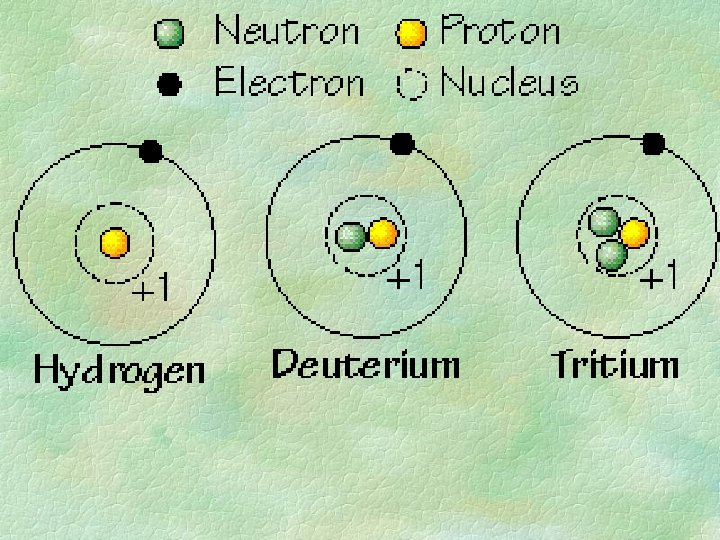
ATOMIC STRUCTURE Label the following Nucleus protons neutrons
ATOMIC STRUCTURE
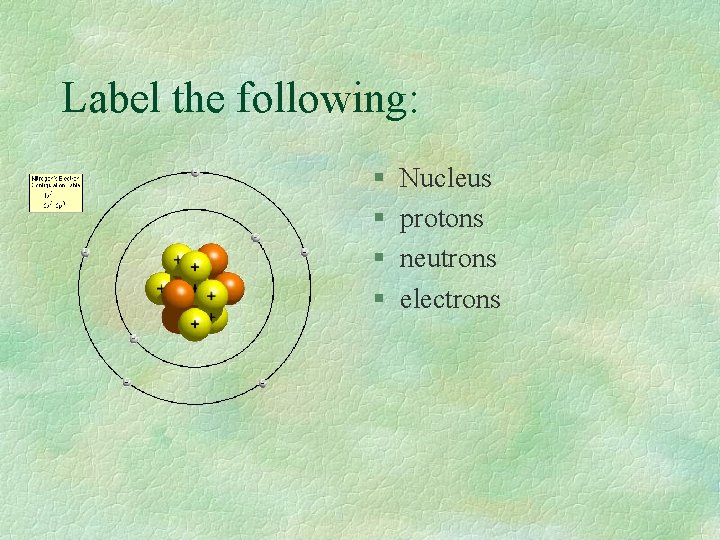
Label the following: § § Nucleus protons neutrons electrons
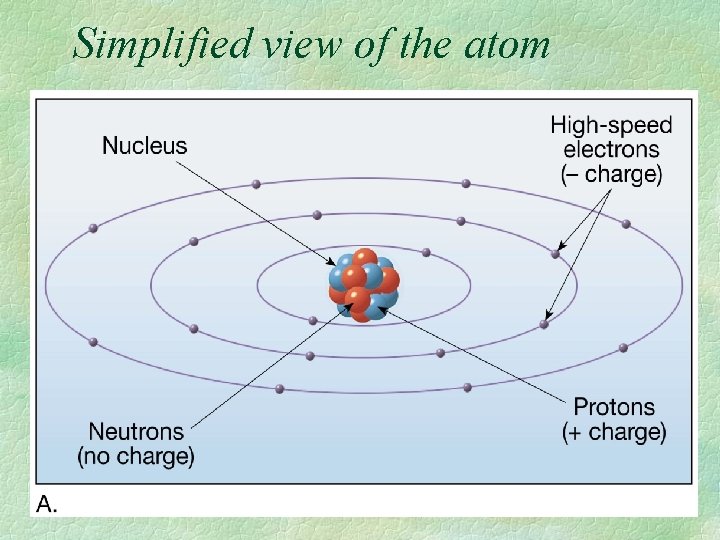
Simplified view of the atom

Protons = positive charge + electrons= negative charge neutrons= no charge 0

ATOMIC MASS § § Inside the nucleus (mainly) protons + neutrons unit is AMU find on periodic table (round to whole #)
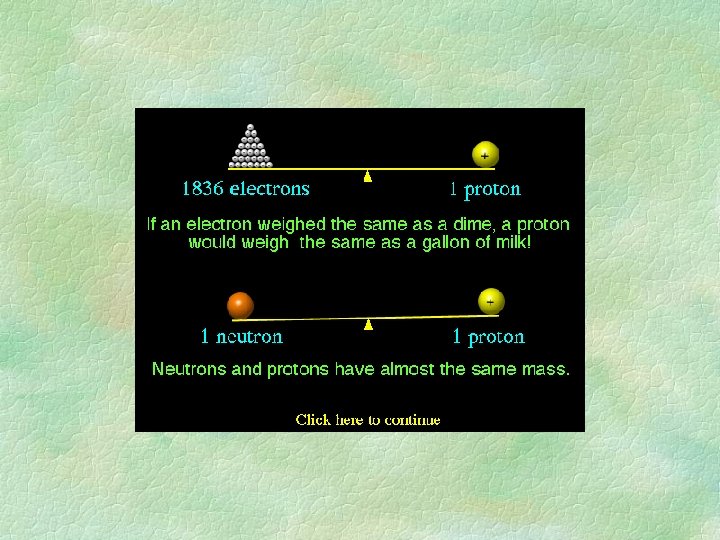

