Atomic Structure The structure of the atom The




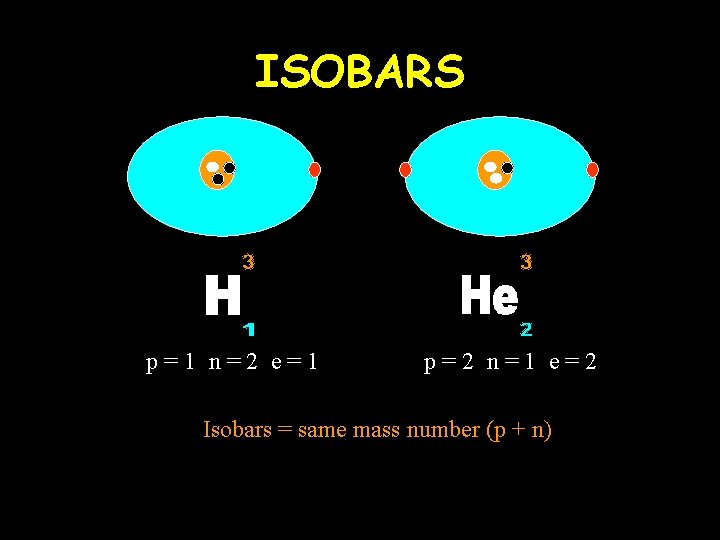

- Slides: 25

Atomic Structure

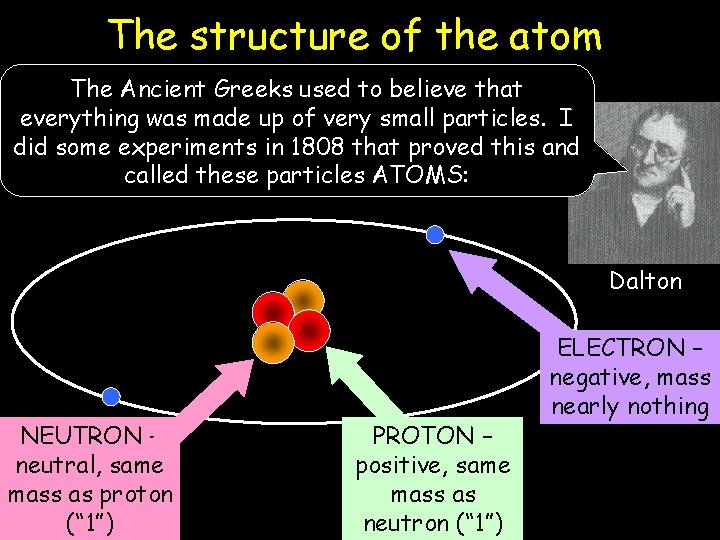
The structure of the atom The Ancient Greeks used to believe that everything was made up of very small particles. I did some experiments in 1808 that proved this and called these particles ATOMS: Dalton NEUTRON – neutral, same mass as proton (“ 1”) PROTON – positive, same mass as neutron (“ 1”) ELECTRON – negative, mass nearly nothing

Mass and atomic number Particle Proton Neutron Electron Relative Mass 1 1 0 Relative Charge 1 0 -1 MASS NUMBER = number of protons + number of neutrons SYMBOL PROTON NUMBER = number of protons (obviously)

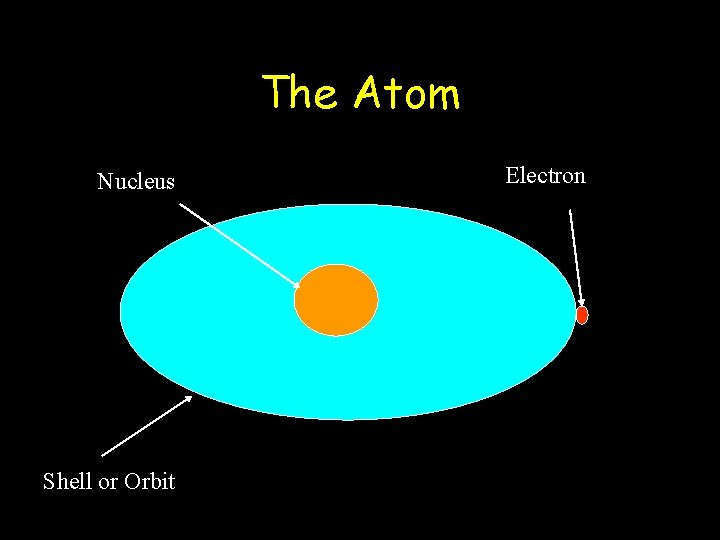
The Atom Nucleus Shell or Orbit Electron

