Atomic Structure Rutherford Scattering Experiment Background Plum Pudding
Atomic Structure Rutherford Scattering Experiment

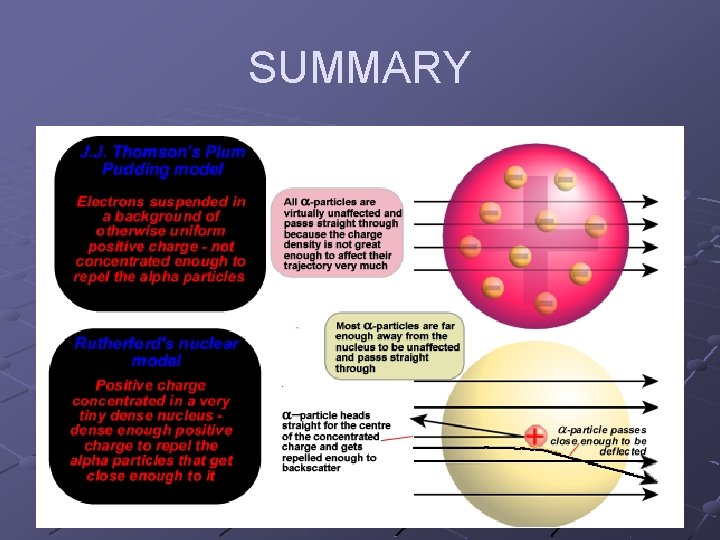
Background: Plum Pudding Model Thomson discovers the electron. Atoms electrically neutral and have e-’s. Atom must contain equal # of __ charge. His model: negative e- in spread out cloud of + charge, hence: plums in pudding.
Analogy to Rutherford’s Experiment What would be seen from the other side of the tent:
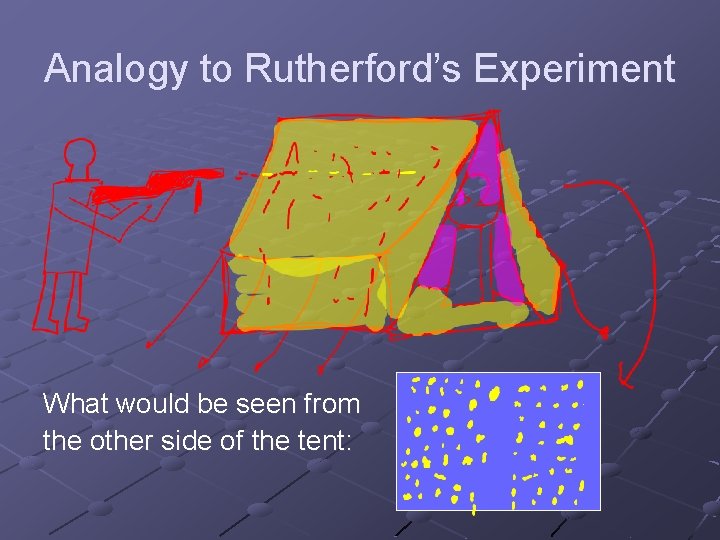
Analogy to Rutherford’s Experiment What would be seen from the other side of the tent:

Shadow Image
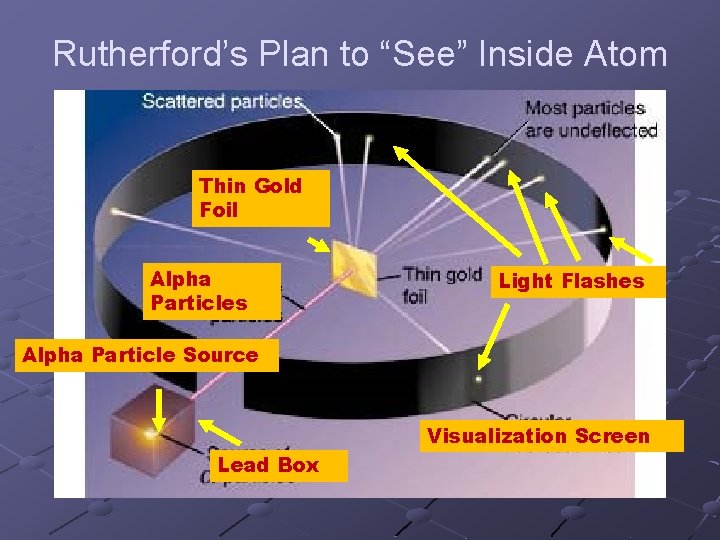
Rutherford’s Plan to “See” Inside Atom Thin Gold Foil Alpha Particles Light Flashes Alpha Particle Source Visualization Screen Lead Box

HW 1 -6 1) Why a very thin sheet of gold foil instead of a thick gold brick? Gold can be pounded into very thin FOIL (0. 0000005 m) VERY LIKELY AT LEAST SOME ALPHA PARTICLES CAN PASS THROUGH THIN FOIL BRICK IS TOO THICK AND DENSE NO ALPHA PARTICLES CAN PASS THROUGH. IF NO PARTICLES PASS THROUGH – NO INFORMATION ABOUT INSIDE CAN BE DEDUCED.

