Atomic Theory A historical overview Democritus 460 BC

Atomic Theory A historical overview

Democritus (460 BC) A philosopher not a scientist Developed the Particle Theory of matter: n All matter is made up of particles which are invisible, indivisible and indestructible

Aristotle (384 -322 BC) Matter consists of varying proportions of earth, air, fire and water

Dalton (1803) English school teacher Developed an atomic theory: n n Matter is made up of atoms Atoms are indivisible spheres with uniform density throughout All atoms of the same element are identical and differ from atoms of any other element Atoms combine in fixed ratios to make compounds

Thomson (1897) Using a partially evacuated discharge tube, he discovered the electron
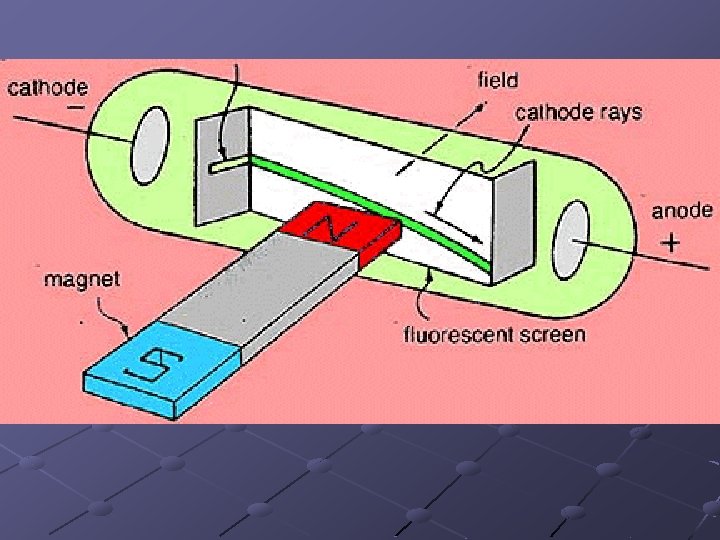

A cathode ray consists of negatively charged particles called electrons Electrons are the same, no matter what substance they come from Electrons have a mass of 0. 00055 u and a charge of -1

J. J. Thomson (1897) Suggested that the atom was composed of negatively charged electrons embedded in a positively charged sphere (i. e. the raisin bun or plum pudding model)
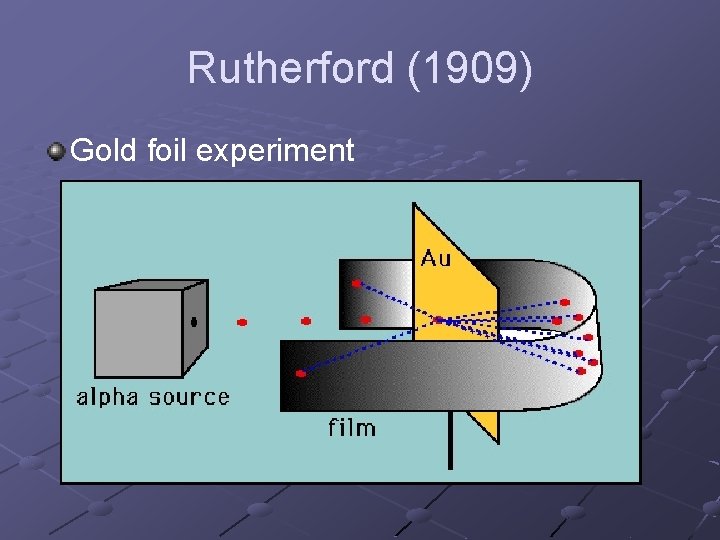
Rutherford (1909) Gold foil experiment

Expected results: since the atom has a uniform density, and since the foil is so thin, all alpha particles should easily pass straight through the foil However, some alpha particles were deflected or even bounced back Conclusion: the positively charged matter must be densely packed in a nucleus and most of the atom must be empty space.

Rutherford Model of the Atom Rutherford suggested that electrons orbit the nucleus in a way analogous to planets orbiting the sun
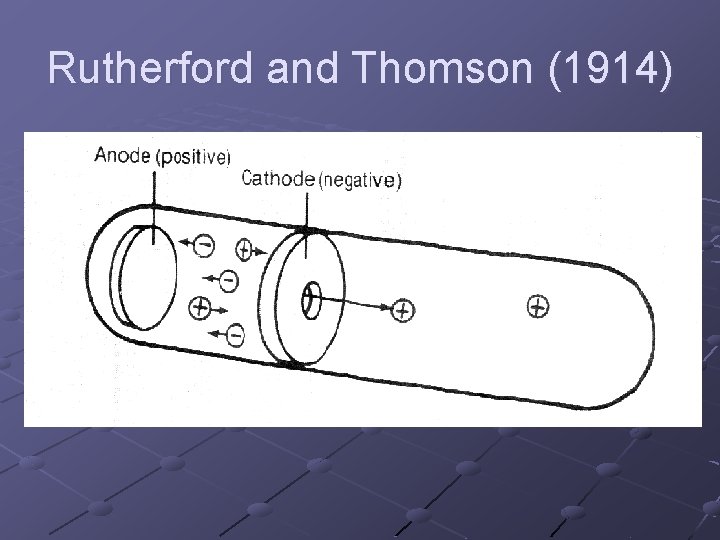
Rutherford and Thomson (1914)