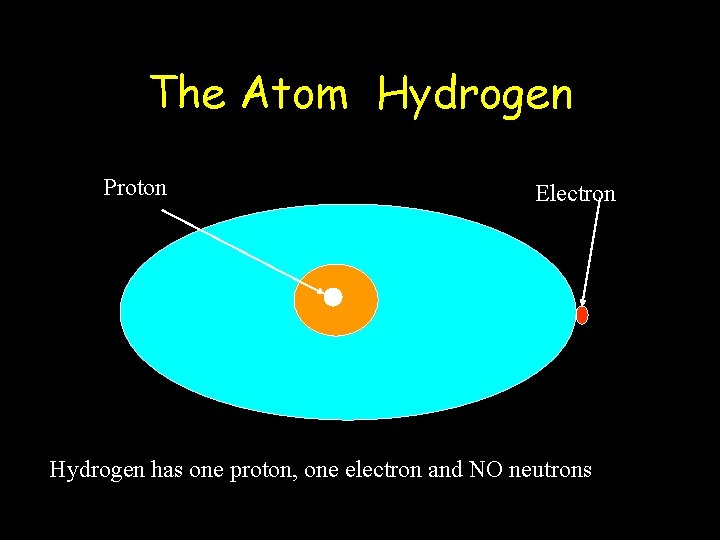
The Atom Hydrogen Proton Electron Hydrogen has one proton, one electron and NO neutrons

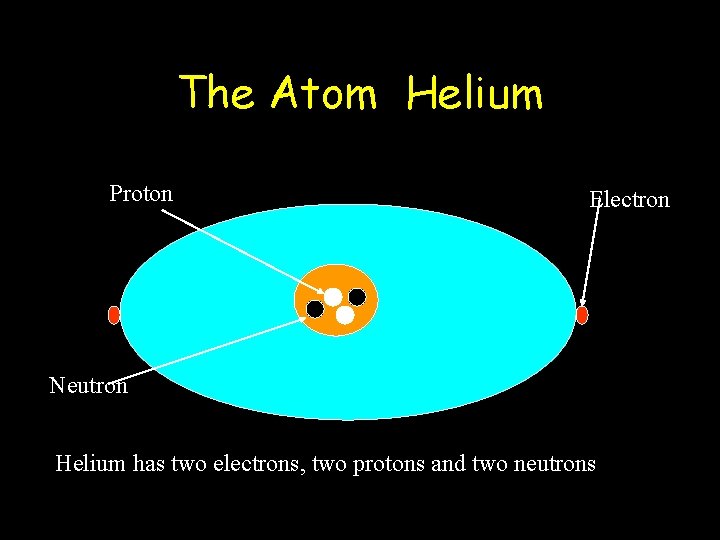
The Atom Helium Proton Electron Neutron Helium has two electrons, two protons and two neutrons

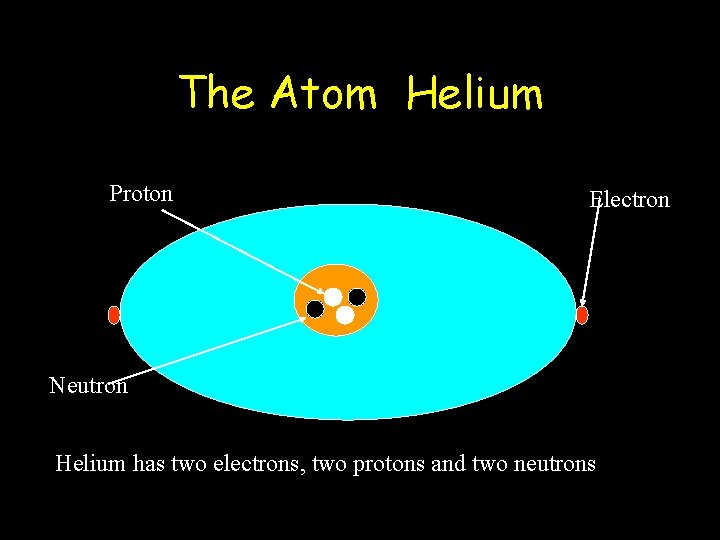
The Atom Helium Proton Electron Neutron Helium has two electrons, two protons and two neutrons

