Atomic Structure What is an atom Atom n




![Atomic Theory n “We might as well attempt to annihilate [a planet], as to Atomic Theory n “We might as well attempt to annihilate [a planet], as to](https://slidetodoc.com/presentation_image_h2/42f680280ec7c99a4f3a9cd3ad93f000/image-5.jpg)

















- Slides: 33

Atomic Structure What is an atom?

Atom n An atom is the smallest physical particle of an element that still retains the properties of that element.

Atomic Theory n First scientist to suggest atoms: Democritus n “Everything that is must be eternal. ” n Democritus believed that there were basic things that always exist, but can be rearranged; he called these “atomos”.

Atomic Theory n John Dalton q q exceptionally bright Quaker put in charge of the local school at age 12 Color blind (was once called Daltonism) First scientist to create a modern definition of “atom”
![Atomic Theory n We might as well attempt to annihilate a planet as to Atomic Theory n “We might as well attempt to annihilate [a planet], as to](https://slidetodoc.com/presentation_image_h2/42f680280ec7c99a4f3a9cd3ad93f000/image-5.jpg)
Atomic Theory n “We might as well attempt to annihilate [a planet], as to create or destroy a particle of hydrogen. ” n Determined relative weights of known elements by breaking apart compounds consisting of them.

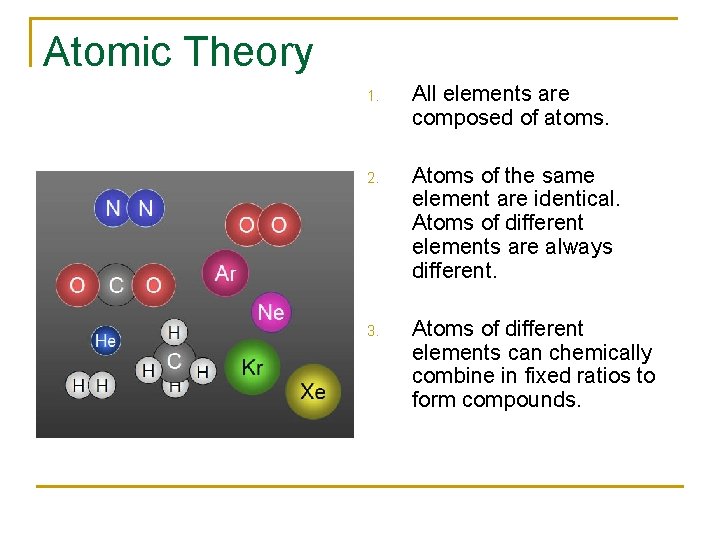
Atomic Theory 1. All elements are composed of atoms. 2. Atoms of the same element are identical. Atoms of different elements are always different. 3. Atoms of different elements can chemically combine in fixed ratios to form compounds.

Atomic Theory 4. Chemical reactions occur when atoms are joined, separated, or rearranged. Atoms of one element are NEVER changed into another during a chemical reaction.

Review of Atoms? n All atoms of the same element are identical. n Chemical reactions occur when atoms of one element are changed into atoms of another element.

Atomic Structure What does an atom look like?

Proof of Atoms n Three scientists that made contributions to atomic structure – J. J. Thompson, Ernest Rutherford, and Neils Bohr.

John Dalton’s Atom n Atoms are solid and indivisible.