ATOMIC NUMBER § Number of protons § cannot change! § Identifies element

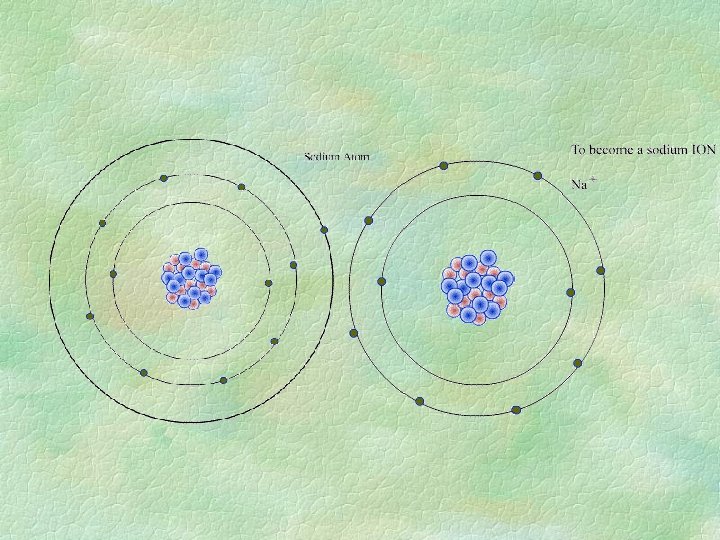
IONS § Charged atoms: NOT neutral § p> e positive § p< e negative § example: 11 p 11 e 12 n neutral Na » 11 p 10 e 12 n ion Na+

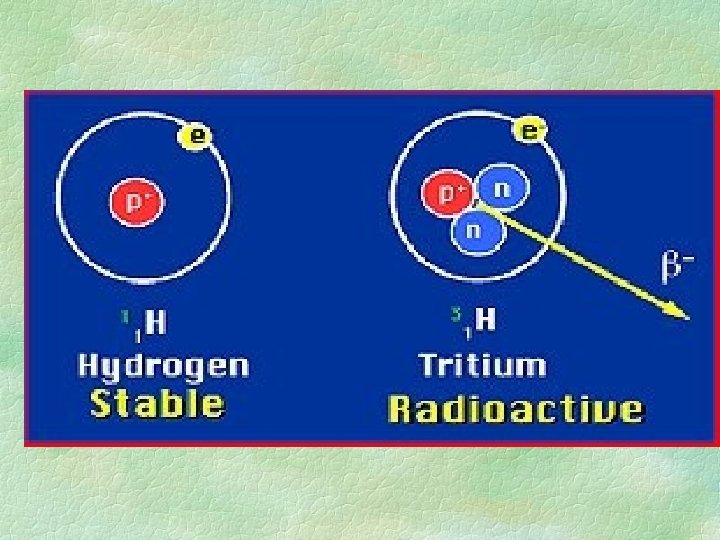
ISOTOPES § Different number of neutrons than stable atom § usually radioactive § atomic mass is different than stable atom § example: 234 U
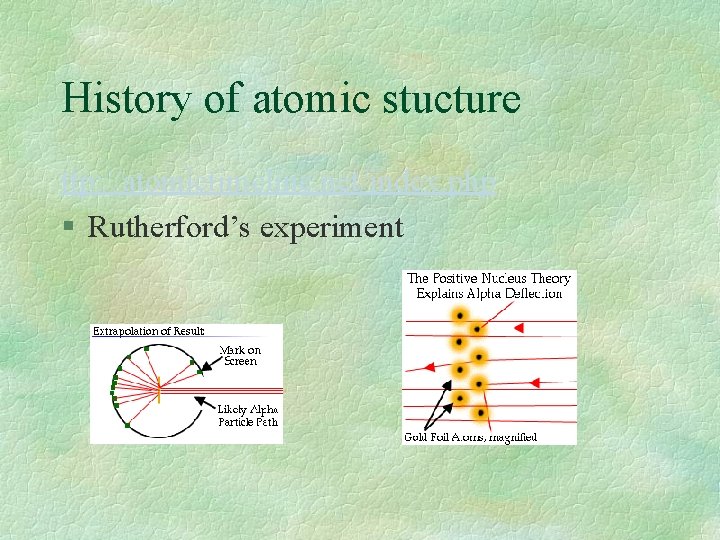
History of atomic stucture ttp: //atomictimeline. net/index. php § Rutherford’s experiment

Can we see atoms? § STM- scanning tunneling microscope § Invented 1981 § Shows arrangement of atoms § Gold

§ element- same atoms bonded together § Compound- two (or more) different atoms bonded § Mixtures- two (or more) different materials NOT bonded § Minerals? -

WORKSHEET § § § § P= e= n= atomic mass= atomic number= how to recognize an ion? How to recognize an isotope?
TO CALCULATE # OF NEUTRONS § Atomic mass (p+n) § atomic number (p) § SUBTRACT!

Sample problems § 11 p 12 n 10 e § 19 p 21 n 19 e § 25 p 30 n 21 e § Cr+3 § 234 Pu
- Slides: 22