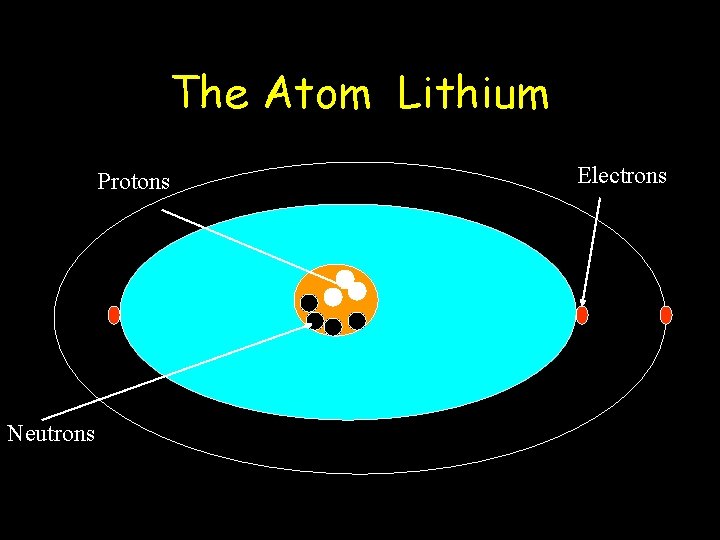
The Atom Lithium Protons Neutrons Electrons

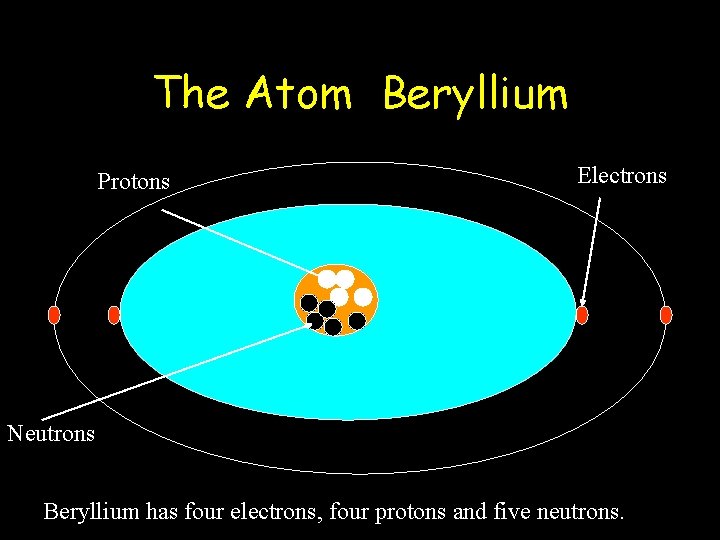
The Atom Beryllium Protons Electrons Neutrons Beryllium has four electrons, four protons and five neutrons.

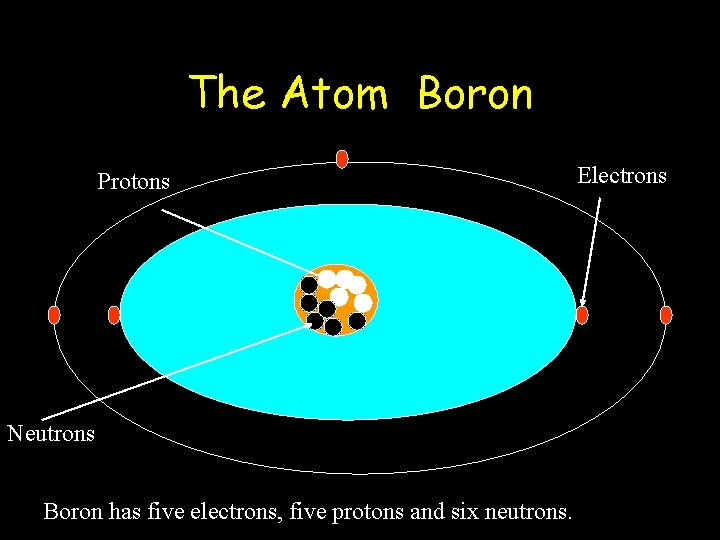
The Atom Boron Protons Neutrons Boron has five electrons, five protons and six neutrons. Electrons

