Atomic Structure The Periodic Table Atomic Theory Whats

Atomic Structure, The Periodic Table & Atomic Theory What’s it all about?

The Basic Atom �Smallest particle of an element that still has the element’s properties �Atomic Theory explains what atoms look like, and how and why they behave the way they do �Democritus (400 BC) proposed the existence of atoms (philosophy) �John Dalton (1800) was the first to have scientific evidence to support this � Considered the Father of Modern Atomic Theory

Dalton’s Atomic Theory �Dalton’s experiments showed him several things about the existence of atoms – he had 5 postulates �All matter is made of atoms �Atoms are small, indestructible spheres �Atoms of the same element are identical �Atoms always combine in compounds in whole number ratios �In chemical reactions atoms are combined, separated or rearranged

Dalton’s Model �Dalton’s idea of the atom �The Marble Model was a small sphere hence

Current model �“building blocks of matter” � Charge: Neutral (no charge) particles �made up of 3 subatomic particles in 2 regions Electron Cloud: • Diffuse (spread out very thinly) • Negatively charged • area outside of the nucleus • Electrons (e-): o each has a (-1) charge o little mass (0 amu) o located outside of the nucleus Nucleus • Tiny • Positively charged • Dense mass in the atom’s center • has 2 particles • (p+) protons o each has a (+1) charge o has a mass of 1 a. m. u. * • (n) Neutrons o no charge o has a mass of 1 a. m. u. * *a. m. u. = atomic mass unit
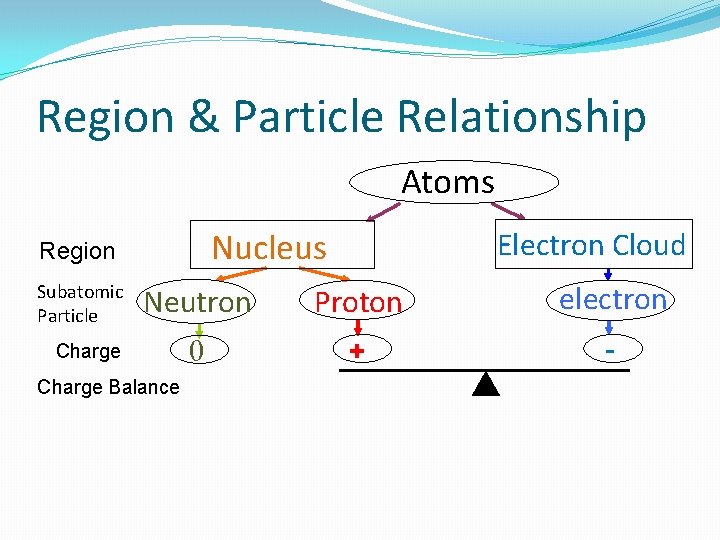
Region & Particle Relationship Atoms Nucleus Region Subatomic Particle Electron Cloud Neutron Proton electron 0 + - Charge Balance

Atomic Particles The Electron

The Electron– Fun Facts �Abbreviated e�Gives the electron cloud its charge �Gives the element its chemical properties �Structure: Lepton (fundamental particle not made of smaller particles) �Mass = 0 amu* (amu = atomic mass unit) �Not really 0 but 1/1200 -1/2000 th the mass of a proton) �Charge: negative (exactly balances the p+ positive charge) �Location: Electron Cloud

The Electron on the Periodic Table How many electrons are there in an element? ? Where do you find it? ? ? # of e- = Atomic Number but only in a neutral atom 20 Ca Calcium 40. 078 Let’s make a note on our PT & test each other

The Electron – History/Theory �Discovery #: 1 st subatomic particle discovered �Discovered by JJ Thomson in 1897 �JJ Made a CRT (cathode ray tube) and found that when he shot electricity through it a beam of light appeared �The beam of light was affected by magnetic & electric forces (does light do that? ? ) �He proposed that it wasn’t light but tiny, negatively charged particles he called electrons (btw: he thought the discovery was useless)

Thomson’s Model �The atom was �Neutral in charge so… � Most of the atom was a diffuse positively charged “goo” or matrix � Electrons were tiny particles that floated around in the “goo”. �Electrons were very tiny compared to the mass of the atom

