms are composed of subatomic particles Not all the atoms of a specific element have the same mass (isotopes)

n 1856 -1940, J. J. Thomson, English physicist q q n n 1897 - discovered the electron A smaller particle than the atom. Electron – negatively charged Plum-pudding model This discovery brought on more questions. Atoms were neutral so what about a balancing charge? 1871 -1937, Ernest Rutherford, worked with Thomson – His work discovered the nuclear atom
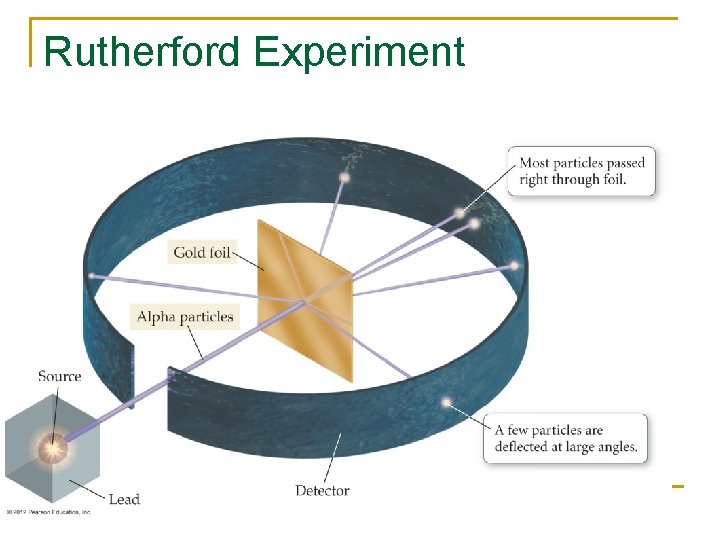
Rutherford Experiment
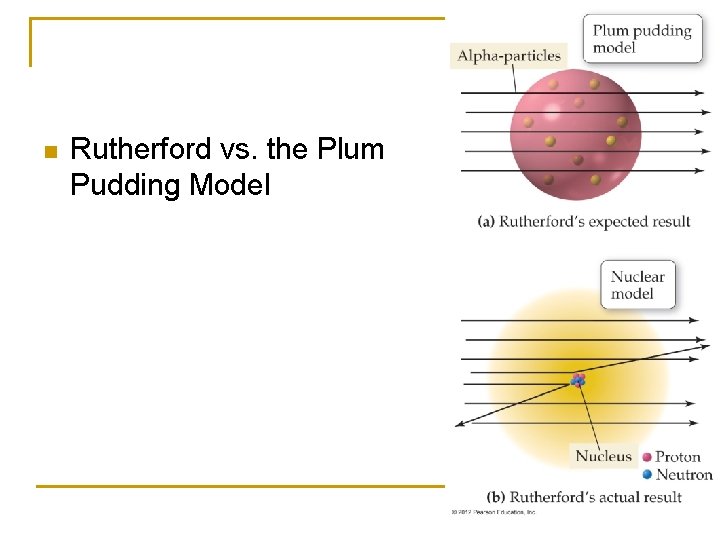
n Rutherford vs. the Plum Pudding Model

Rutherford’s Discovery n n n Most of the atom’s mass and all of its positively charged particles are contained in a small core called the nucleus Negatively charged particles are dispersed in the empty area surrounding the nucleus The number of negatively charged particles is equal to the number of positively charged particles. Later work by Rutherford and others demonstrated that the atom’s nucleus contains both positively charged protons and neutral particles call neturons. The dense nucleus makes up 99. 9% of the atoms mass but only a small fraction of the volume.

Nucleus n n Contains protons and neutrons In a neutral atom (not in ionic form), the number of protons equals the number of electrons circling the nucleus. Protons positive charge, neutrons no charge Protons in the nucleus identify the element

Properties of electric charges n n n Proton positive charge Electrons negative charge Atoms have no charge because the number of protons equals the number of neutrons