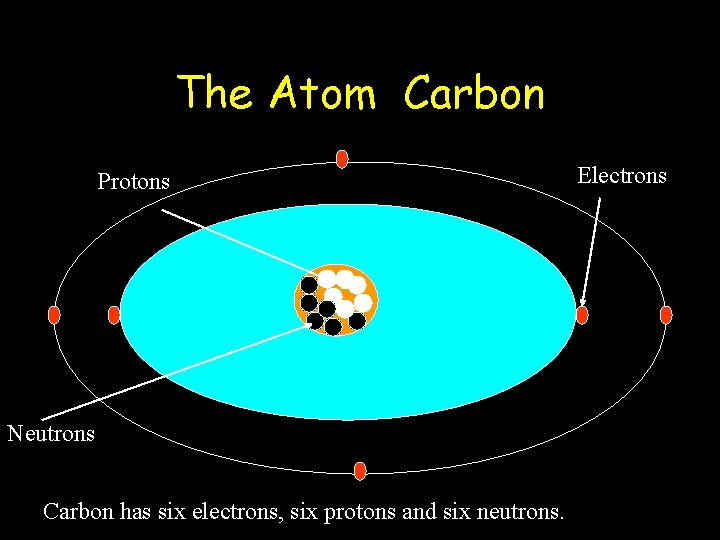
The Atom Carbon Protons Neutrons Carbon has six electrons, six protons and six neutrons. Electrons

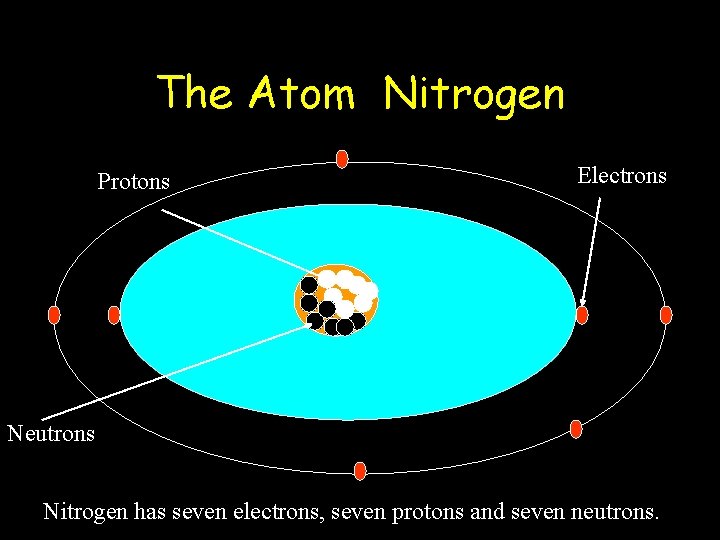
The Atom Nitrogen Protons Electrons Neutrons Nitrogen has seven electrons, seven protons and seven neutrons.

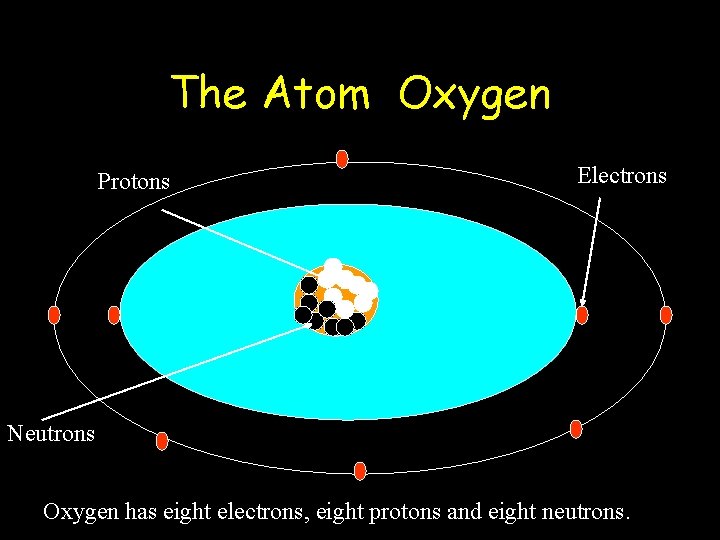
The Atom Oxygen Protons Electrons Neutrons Oxygen has eight electrons, eight protons and eight neutrons.