Studied positive rays in a discharge tube and discovered the smallest positively charged particle is a hydrogen ion; AKA a proton Mass of proton is 1. 0073 u; charge +1 Atoms of different elements have different numbers of protons in their nucleus

Chadwick (1932) Hit beryllium atoms with alpha particles. This generated a beam of particles which had approx. the same mass as protons but no charge: the neutron.

Max Planck (1900) A black body is an object that absorbs all light that hits it. When heated, it will emit light. At low temperatures it will glow red. At high temperatures, it will emit white light.
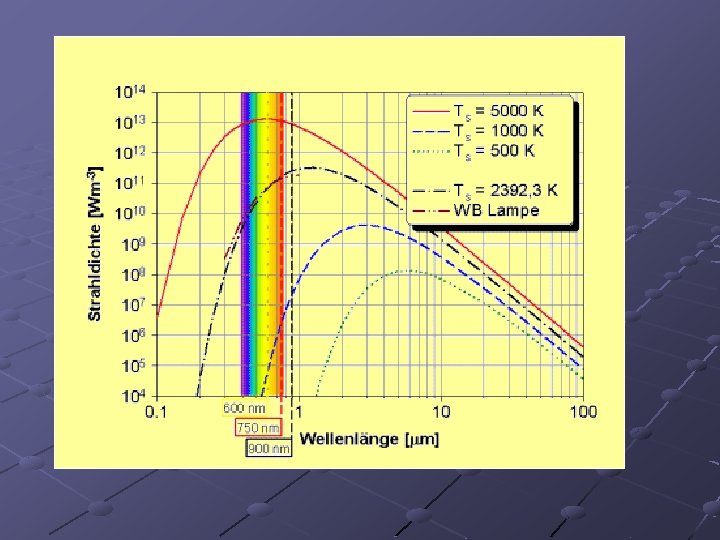

Theories to try to explain black body radiation failed because they all predicted a continuous increase in light intensity as the wavelength became shorter. This did not occur in the lab. (called the UV catastrophe) In order to explain black body radiation, Planck suggested the following: n n n An atom can only emit certain exact amounts of energy. (up until Planck, it was thought that an atom could emit energy of any amount) There is a minimum amount of energy that an atom can emit: a quantum of energy If more energy is emitted, it is an integer multiple of the minimum energy.

Photoelectric Effect Background: light of different colours has different energy i. e. red light has less energy than blue When light strikes some metal surfaces, electrons are ejected from the surface. This is called the photoelectric effect. It was observed that high frequency light (UV) would cause electrons to be ejected while low frequency light (red) wouldn’t
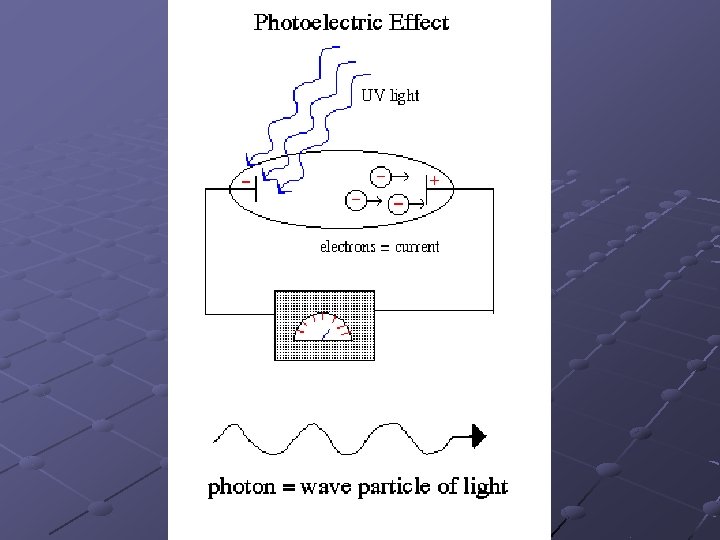

Initially it was thought that if higher intensity red light (brighter source) was used, electrons would be ejected. i. e. a lot of low frequency light would equal a small amount of high frequency light. This was shown to be false. There was a minimum frequency of light that was needed to eject electrons. Why? ? ?

Einstein (prompted by Planck’s ideas) suggested that light comes in packets or quanta of energy called photons. Photons of red light contain less energy than photons of UV light If a high energy photon collides with an electron on a metal surface, the electron will gain the energy of the photon and have sufficient energy to leave the surface. Since this is a one electron – one photon event, having more low energy photons will not result in electron ejection (p. 172)

Problems with Rutherford’s Model Negatively charged electrons would be expected to spiral into the positively charged nucleus since they should emit energy as they accelerate around the nucleus. This does not occur.