Periodic Table
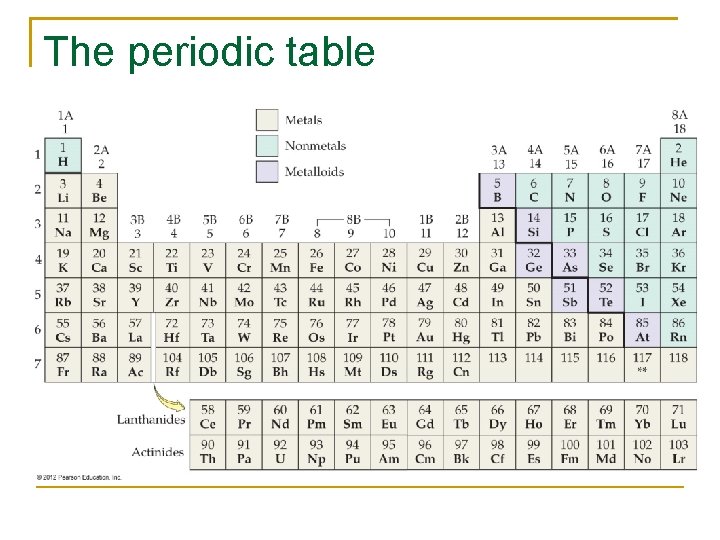
The periodic table

Periodic Table n n n Arranged in ascending order of number of protons Shows symbols of elements Shows molar mass (atomic mass) Separated into metals, metalloids, nonmetals The columns are groups or families. q Alkali metals q Alkaline earth metals q Halogens q Noble gases The rows are periods.

Atomic Mass n n n Atomic masses of elements are shown on the periodic table. The atomic mass shown takes into account an atom’s isotopes and the percent which the isotopes are found in nature (percent abundance). Atomic mass is the average relative mass of all the atom’s isotopes

Atomic Mass Units n n n Mass of an atom is too small to weigh on a balance Can be measured with a mass spectrometer. Atomic masses are measured in atomic mass units All are based on the carbon-12. One atomic mass unit (amu) is equal to 1/12 of the mass of a carbon-12 atom

Atomic Number (Z) of Elements n n Atomic number is the number of protons in the nucleus of an atom. It is denoted by Z. The number of protons gives an atom its identity. The number of protons never change in the nucleus of an atom unless the atom undergoes a nuclear reaction. The elements on the periodic table are listed in ascending order of atomic number (protons)

Mass number n n n The number of protons and neutrons found in the nucleus of an atom. Denoted by A. Mass number = protons + neutrons

Relationship between mass number (A) and atomic number (Z) n Given a mass number and an atomic number we can determine the number of neutrons in an atom. n A = number of protons plus number of neutrons Number of neutrons = A - Z n
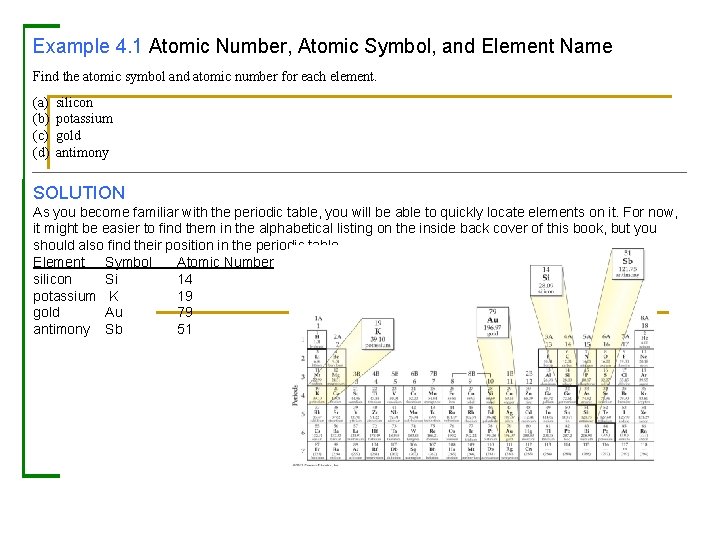
Example 4. 1 Atomic Number, Atomic Symbol, and Element Name Find the atomic symbol and atomic number for each element. (a) (b) (c) (d) silicon potassium gold antimony SOLUTION As you become familiar with the periodic table, you will be able to quickly locate elements on it. For now, it might be easier to find them in the alphabetical listing on the inside back cover of this book, but you should also find their position in the periodic table. Element Symbol Atomic Number silicon Si 14 potassium K 19 gold Au 79 antimony Sb 51

Example 4. 1 Atomic Number, Atomic Symbol, and Element Name Continued SKILLBUILDER 4. 1 Atomic Number, Atomic Symbol, and Element Name Find the name and atomic number for each element. (a) Na (b) Ni (c) P (d) Ta Answers: (a) sodium, 11 (b) nickel, 28 (c) phosphorus, 15 (d) tantalum, 73 For More Practice Problems 41, 42, 45, 46, 47, 48, 49, 50.