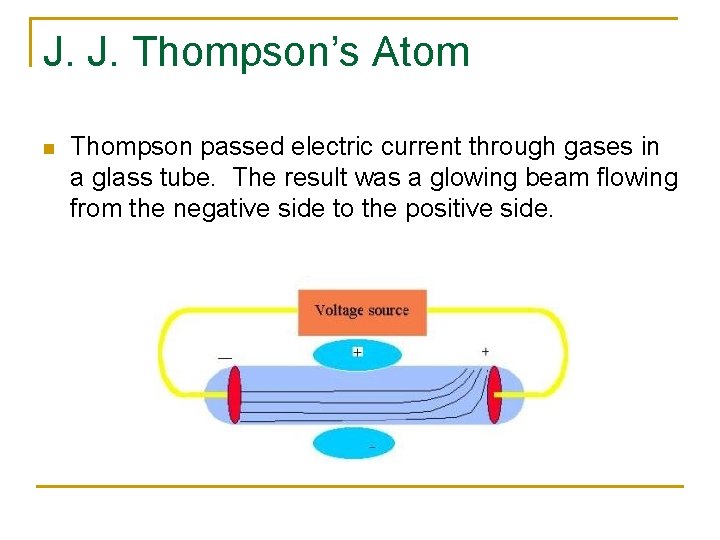
J. J. Thompson’s Atom n Thompson passed electric current through gases in a glass tube. The result was a glowing beam flowing from the negative side to the positive side.

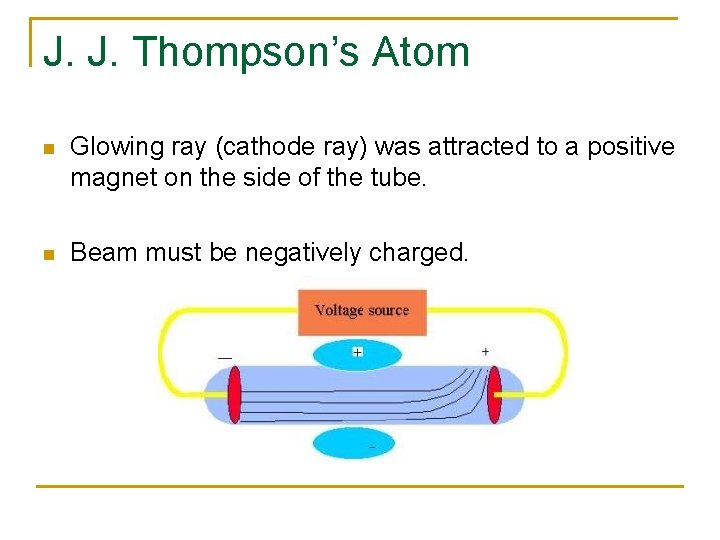
J. J. Thompson’s Atom n Glowing ray (cathode ray) was attracted to a positive magnet on the side of the tube. n Beam must be negatively charged.

J. J. Thompson’s Atom n Thompson hypothesized that all atoms had such negative charges within. (plum pudding model) n Later, scientists proved that atoms contained both positive and negative charges (which balance each other out).

Electrons and Protons n Electrons – negatively charged portion of an atom n Protons – positively charged portion of an atom

James Chadwick’s Atom n Chadwick found that the mass of an atom is greater than the mass of protons plus electrons. n Neutrons – portion of an atom with no charge n Protons, neutrons, and electrons are called subatomic particles.

Ernest Rutherford’s Atom n Born in New Zealand. n Terrible at mathematics. n Diligent problem solver. n Did most of his work at Mc. Gill in Montreal

