Atomic Structure The Atom Proton Positively charged subatomic

Atomic Structure
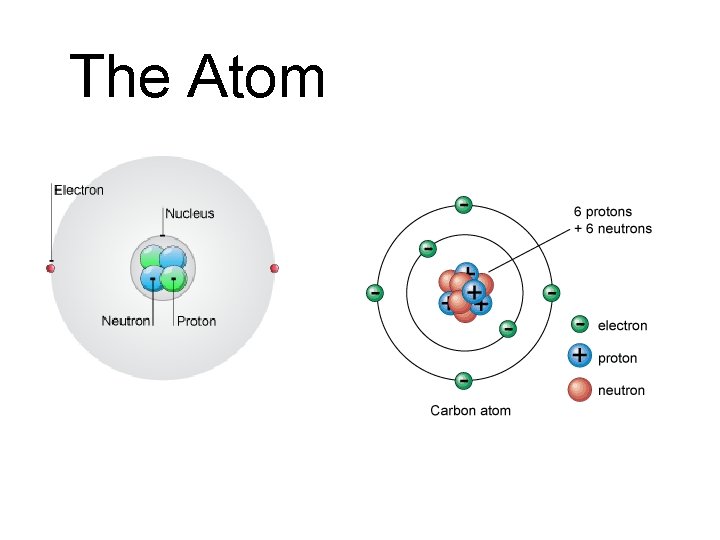
The Atom

Proton Positively charged subatomic particle • Symbol: p+ • Relative Mass: 1 • Charge: +1 • Location: nucleus

Neutron Subatomic particle with no charge • Symbol: n 0 • Relative Mass: 1 • Charge: 0 • Location: nucleus

Electron Negatively charged subatomic particle • Symbol: e− • Relative Mass: 0 • Charge: − 1 • Location: outside the nucleus

Atomic Variation
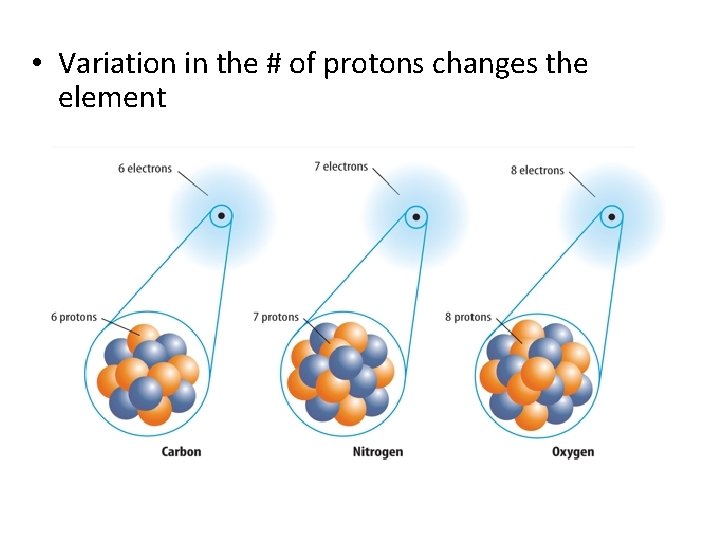
• Variation in the # of protons changes the element