To see image at least some light must be able to pass through The experiment only works if a shadow is cast on the other side with some passing through and some blocked.

HW 1 -6, #2 Characteristic of Alpha Particles The relative sizes of an alpha particle and a gold atom are shown below. How does the size of an alpha particle compare to a gold atom? (Does the probe particle need to be bigger or smaller than the atom it is attempting to pass through? ) Ans: Alpha particles are much smaller than gold atoms and therefore should be able to pass through the gold atoms.

HW 1 -6, #3 Based on the data below, what kind of charge does an alpha particle have? What kind of charge would repel an alpha particle? Alpha particles (blue line) are attracted to the negative and repelled by + charge, thus Alpha particles must be +.
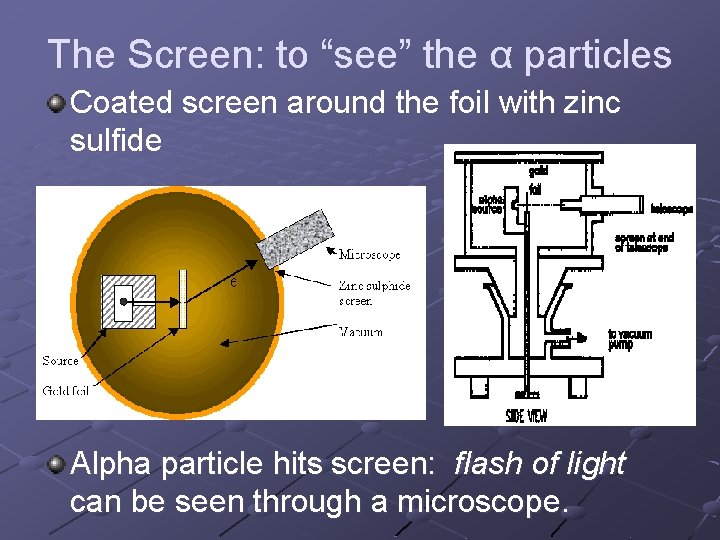
The Screen: to “see” the α particles Coated screen around the foil with zinc sulfide Alpha particle hits screen: flash of light can be seen through a microscope.
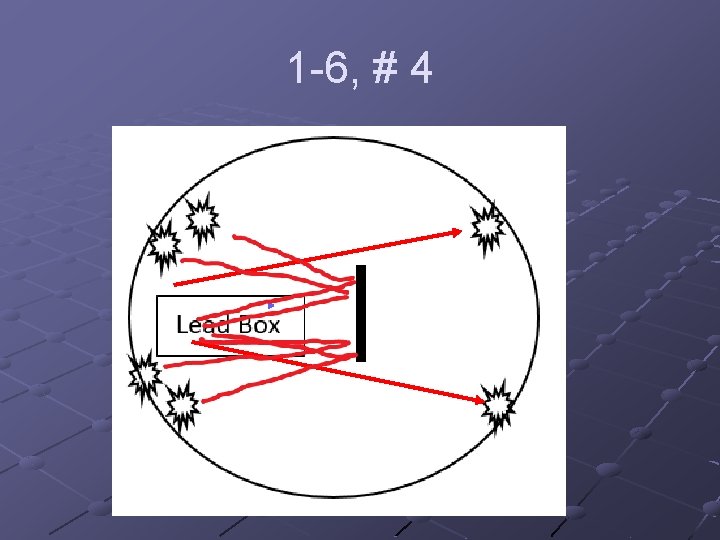
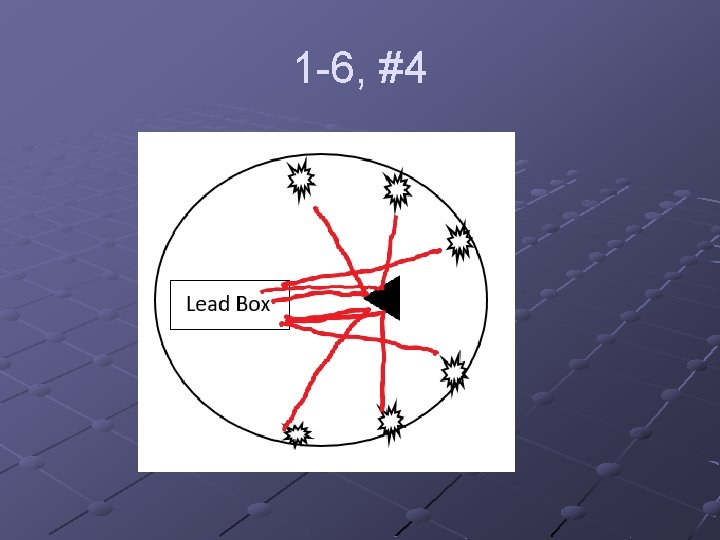
1 -6, #4
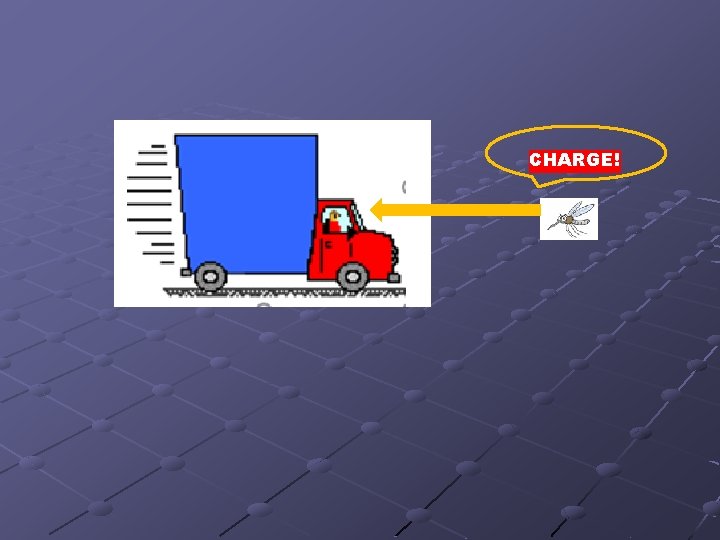
CHARGE!
SPLAT!!

