Atomic Structure Composition of an atom Atoms are


























- Slides: 42

Atomic Structure Composition of an atom Atoms are made up of 3 fundamental subatomic particles: Position in atom Relative electric charge Relative mass Protons (p) nucleus +1 1 Neutrons (n) nucleus 0 1 Electrons (e-) around the nucleus -1 1/ 1840

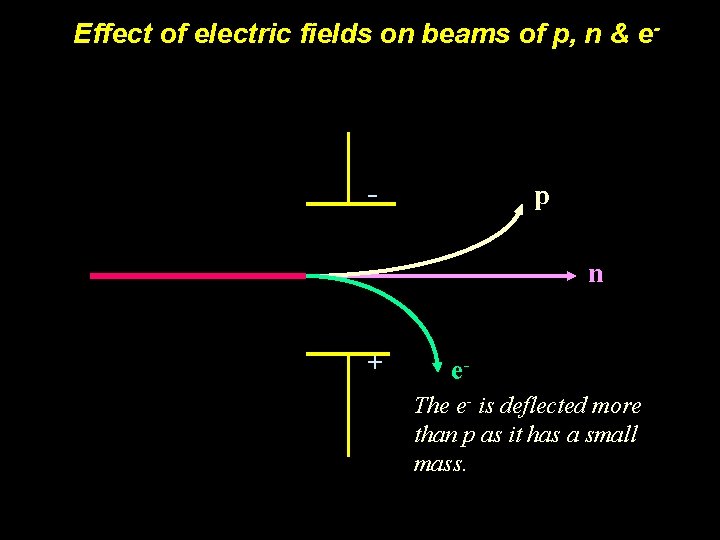
Effect of electric fields on beams of p, n & e. In an electric field: - p n + e. The e- is deflected more than p as it has a small mass.

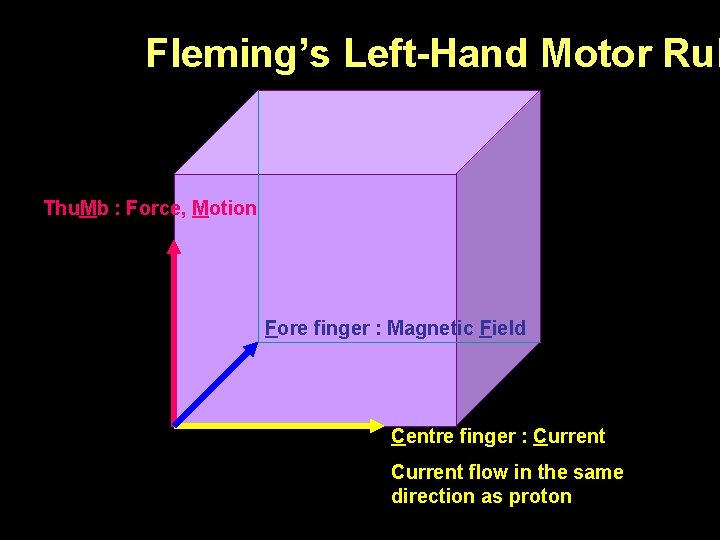
Fleming’s Left-Hand Motor Rul Thu. Mb : Force, Motion Fore finger : Magnetic Field Centre finger : Current flow in the same direction as proton