Ions Variation in the number of electrons Anion • gain of electrons • overall charge is negative F 1− Cation • lose of electrons • overall charge is positive 11 protons 12 neutrons 11 electrons 11 protons 12 neutrons 10 electrons Sodium atom Sodium ion 1+ Na
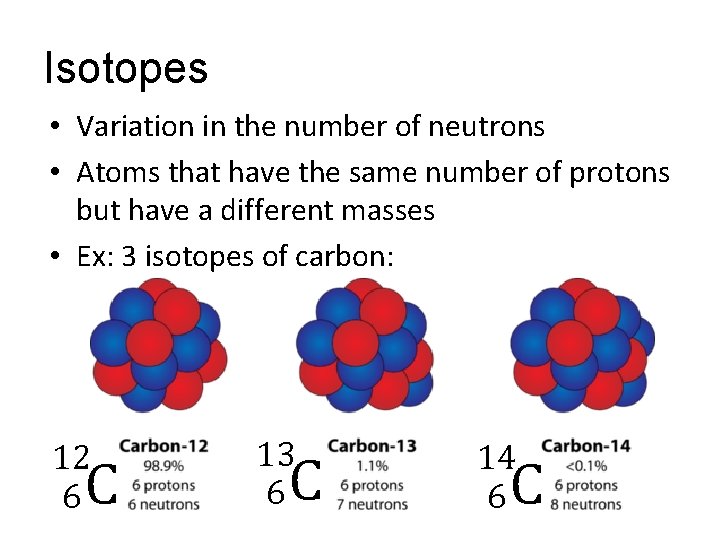
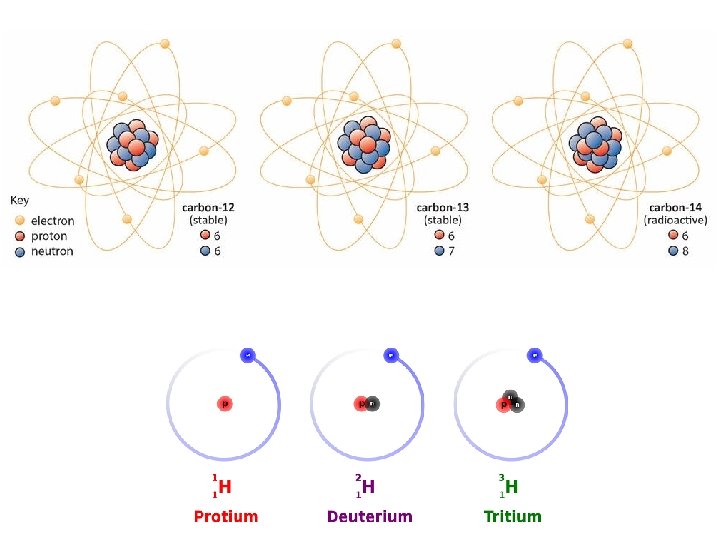
Isotopes • Variation in the number of neutrons • Atoms that have the same number of protons but have a different masses • Ex: 3 isotopes of carbon: 12 6 C 13 6 C 14 6 C

Nuclide Symbol A Z X Atomic Number Mass Number • the number of protons in an atom • Identifies element • Symbol: Z • Represents the total number of protons and neutrons in the nucleus (p+ + n 0) • Symbol: A Charge • States whether electrons were gained or lost from the neutral atom • Helps indicate chemical bonding • Symbol: ± # ±# Chemical Symbol • Short-hand abbreviation for the element name • Only first letter should be capital


Naming Isotopes • State the element name or symbol followed by a hyphen and the mass number • Ex: Carbon— 14

Practice 1. How many protons, neutrons and electrons are in Cobalt— 59? 1. How many protons, neutrons and electrons are in 2. Write the nuclide symbol and name of an isotope that has 15 protons, 17 neutrons, and 18 electrons. 3. Write the nuclide symbol of an isotope that has 18 protons, 22 neutrons, and 18 electrons.

Atomic Mass • Mass of a single atom in atomic mass units (amu)
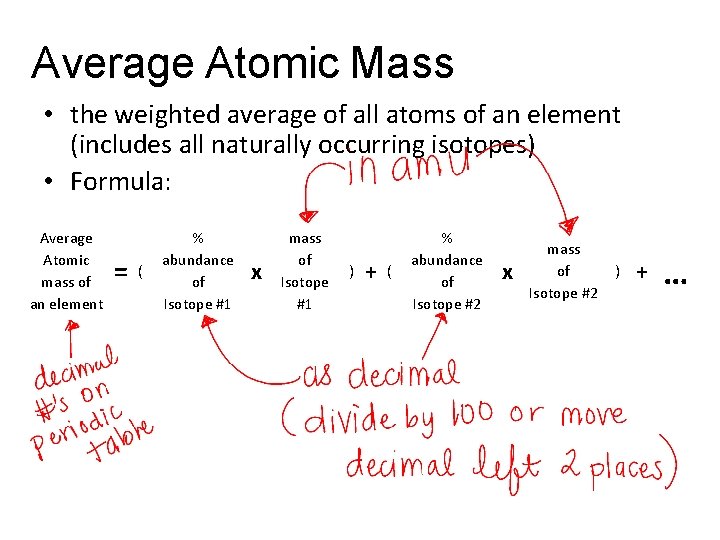
Average Atomic Mass • the weighted average of all atoms of an element (includes all naturally occurring isotopes) • Formula: Average Atomic mass of an element = ( % abundance of Isotope #1 x mass of Isotope #1 ) + ( % abundance of Isotope #2 x mass of Isotope #2 ) + …

Example 1 Silver has two naturally occurring isotopes. Ag-107 has an abundance of 51. 82% and mass of 106. 9 amu. Ag-109 has a relative abundance of 48. 18% and a mass of 108. 9 amu. Calculate the average atomic mass of silver.

Example 2 Rubidium is a soft, silvery-white metal that has two 87 common isotopes, 85 Rb and Rb. 37 37 If the abundance of 85 Rb is 72. 7% with an atomic mass of 84. 91 amu and the abundance of 87 Rb is 27. 8% with an atomic mass of 86. 91 amu, what is the average atomic mass of rubidium?
- Slides: 18