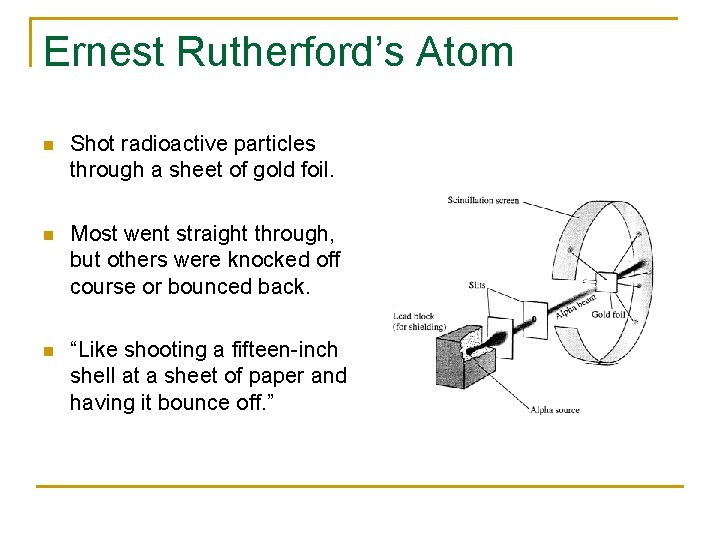
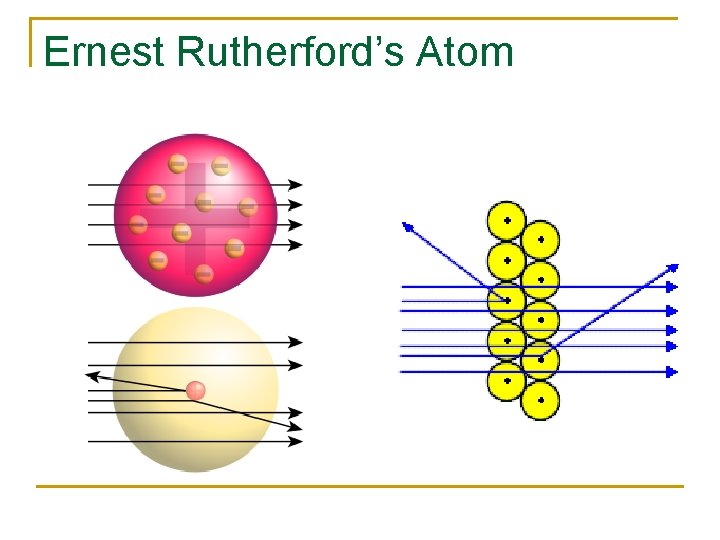
Ernest Rutherford’s Atom n Shot radioactive particles through a sheet of gold foil. n Most went straight through, but others were knocked off course or bounced back. n “Like shooting a fifteen-inch shell at a sheet of paper and having it bounce off. ”

Ernest Rutherford’s Atom

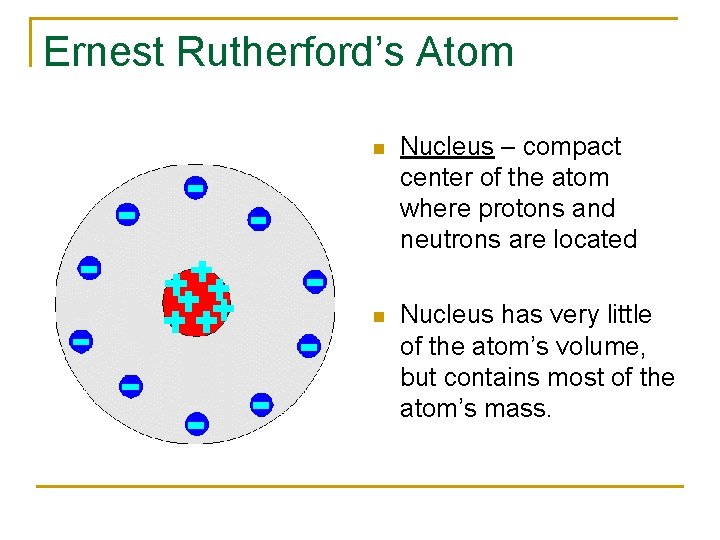
Ernest Rutherford’s Atom n Nucleus – compact center of the atom where protons and neutrons are located n Nucleus has very little of the atom’s volume, but contains most of the atom’s mass.