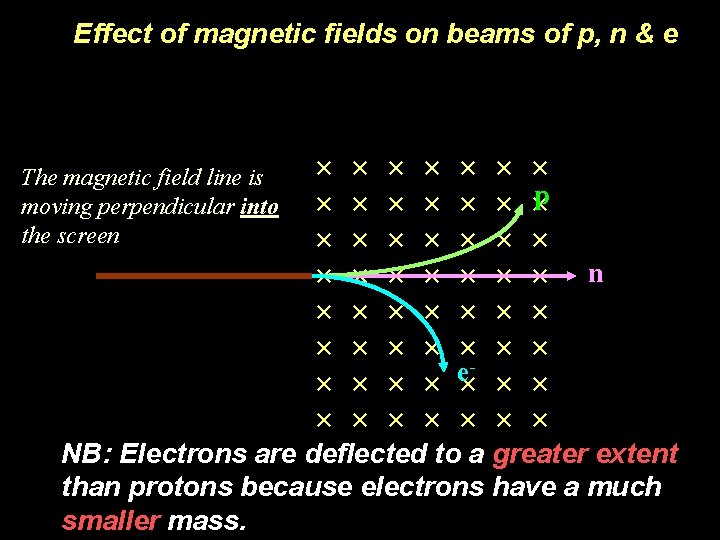
Effect of magnetic fields on beams of p, n & e In a magnetic field: The magnetic field line is moving perpendicular into the screen p n e- NB: Electrons are deflected to a greater extent than protons because electrons have a much smaller mass.

Atomic number & Atomic mass A Z E where A: mass no. (total no. of p & n) Z: atomic no. (no. of p) N: no. of neutrons ( = A - Z ) NB: 1. Since the atomic no. (no. of p) = no. of electrons, therefore, there is no net charge on an atom 2. Loss of electrons give rise to cations (positively charged ions, no. of p > e-) while the gain of electrons give rise to anions (negatively charged ions, no. of p < e-)

Try these : 29 13 32 16 23 11 Al 13 p 16 n 13 e- S 16 p 16 n 18 e- 11 p 12 n 10 e- 2 - + Na

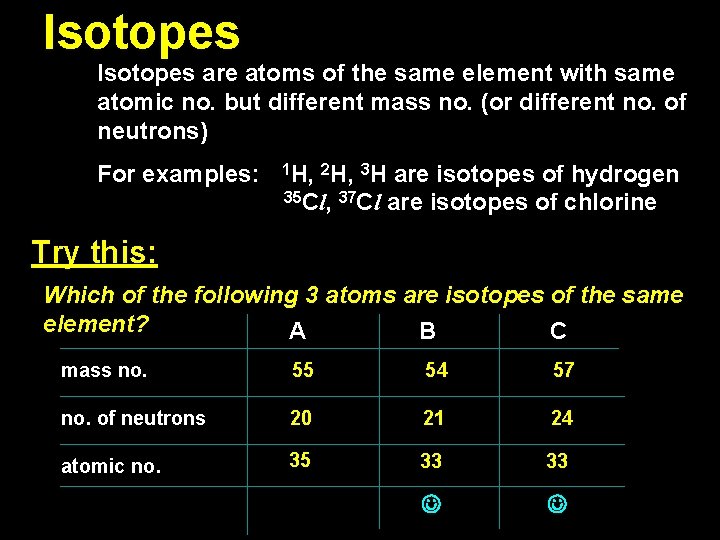
Isotopes are atoms of the same element with same atomic no. but different mass no. (or different no. of neutrons) For examples: 1 H, 2 H, 3 H are isotopes of hydrogen 35 Cl, 37 Cl are isotopes of chlorine Try this: Which of the following 3 atoms are isotopes of the same element? A B C mass no. 55 54 57 no. of neutrons 20 21 24 atomic no. 35 33 33

Isotopes n Isotopes of an element have similar chemical properties This is because they have the same no. of electrons and the chemical properties depend upon the transfer and redistribution of electrons. n Isotopes of an element have different physical properties. This is because they have different no. of neutrons and hence different masses. eg. 37 Cl has a higher density than 35 Cl.