The Thomson Plum Pudding Model 1897 – The Plum Pudding Model I prefer the “Chocolate Chip Cookie Dough Model negative electrons positive matrix or goo that made up the rest of the atom

Atomic Particles The Proton

The Proton – Fun Facts �Abbreviated p+ �Gives the nucleus a positive charge �Gives the element its unique characteristics & identifies the element (# p + is unique to each element �Structure: Hadron (composite particle made of smaller particles) �Mass = 1 amu* �Charge: Positive �Location: Nucleus

The Proton on the Periodic Table How many protons are there in an element? ? Where do you find it? ? ? # of p+ = Atomic Number 20 Ca Calcium 40. 078 Let’s make a note on our PT & test each other

The Proton – History/Theory �Discovery #: 2 nd subatomic particle discovered �Discovered by Ernest Rutherford in 1918 �Rutherford first proposed the positive nucleus in 1911 from the “Gold Foil Experiment” changing the model of the atom again �http: //www. mhhe. com/physsci/chemistry/essentialch emistry/flash/ruther 14. swf

Rutherford’s Models �The nucleus was �Very dense �Had most of the mass of the atom �Was positive �Very, very tiny compared to the entire atom �Atoms are mostly empty space �The electrons move around the outside of the nucleus �Rutherford model – nuclear model – kind of like a plasma ball w/ a tiny positive nucleus and electrons floating around outside it �Discovered the proton later in the first nuclear reaction �How tiny? 400 x tinier than this – and that’s only the nucleus!!! Protons are smaller yet!!!!

The Rutherford (Nuclear) Models 1911 Model with just a positive nucleus in the center of the electrons 1918 Model with protons making up the nucleus

Atomic Particles The Neutron

The Neutron – Fun Facts �Abbreviated n 0 �Adds mass to the nucleus �Helps hold the nucleus together (prevents proton repulsion from destroying the nucleus) �Structure: Hadron (composite particle made of smaller particles) �Mass = 1 amu* �Charge: Neutral (no charge) �Location: Nucleus

The Neutron on the Periodic Table How many neutrons are there in an element? ? Where do you find it? ? ? #n 0 is NOT found on the PT 20 Ca Calcium 40. 078 More on this later

The Neutron – History/Theory �Discovery #: 3 rd subatomic particle discovered �Discovered by James Chadwick (also a student of JJ Thomson) in 1936 – (Rutherford said it should be there, Chadwick gave them evidence) �Chadwick produced a beam of neutral particles that knocked protons out of paraffin wax �https: //www. youtube. com/watch? v=Hnm. EI 94 URK 8 �Led to the development of the atomic bomb

Chadwick’s Model �Similar to the current model with p+ & n 0 in the nucleus and e- orbiting the nucleus in the electron cloud

Atomic Particles Isotopes & The Neutron

Isotopes & the Neutron �Isotopes: Atoms of the same element with different numbers of neutrons �The number of neutrons can be different in an element (that’s why there is no neutron number on the PT) �The number of neutrons must be calculated for each isotope �Example: Magnesium has 3 isotopes �Symbol #p+ #n 0 #e. Mg - 24 12 12 12 Mg - 25 13 12 12 Mg - 26 14 12 12

Isotopes & Mass Number �Mass number (mass #) is the number of p+ & no in a specific isotope of an element �Mass # = #p+ + no �or #no = Mass # - #p+ �Lets put this on the PT somewhere