Example 4. 2 Classifying Elements as Metals, Nonmetals, or Metalloids Classify each element as a metal, nonmetal, or metalloid. (a) Ba (b) I (c) O (d) Te SOLUTION (a) (b) (c) (d) Barium is on the left side of the periodic table; it is a metal. Iodine is on the right side of the periodic table; it is a nonmetal. Oxygen is on the right side of the periodic table; it is a nonmetal. Tellurium is in the middle-right section of the periodic table, along the line that divides the metals from the nonmetals; it is a metalloid. SKILLBUILDER 4. 2 Classifying Elements as Metals, Nonmetals, or Metalloids Classify each element as a metal, nonmetal, or metalloid. Answers: (a) S (a) nonmetal (b) Cl (b) nonmetal (c) Ti (c) metal (d) Sb (d) metalloid For More Practice Problems: 51, 52, 53, 54.

Example 4. 3 Groups and Families of Elements To which group or family of elements does each element belong? (a) Mg (b) N (c) K (d) Br SOLUTION (a) (b) (c) (d) Mg is in Group 2 A; it is an alkaline earth metal. N is in Group 5 A. K is in Group 1 A; it is an alkali metal. Br is in Group 7 A; it is a halogen. SKILLBUILDER 4. 3 Groups and Families of Elements To which group or family of elements does each element belong? (a) (b) (c) (d) Li B I Ar For More Practice Problems 57, 58, 59, 60, 61, 62, 63, 64. Answers: (a) alkali metal, group 1 A (b) group 3 A (c) halogen, group 7 A (d) noble gas, group 8 A

Isotopes n n All atoms of a given element have the same number of protons. All atoms of a given element DO NOT have identical masses. This is due to differing numbers of neutrons in the nucleus of an atom. Isotopes – atoms of an element having the same atomic number but different atomic masses
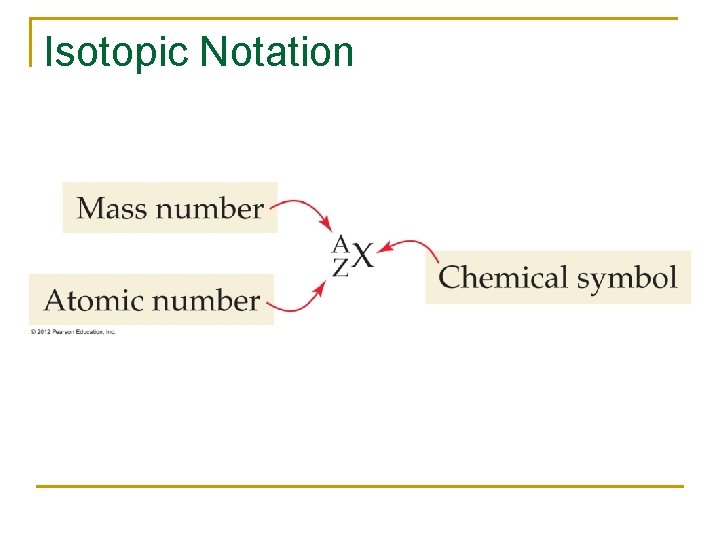
Isotopic Notation

Give the nuclear composition and isotopic notation for n n A) an atom containing 27 protons, 32 neutrons, and 27 electrons B) an atom containing 15 protons, 16 neutrons, and 15 electrons C) an atom containing 110 neutrons, 74 electrons, and 74 protons D) an atom with 143 neutrons, 92 electrons and an atomic number of 92

Write the isotopic notation symbols for each of the following n n n A) Z = 29 B) Z = 20 C) Z = 36 A = 65 A = 45 A = 84

Example 4. 7 Atomic Numbers, Mass Numbers, and Isotope Symbols What are the atomic number (Z), mass number (A), and symbols of the carbon isotope with 7 neutrons? SOLUTION Find that the atomic number (Z) of carbon is 6 (from the periodic table). This tells you that carbon atoms have 6 protons. The mass number (A) for the isotope with 7 neutrons is the sum of the number of protons and the number of neutrons. A = 6 + 7 = 13 So, Z = 6, A = 13, and the symbols for the isotope are C-13 and. SKILLBUILDER 4. 7 Atomic Numbers, Mass Numbers, and Isotope Symbols What are the atomic number, mass number, and symbols for the chlorine isotope with 18 neutrons? Answers: For More Practice Example 4. 12; Problems: 85, 87, 89, 90.