Electronic Energy levels or shells Electrons move around the nucleus of an atom in definite energy levels or shells which are identified by numbers called principal quantum numbers (p. q. n. ), n. 19 K : electrons are arranged in 4 shells ( 2, 8, 8, 1) energy level / shell no. of electrons farthest from Highest energy level 1 ____ n=4 nucleus (most easily removed) 8 ____ n=3 Increasing energy 8 ____ n=2 nearest to nucleus n=1 2 ____ Lowest energy level

Energy sublevels or subshells Energy levels are split into sublevels / subshells. ( labelled s, p, d, f ) Energy level no. of subshells types of subshells labelled as n=1 1 s 1 s n=2 2 s, p 2 s, 2 p n=3 3 s, p, d 3 s, 3 p, 3 d n=4 4 s, p, d, f 4 s, 4 p, 4 d, 4 f

Electrons & Orbitals Electrons are in constant motion. Within each subshells, electrons can be found in certain regions known as orbitals. An orbital is a region or volume of space within which there is a high probability of finding an electron. Each orbital can accommodate two electrons. type of subshell no. of orbitals max. no. of electrons s 1 2 p 3 6 d 5 10 f 7 14

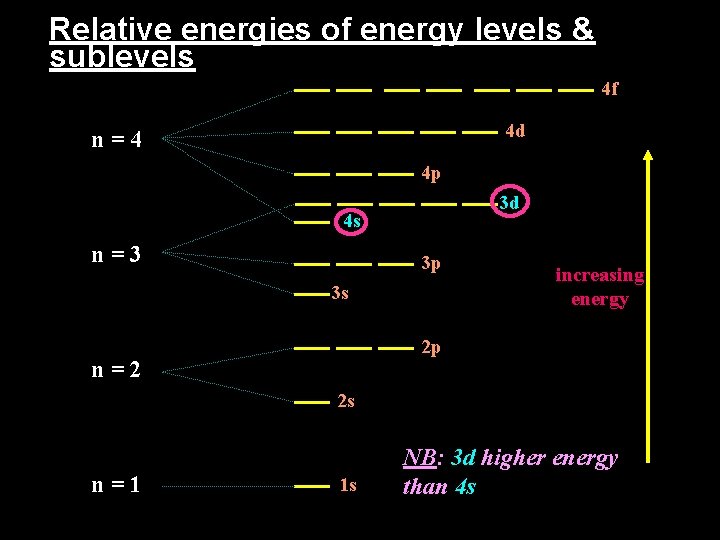
Relative energies of energy levels & sublevels 4 f 4 d n=4 4 p 3 d 4 s n=3 3 p 3 s increasing energy 2 p n=2 2 s n=1 1 s NB: 3 d higher energy than 4 s

Try this: Shell no. Maximum no. of electrons n=1 2 n=2 2+6=8 n=3 2 + 6 + 10 = 18 n=4 2 + 6 + 10 + 14 = 32 General Formula = 2 n 2

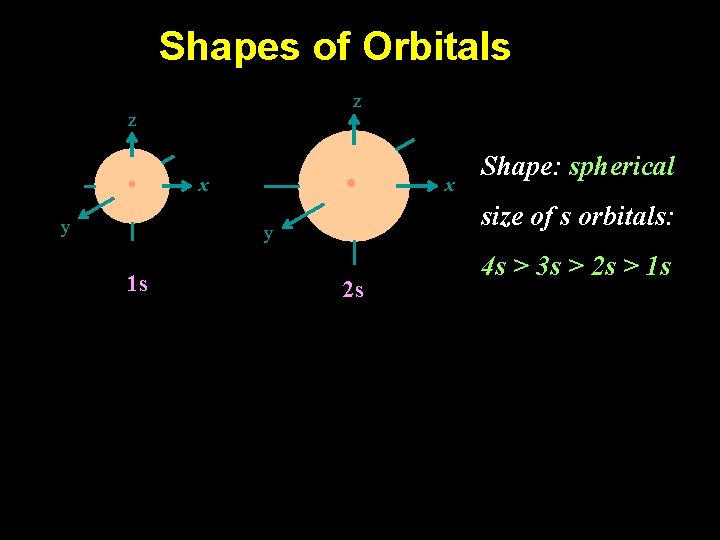
Shapes of Orbitals z z y x size of s orbitals: y 1 s x Shape: spherical 2 s 4 s > 3 s > 2 s > 1 s