Isotopes & Mass Number #1 How do you know which isotope you have? ? Mass # is found in one of 2 ways – prioritized list 1. Isotope Notation • (given to you with the element symbol) – 100% accurate Element Symbol C-14 Mass # OR 14 Atomic # 6 C Element Symbol So in C-14 there are… • 6 p+ & 6 e- (atomic number of C is 6) • 8 n 0 (Mass # - # p+ = #n 0 or 14 - 6 = 8 )
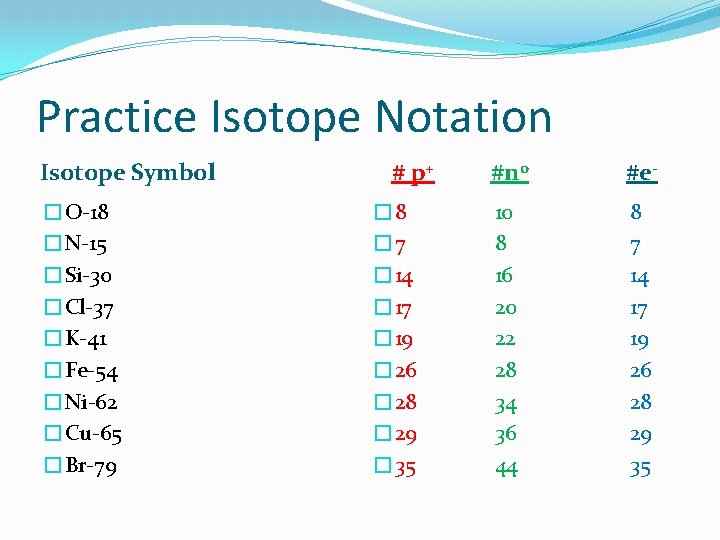
Practice Isotope Notation Isotope Symbol �O-18 �N-15 �Si-30 �Cl-37 �K-41 �Fe-54 �Ni-62 �Cu-65 �Br-79 # p+ � 8 � 7 � 14 � 17 � 19 � 26 � 28 � 29 � 35 #n 0 #e- 10 8 16 20 22 28 34 36 44 8 7 14 17 19 26 28 29 35

Isotopes & Mass Number #2 How do you know which isotope you have? ? 2. MCI (most common isotope) – 50% accuracy • Round the Average Mass to a whole number to get mass # for the m. c. i. 20 Ca Average mass = 40. 078 amu so…. round it and … The mass number of the most common isotope is 40 so Ca-40 Calcium 40. 078 Average mass = 10. 806 amu so…. round it and … The mass number of the most common isotope is 11 so B-11 5 B Boron 10. 806
Average Mass
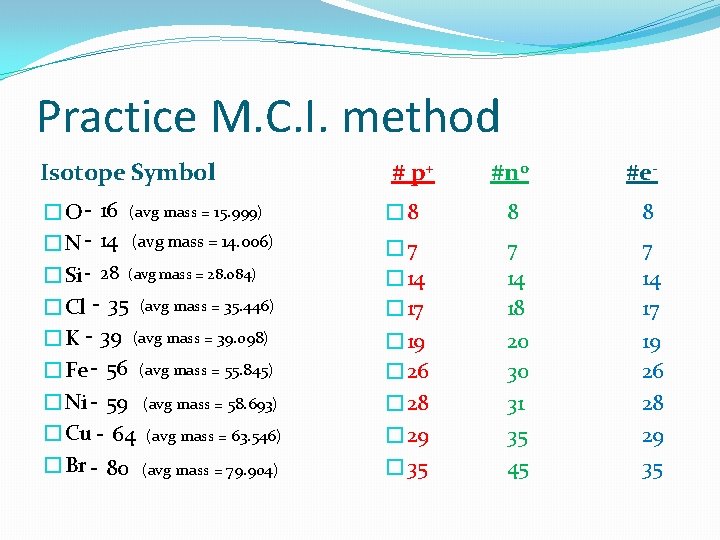
Practice M. C. I. method Isotope Symbol �O - 16 (avg mass = 15. 999) �N - 14 (avg mass = 14. 006) �Si - 28 (avg mass = 28. 084) �Cl - 35 (avg mass = 35. 446) �K - 39 (avg mass = 39. 098) �Fe - 56 (avg mass = 55. 845) �Ni - 59 (avg mass = 58. 693) �Cu - 64 (avg mass = 63. 546) �Br - 80 (avg mass = 79. 904) # p+ #n 0 #e- � 8 8 8 � 7 � 14 � 17 7 14 18 7 14 17 � 19 � 26 20 30 31 35 45 19 26 28 29 35 � 28 � 29 � 35
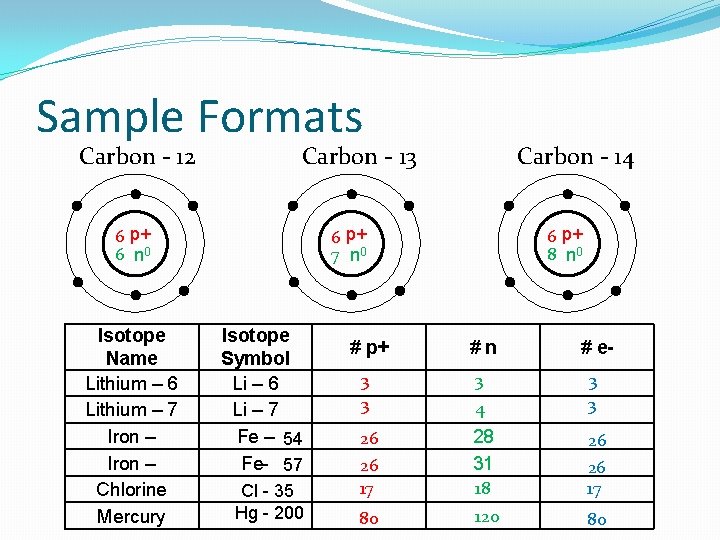
Sample Formats Carbon - 12 Carbon - 13 6 p+ 6 n 0 Isotope Name Lithium – 6 Lithium – 7 Iron – Chlorine Mercury Carbon - 14 6 p+ 7 n 0 Isotope Symbol Li – 6 Li – 7 Fe – 54 Fe- 57 Cl - 35 Hg - 200 6 p+ 8 n 0 # p+ #n # e- 3 3 3 26 28 31 26 17 80 4 26 18 26 17 120 80