Spectroscopy Bright line spectra were observed for atoms rather than continuous spectra. Bohr’s explanation: n n If only certain quanta of light (photon energies) can be emitted from an atom then the energy of the electron must be quantized. That is: electrons can only occupy certain energy levels around the nucleus. They can never occupy intermediate energy levels.


Fixing Bohr’s Model Bohr’s model did not explain why electrons can only have certain energies or why electrons do not collapse into the nucleus.

de Broglie (1923) Suggested that all matter, including electrons behaves like a wave. This was later shown to be true experimentally

Schrodinger Used de Broglie’s idea to explain Bohr’s model. Electrons can only have certain quantized energies because of the requirement for only whole numbers of wavelengths for an electron wave. (p. 199) Demonstrate with waves in a spring.


Where is an electron at any given time? Can we know? By trying to measure where an electron is (hitting it with a photon), we change its energy and its position. Therefore, it is impossible to know exactly where it is at any given time. This is called the Heisenberg uncertainty principle. Heisenberg suggested that we can only know a volume of space where the electron is most probably to be found. These volumes are called orbitals rather than orbits.
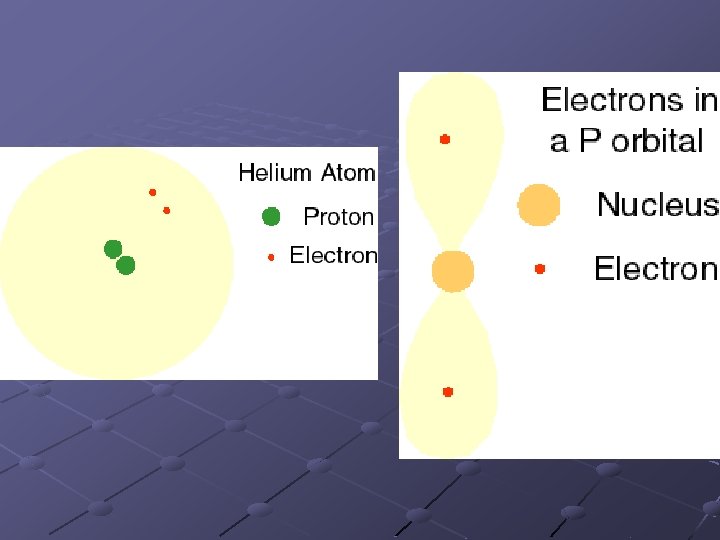

Quantum Numbers Principle Quantum Number (n) n n The energy level of an electron is designated by the letter n. Any energy level can hold up to 2 n 2 electrons

Secondary Quantum Number (l) n n The main lines of the bright line spectrum are actually a number of very small lines. This suggested that there were subshells within every shell of electrons. The number of subshells equals n; for n = 4, there are 4 subshells; l = 0, 1, 2, 3 The subshells have different shapes (volumes of space where electrons are likely to be found. )

l = 0 is spherical in shape and is called an s orbital l = 1 is dumbell shaped and is called a p orbital Above l = 1 it is difficult to describe the shapes in words. Called a d orbital In order, the first 4 types of shapes are called s, p, d, f orbitals

Magnetic quantum number (ml): describes the orientation of the orbitals n n S orbitals have only one orientation around the nucleus since they are spherical P orbitals have 3 orientations: in the x, y and z planes. ml can have values from –l to +l Each orbital can hold up to 2 electrons.
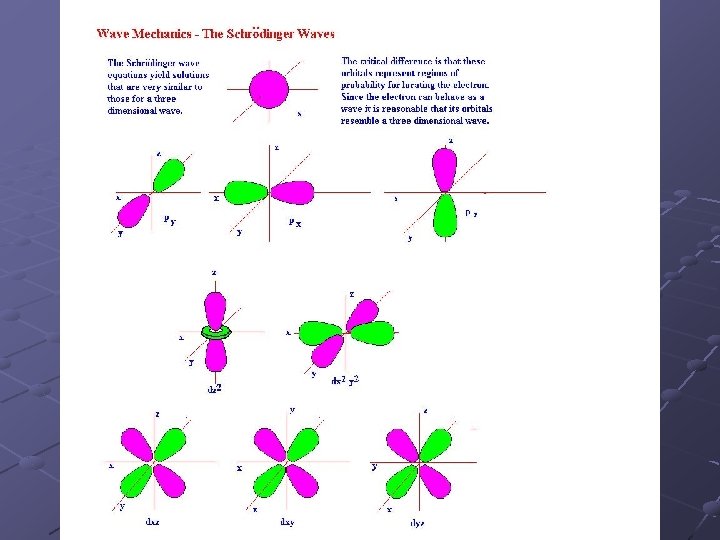

Spin quantum number (ms): describes the direction that an electron spins on its axis Two electrons in the same orbital have opposite spins. ms can be +1/2 or -1/2
- Slides: 36