Atomic Theory and Radioactive Decay n Natural background

Atomic Theory and Radioactive Decay n. Natural background radiation exists all around us. n. Radioactivity is the release of high energy particles or waves that we call radiation.
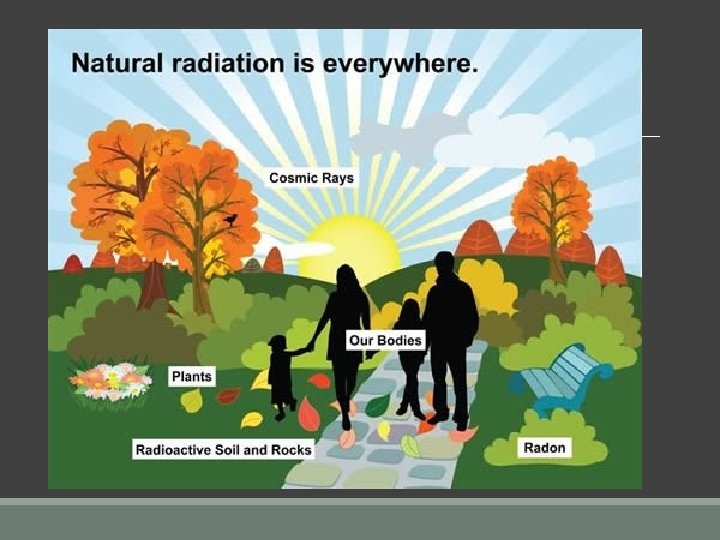

When atoms lose high energy particles and waves, new atoms can be formed. Radiation can be beneficial or harmful to us Benefits §X-rays, §radiation therapy §electricity generation are beneficial. Harms caused by radiation sources §High energy particles and waves can do damage to DNA in our cells.
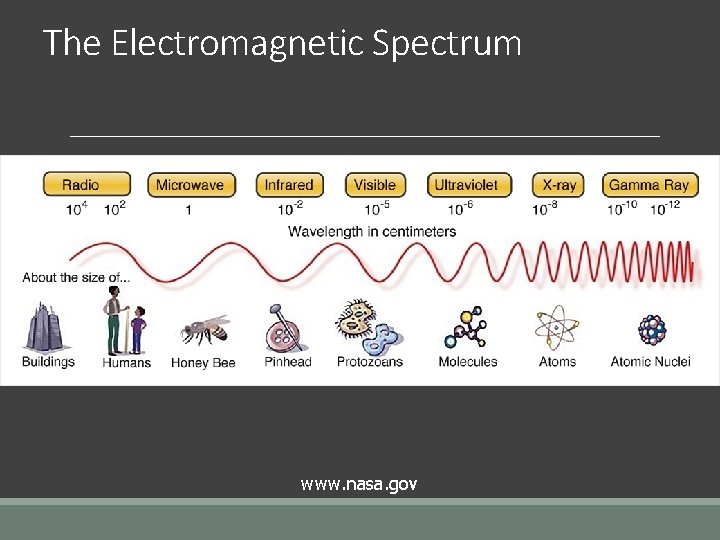
The Electromagnetic Spectrum www. nasa. gov

Early Discoveries of Radiation Roentgen named X rays with an “X” more than 100 years ago because they were previously unknown. Later, more sophisticated devices such as the Geiger-Müller counter
Becquerel realized uranium emitted seemingly invisible energy as well. Marie and Pierre Curie named this energy radioactivity.
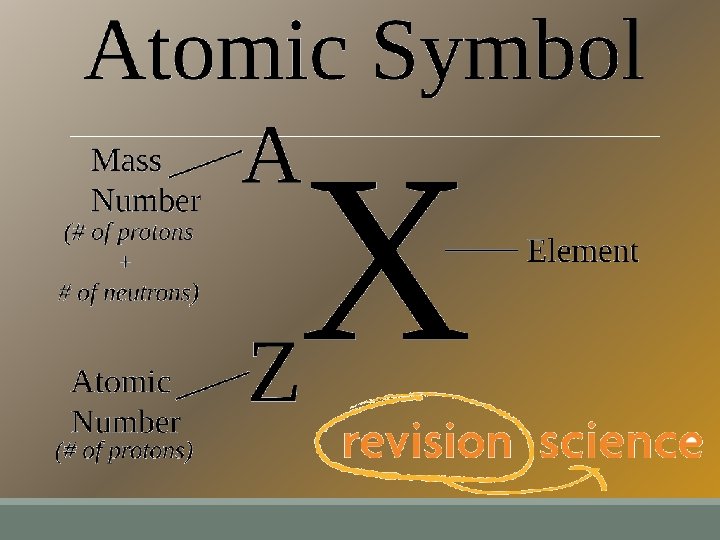