Example 4. 8 Numbers of Protons and Neutrons from Isotope Symbols How many protons and neutrons are in the chromium isotope The number of protons is equal to Z (lower left number). The number of neutrons is equal to A (upper left number) –Z (lower left number). SOLUTION #p+ = Z = 24 #n = A – Z = 52 – 24 = 28 SKILLBUILDER 4. 8 Numbers of Protons and Neutrons from Isotope Symbols How many protons and neutrons are in the potassium isotope ? Answers: 19 protons, 20 neutrons For More Practice Example 4. 13; Problems 91, 92. ?

Example 4. 9 Calculating Atomic Mass Gallium has two naturally occurring isotopes: Ga-69 with mass 68. 9256 amu and a natural abundance of 60. 11%, and Ga-71 with mass 70. 9247 amu and a natural abundance of 39. 89%. Calculate the atomic mass of gallium. Convert the percent natural abundances into decimal form by dividing by 100. SOLUTION Use the fractional abundances and the atomic masses of the isotopes to compute the atomic mass according to the atomic mass definition given earlier. Atomic mass = (0. 6011 68. 9256 amu) + (0. 3989 70. 9247 amu) = 41. 4321 amu + 28. 2919 amu = 69. 7231 = 69. 72 amu SKILLBUILDER 4. 9 Calculating Atomic Mass Magnesium has three naturally occurring isotopes with masses of 23. 99, 24. 99, and 25. 98 amu and natural abundances of 78. 99%, 10. 00%, and 11. 01%. Calculate the atomic mass of magnesium. Answer: 24. 31 amu For More Practice Example 4. 14; Problems 95, 96.

Example 4. 12 Determining Atomic Numbers, Mass Numbers, and Isotope Symbols for an Isotope What are the atomic number (Z), mass number (A), and symbols for the iron isotope with 30 neutrons? SOLUTION The atomic number of iron is 26. A = 26 + 30 = 56 The mass number is 56. Fe-56

What is the nuclear composition of five naturally occurring isotopes of germanium having mass numbers of 70, 72, 73, 74, and 76?

Discovery of Ions n Michael Faraday – q q q n Svante Arrhenius – q n n certain substances when dissolved in water conduct an electrical current. Atoms of some elements are attracted to the positive electrode, while atoms of other elements are attracted to the negative electrode. Coined the phrase “ion”. An ion is an atom carrying a positive or negative charge. G. J. Stoney – some fundamental unit of electricity associated with atoms; named the unit electron J. J. Thomson – experimentally proved the existence of the electron
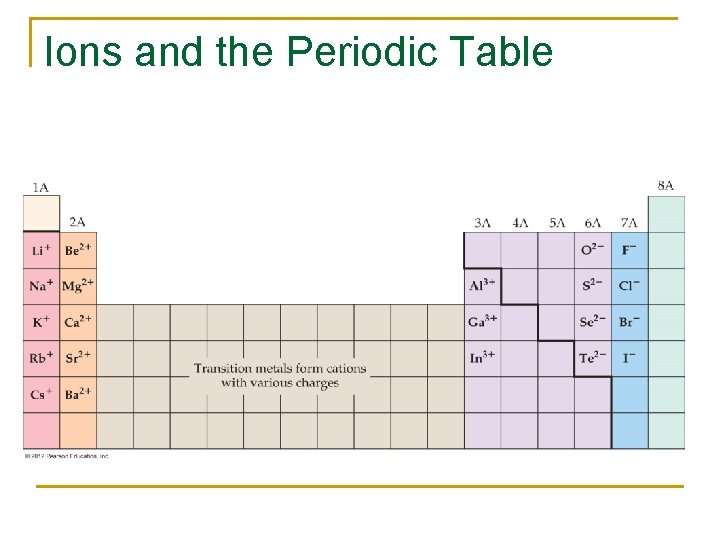
Ions and the Periodic Table

Formation of Ions n n Atoms lose or gain electrons. Cations are positively charged ions. q n Fformed by metals losing electrons Anions are negatively charged ions q Formed by nonmetals gaining electrons

Example 4. 4 Determining Ion Charge from Numbers of Protons and Electrons Determine the charge of each ion. (a) a magnesium ion with 10 electrons (b) a sulfur ion with 18 electrons (c) an iron ion with 23 electrons SOLUTION To determine the charge of each ion, use the ion charge equation. . Ion charge = #p – #e– The number of electrons is given in the problem. The number of protons is obtained from the element’s atomic number in the periodic table (a) magnesium with atomic number 12 Ion charge = 12 – 10 = 2+ (Mg 2+) (b) sulfur with atomic number 16 Ion charge = 16 – 18 = 2– (S 2–) (c) iron with atomic number 26 Ion charge = 26 – 23 = 3+ (Fe 3+)