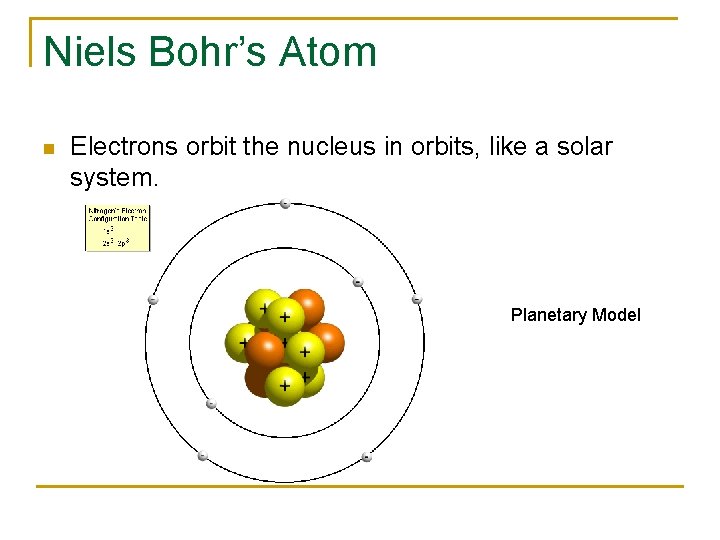
Niels Bohr’s Atom n Electrons orbit the nucleus in orbits, like a solar system. Planetary Model

Atomic Properties How do atoms differ?

Atomic Number n Atoms of different elements have different structures. n Elements differ because their atoms contain different numbers of protons! n Atomic Number – number of protons in the atom

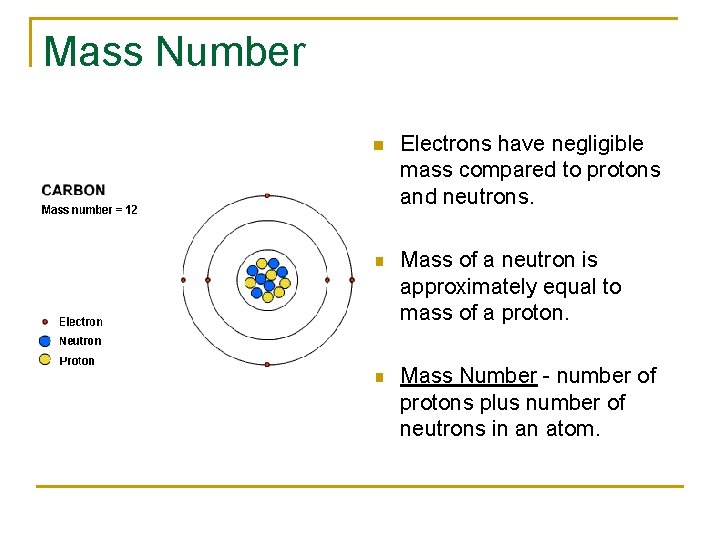
Mass Number n Electrons have negligible mass compared to protons and neutrons. n Mass of a neutron is approximately equal to mass of a proton. n Mass Number - number of protons plus number of neutrons in an atom.

Names and Symbols n Gold – 118 neutrons q n Beryllium – 5 neutrons q n Mass Number = ? Mass Number – ? Sodium – 12 neutrons q Mass Number – ?

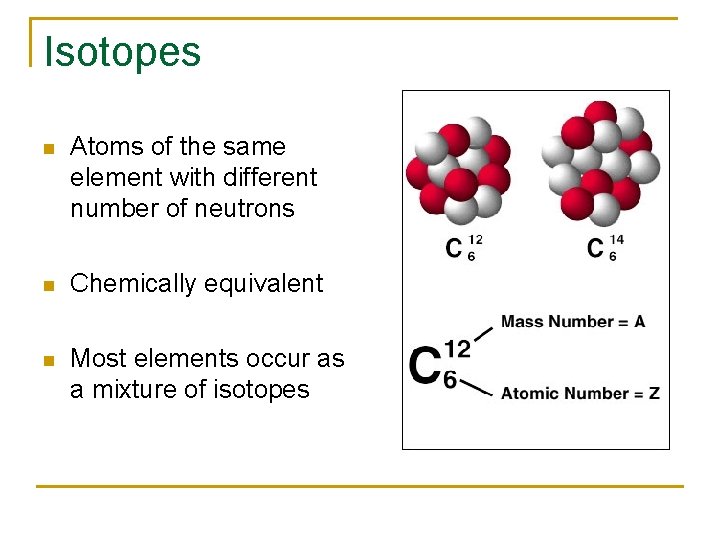
Isotopes n Atoms of the same element with different number of neutrons n Chemically equivalent n Most elements occur as a mixture of isotopes