Isotopes are atoms of the same element, with a different number of neutrons. ◦ changing the # of neutrons changes the mass number § Remember: mass # = # protons + # neutrons ◦ isotopes still have the same number of protons and the same element symbol Atomic mass = average of the mass numbers for all isotopes of an element.
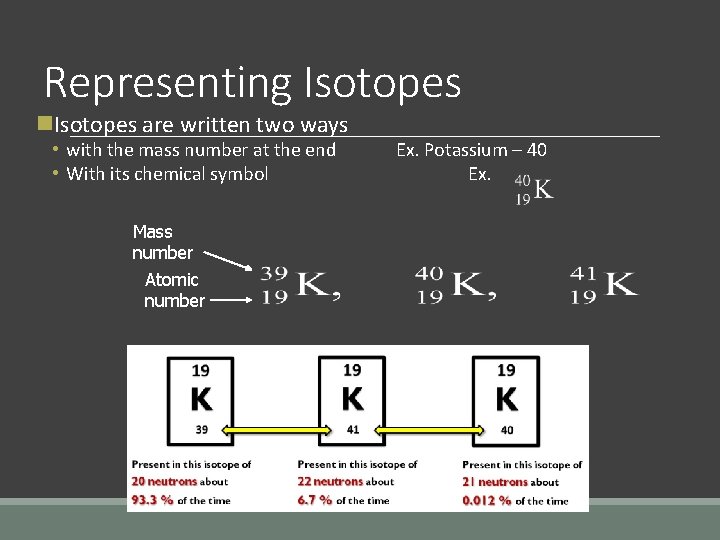
Representing Isotopes n. Isotopes are written two ways • with the mass number at the end • With its chemical symbol Mass number Atomic number Ex. Potassium – 40 Ex.

Radioactive Decay n. Can result in new atoms forming. ◦ Radioactivity results from having an unstable nucleus. ◦ Radioactive decay = when nuclei break apart + release energy from the nucleus. § Radioactive decay continues until a stable element forms. § An element may have isotopes that are radioactive called radioisotopes ◦ Ex. carbon-12, carbon-13 and carbon-14 (only C-14 is radioactive)

n. Rutherford identified three types of radiation using an electric field. ◦ Positive alpha particles were attracted to the negative plate. ◦ Negative beta particles were attracted to the positive plate. ◦ Neutral gamma particles did not move towards any plate.

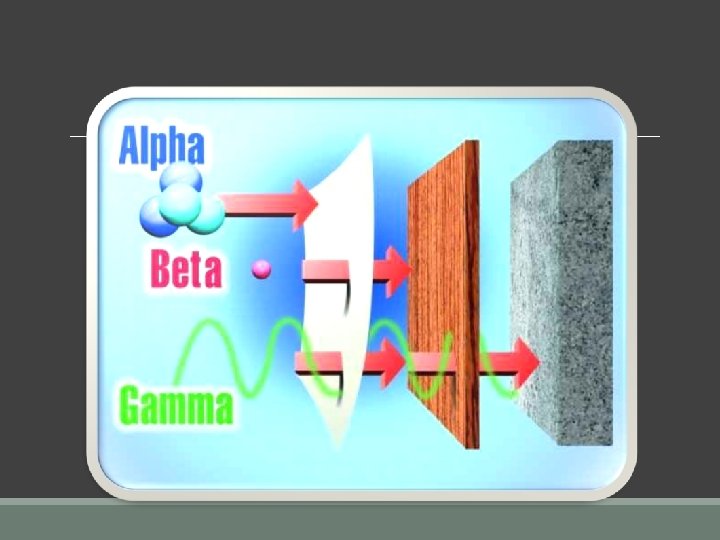
Alpha Radiation: is a stream of alpha particles, (shown as ) positively charged weighs the most are the same as a helium nucleus Alpha particles are represented by the symbols 2 protons and 2 neutrons make a mass number of 4 it has a charge of 2+ because of the protons


Alpha particles are big and slow even a sheet of paper can stop it.

Beta Radiation: n. A Beta particle, , is a high energy electron. ◦ negatively charged, and weigh less than alpha particles. ◦ Beta particles are represented by the symbols § electrons are very tiny, so beta particles are assigned a mass of 0. § one electron gives a beta particle has a charge of 1–.

n. Beta decay occurs when a neutron changes into a proton + an electron. ◦ The proton stays in the nucleus, and the electron is released. Example: The beta decay of iodine - 131 n. It takes a thin sheet of aluminum foil to stop a beta particle.

Gamma Radiation: n. Gamma radiation, , is a ray of high energy, shortwavelength radiation. ◦ has no charge and no mass. ◦ is the highest energy form of electromagnetic radiation. ◦ It takes thick blocks of lead or concrete to stop gamma rays. ◦ Gamma decay results from energy being released from a highenergy nucleus. Shows unstable nucleus for gamma decay

n. Often, other kinds of radioactive decay will also release gamma radiation. ◦ Uranium-238 decays into an alpha particle and also releases gamma rays.
- Slides: 19