Example 4. 4 Determining Ion Charge from Numbers of Protons and Electrons Continued SKILLBUILDER 4. 4 Determining Ion Charge from Numbers of Protons and Electrons Determine the charge of each ion. (a) a nickel ion with 26 electrons (b) a bromine ion with 36 electrons (c) a phosphorus ion with 18 electrons Answers: (a) 2+ (b) 1– (c) 3 – For More Practice Example 4. 10; Problems 73, 74.

Example 4. 5 Determining the Number of Protons and Electrons in an Ion Find the number of protons and electrons in the Ca 2+ ion. From the periodic table, find that the atomic number for calcium is 20, so calcium has 20 protons. The number of electrons can be found using the ion charge equation. SOLUTION Ion charge = #p – #e– 2+ = 20 – #e– = 20 – 2 = 18 Therefore the number of electrons is 18. The Ca 2+ ion has 20 protons and 18 electrons. SKILLBUILDER 4. 5 Determining the Number of Protons and Electrons in an Ion Find the number of protons and electrons in the S 2 ion. Answer: 16 protons, 18 electrons For More Practice Example 4. 11; Problems 75, 76.

Example 4. 10 Determining Ion Charge from Numbers of Protons and Electrons Determine the charge of a selenium ion with 36 electrons. SOLUTION Selenium is atomic number 34; therefore, it has 34 protons. Ion charge = 34 – 36 = 2–
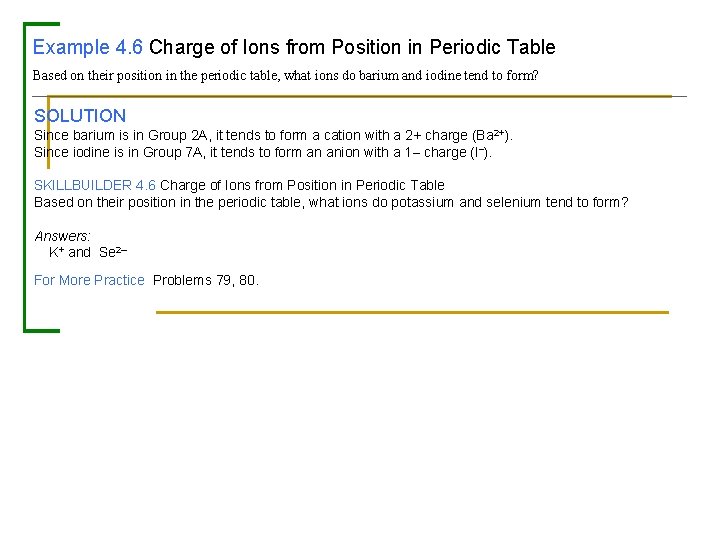
Example 4. 6 Charge of Ions from Position in Periodic Table Based on their position in the periodic table, what ions do barium and iodine tend to form? SOLUTION Since barium is in Group 2 A, it tends to form a cation with a 2+ charge (Ba 2+). Since iodine is in Group 7 A, it tends to form an anion with a 1– charge (I–). SKILLBUILDER 4. 6 Charge of Ions from Position in Periodic Table Based on their position in the periodic table, what ions do potassium and selenium tend to form? Answers: K+ and Se 2– For More Practice Problems 79, 80.

Composition of Compounds n n Law of definite composition – compounds always contain two or more elements chemically combined in a definite proportion by mass. Law of multiple proportions – atoms of two or more elements combine in different ratios to produce more than one compound.

Percent by mass n 100. 0 g of Cu. Cl is decomposed into 64. 2 g of copper and 35. 8 g of chlorine. What is the % by mass of each atom? n 50. 0 g of Cu. Cl 2 is formed from 23. 65 g of copper and 26. 35 g of chlorine. What is the % by mass of each atom?

Example 4. 14 Calculating Atomic Mass from Percent Natural Abundances and Isotopic Masses Copper has two naturally occurring isotopes: Cu-63 with mass 62. 9395 amu and a natural abundance of 69. 17%, and Cu-65 with mass 64. 9278 amu and a natural abundance of 30. 83%. Calculate the atomic mass of copper. SOLUTION
- Slides: 42