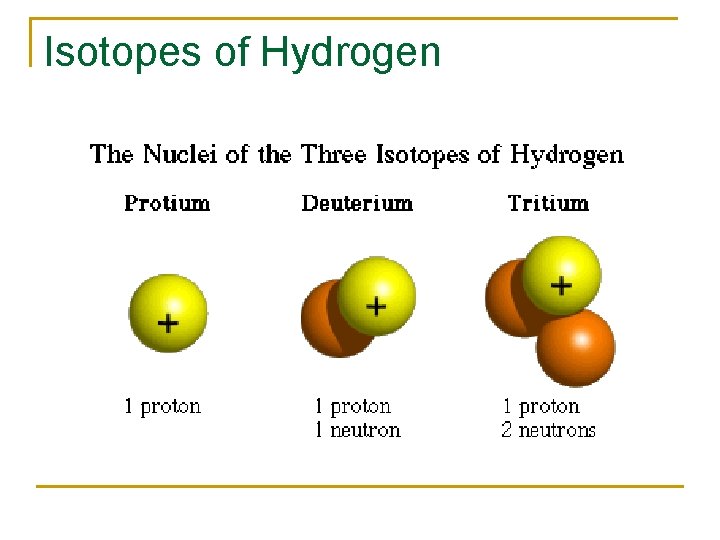
Isotopes of Hydrogen

Calculating Atomic Mass n Mass of a proton is more than 1800 times the mass of an electron n Spectrometer used to find masses of atoms n Masses of subatomic particles too small to work with, so a reference isotope is used

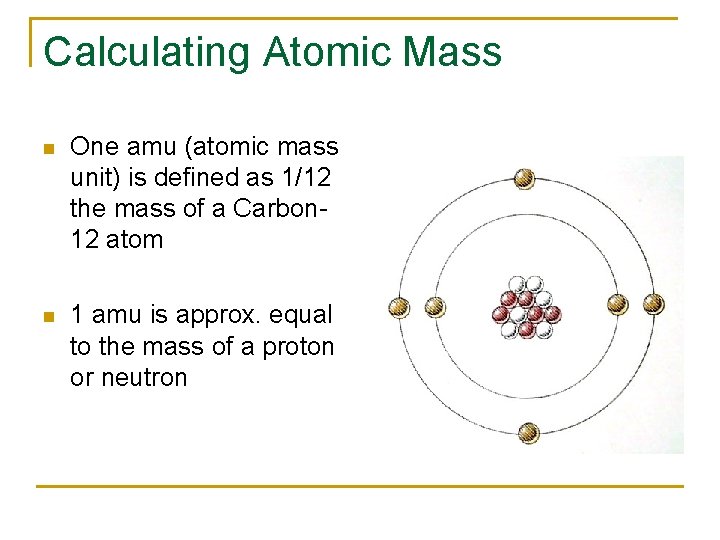
Calculating Atomic Mass n One amu (atomic mass unit) is defined as 1/12 the mass of a Carbon 12 atom n 1 amu is approx. equal to the mass of a proton or neutron

Self Quiz, Part 1 n What is an atom? n What is the symbol for… q q q q Calcium? Phosphorus? Potassium? Copper? Silver? Tin? Mercury?

Self Quiz, Part 2 n What is the atomic number of… q q Lithium? Lead? Gold? Neon? n Describe Dalton’s atomic model. n Name the four premises of Dalton’s atomic theory.

Self Quiz, Part 3 n Explain the difference between atoms and molecules. n Describe Thompson’s experiments and his atomic model. n Describe Rutherford’s experiments and his atomic model. n What is an atomic number?

Self Quiz, Part 4 n Compare and contrast protons, neutrons, and electrons. n Chlorine-35? q q q Mass Number? Number of Protons? Number of Electrons?