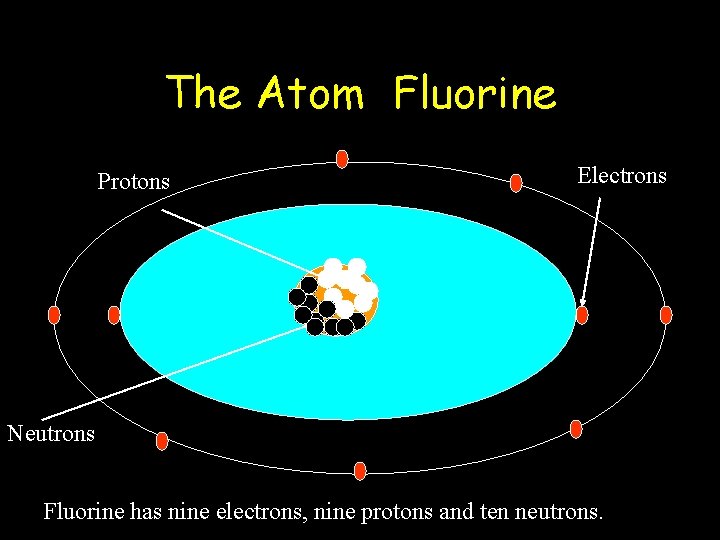
The Atom Fluorine Protons Electrons Neutrons Fluorine has nine electrons, nine protons and ten neutrons.

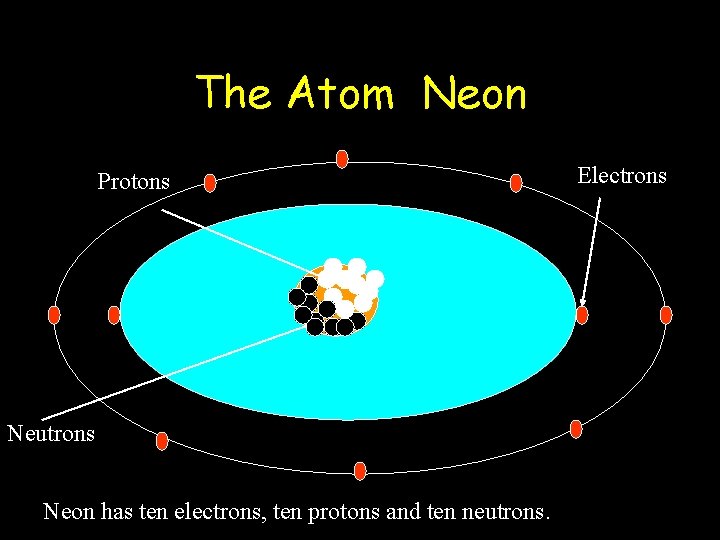
The Atom Neon Protons Neutrons Neon has ten electrons, ten protons and ten neutrons. Electrons

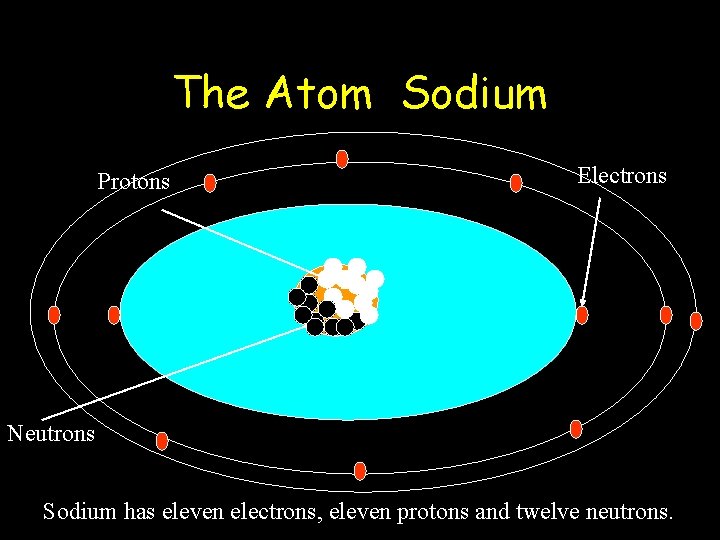
The Atom Sodium Protons Electrons Neutrons Sodium has eleven electrons, eleven protons and twelve neutrons.