Atomic Particles The Electron Cloud
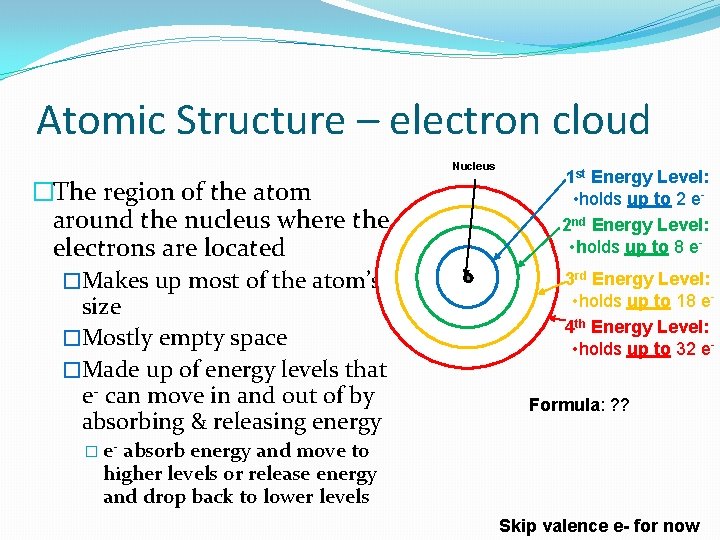
Atomic Structure – electron cloud Nucleus �The region of the atom around the nucleus where the electrons are located �Makes up most of the atom’s size �Mostly empty space �Made up of energy levels that e- can move in and out of by absorbing & releasing energy 1 st Energy Level: • holds up to 2 e 2 nd Energy Level: • holds up to 8 e 3 rd Energy Level: • holds up to 18 e 4 th Energy Level: • holds up to 32 e. Formula: ? ? � e- absorb energy and move to higher levels or release energy and drop back to lower levels Skip valence e- for now

Atomic Theory – Electron Cloud �Energy levels were proposed by Neils Bohr in 1913 �His data supported that e- absorb heat/electrical energy & then release the energy as certain colors of light �He concluded that e- orbited the nucleus in energy levels �Jumped to higher levels when absorbing energy �Released light energy when they fell back down �https: //www. youtube. com/watch? t=175&v=CUk 3 enr-m 0 w

Atomic Model– Bohr Model �Neils Bohr’s model is sometimes called the planetary model but we usually call it the… �Bohr Model light released e. l. = 1 e. l. = 2 e. l. = 3 Time for a demo

Electron Cloud – Later Discoveries �In 1926 Erwin Schrodinger & Max Born proposed that electrons actually behaved more like waves than particles and therefore proposed the wave model. Schrodinger is famous for his “Schrodinger’s Cat” example used to explain this model � The quantum mechanical model of the electron cloud is our current model and many scientists have contributed to its development. � discovered e- exist within orbitals, which are not circular, within the energy levels � dark, shaded areas of the cloud are more likely to contain e-
- Slides: 37