View from inside truck Mosquito (after collision) Momentum Truck >>> Momentum Mosquito

HW 1 -6, #6 Since mass of alpha particle is 7500 X greater than electron MOMENTUM ALPHA >>> ELECTRON In a collision with a tiny electron, the path of the massive α particles would not be altered or changed.

HW 1 -6, #7 Analogy: Titantic passes through spread out cloud of water vaper, but not packed ice. In Plum Pudding Model alpha particles should pass through spread out cloud of positive charge.

Expected Results If the Plum Pudding Model was correct, α particles would pass through the gold atoms undeflected. α particles would pass through diffuse + “pudding” (like a plane through a cloud). α particles traveling too fast and too big to be attracted by e-

Link to Brian Cox Video Link to Rutherford Animation Link to Video with equipment Video clip with + charges
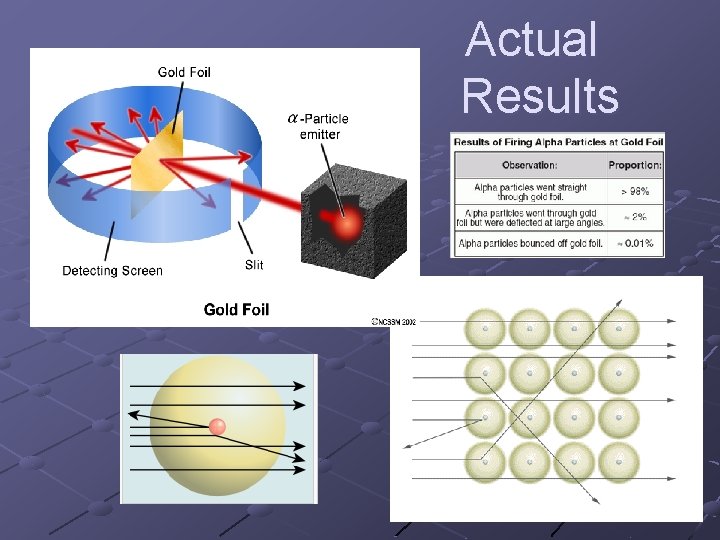
Actual Results

Rutherford’s Conclusions and Supporting Evidence (know these!) Evidence: small number of α particles were scattered. Most passed straight through the Au atoms undeflected. Conclusion: Most of the atom is EMPTY SPACE

Rutherford’s Conclusions and Supporting Evidence (know these!) Ø α particles are + charged. Ø α particles would be repelled by a + charge. Had to be concentrated positive charge to show large deflections. Video clip with + charges Ø Conclusion: ALL ATOMS CONTAIN A TINY, DENSE CONCENTRATION OF POSITIVE CHARGE CALLED THE NUCLEUS.

ALL ATOMS CONTAIN A TINY, DENSE CONCENTRATION OF POSITIVE CHARGE CALLED THE NUCLEUS.

If the nucleus of a hydrogen atom were scaled up to the size of golf ball, what would the average distance of the electron from nucleus be?

Ans: About 1 mile; on this picture the far left edge (approximately) Link to Video with equipment
SUMMARY
- Slides: 27