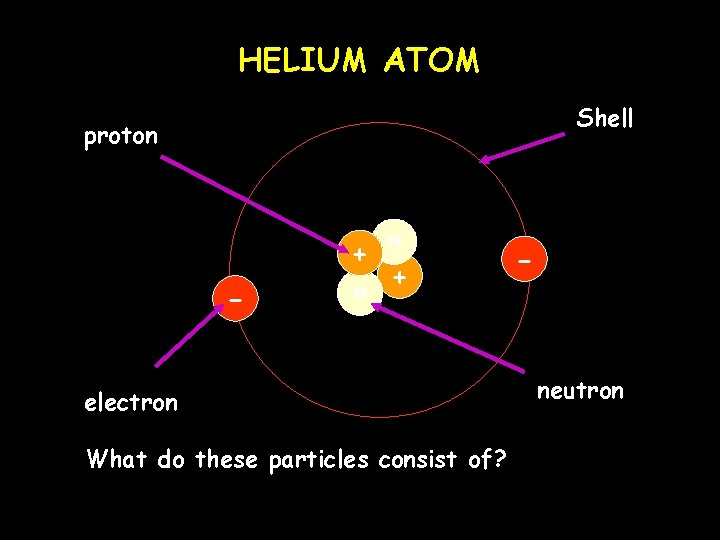
HELIUM ATOM Shell proton + - N N + electron What do these particles consist of? - neutron

How many protons, neutrons and electrons?

Ionic and Sub Atomic Particles Ion = atom or group of atoms who gain or lose electron(s) CATION +1 +2 lose 1 electron in its outer shell Number of proton = 11 Number of electron = 11 – 1 = 10 Number of neutron = 23 – 11 = 12 lose 2 electrons in its outer shell Number of proton = 12 Number of electron = 12 – 2 = 10 Number of neutron = 24 – 12 = 12

Ionic and Sub Atomic Particles ANION -2 -1 gain 2 electron in its outer shell Number of proton = 8 Number of electron = 8 + 2 = 10 Number of neutron = 16 – 8 = 8 gain 1 electrons in its outer shell Number of proton = 17 Number of electron = 17 + 1 = 18 Number of neutron = 35 – 17 = 18

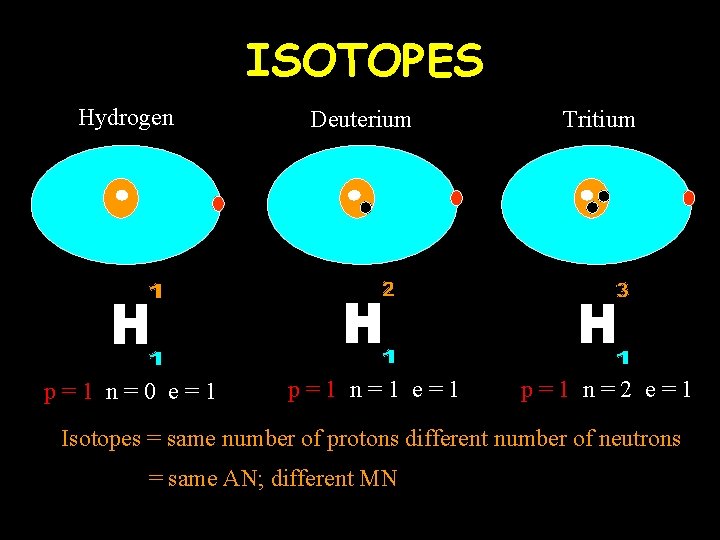
ISOTOPES Hydrogen p=1 n=0 e=1 Deuterium p=1 n=1 e=1 Tritium p=1 n=2 e=1 Isotopes = same number of protons different number of neutrons = same AN; different MN
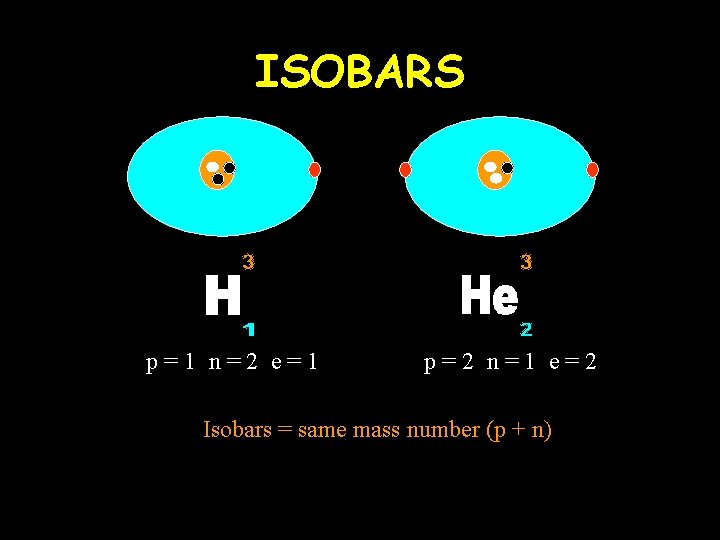
ISOBARS p=1 n=2 e=1 p=2 n=1 e=2 Isobars = same mass number (p + n)

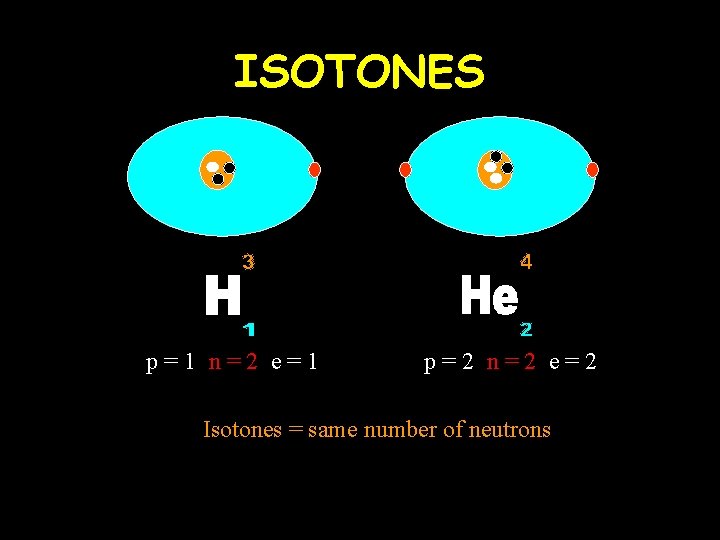
ISOTONES p=1 n=2 e=1 p=2 n=2 e=2 Isotones = same number of neutrons

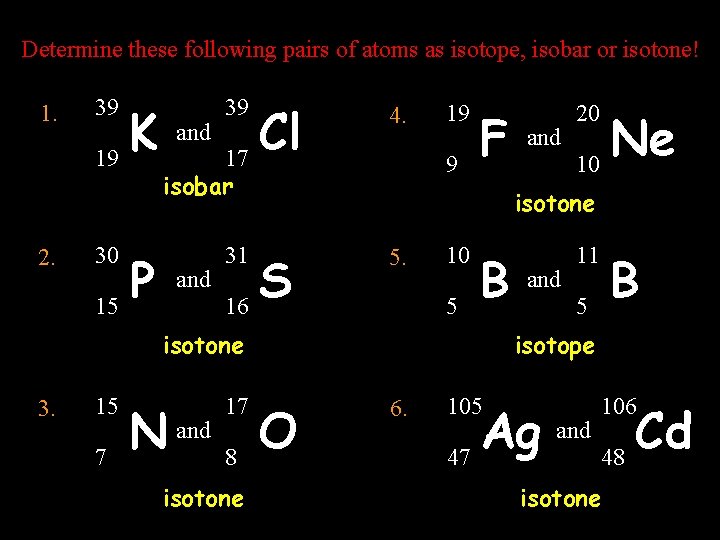
Determine these following pairs of atoms as isotope, isobar or isotone! 1. 2. 39 K 19 30 P 15 and 39 Cl 17 4. 9 isobar and 19 F 15 31 S 16 7 N and 17 8 isotone Ne 10 isotone 5. 10 5 B isotone 3. and 20 and 11 B 5 isotope O 6. 105 Ag 47 and 106 Cd 48 isotone

Go To Worksheet