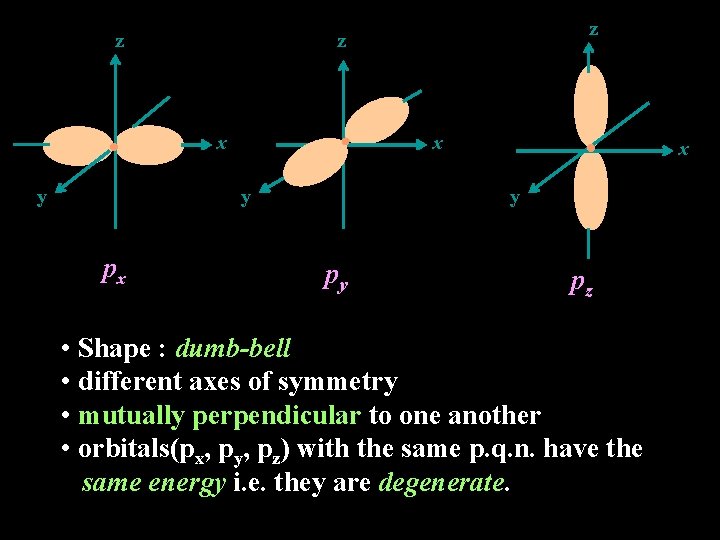
z x y y px z z x y py pz • Shape : dumb-bell • different axes of symmetry • mutually perpendicular to one another • orbitals(px, py, pz) with the same p. q. n. have the same energy i. e. they are degenerate. x

Electronic Configuration Electrons are arranged in orbitals according to a set of rules. 1. Pauli Exclusion Principle: An atomic orbital can be occupied by only two electrons and their spins are always opposite.

Electronic Configuration 2. Aufbau (or building up) Principle: Electrons fill the orbitals of lowest energy first and then a higher energy. Increasing energy 6 s . . . 5 s 5 p . . . 4 s 4 p 4 d 3 s 3 p 3 d 2 s 2 p 1 s 4 f

Electronic Configuration 3. Hund’s Rule: When electrons are added successively to a set of orbital of the same energy level, they occupy them singly first before any pairing occurs. 2 p 2 p

Ionisation Energies Information about the arrangement of electrons has been obtained by bombarding the atoms with streams of fastmoving electrons and so removing electrons from the atom. This method allows us to measure approximately the energy required to remove an electron, called the ionisation energy. First ionisation energy of an element is the amount of energy required to remove one mole of electrons from one mole of gaseous atoms. M(g) M+(g) + e H = + value k. J mol -1

Examples: Na(g) Na+(g) + e H = + 494 k. J mol -1 Mg(g) Mg+(g) + e H = + 736 k. J mol -1 Second ionisation energy of an element is the amount of energy required to remove one mole of electrons from one mole of gaseous ions. M +(g) M 2+(g) + e H = + value k. J mol -1

Example: Na +(g) Na 2+(g) + e H = + 4560 k. J mol -1 Mg +(g) Mg 2+(g) + e H = + 1450 k. J mol -1 NB: 1 st IE < 2 nd IE < 3 rd IE <. . . . .

Factors affecting the first IE of elements Atomic radius (distance of outermost electron from the nucleus) As atomic radius increases, outer electrons experience less nuclear attraction, hence 1 st IE decreases. Size of nuclear charge (no. of p in nucleus) As size of nuclear charge increases, outer electrons experience greater nuclear attraction, hence 1 st IE increases.

Outer electrons are shielded Screening or from nuclear attraction by shielding effect repelling effect of inner-shells by inner-shells electrons. As no. of inner shells increases, the shielding effect electrons by inner-shells electron increases, and hence 1 st IE decreases. NB: Electrons in the same shell exert a negligible shielding effect on each other.

Try this: Compare the 1 st IE of 5 B & 6 C. 2 2 1 2 2 2 5 B : 1 s 2 s 2 p 6 C : 1 s 2 s 2 p C has a greater nuclear charge than B (because C has a greater no. of proton) atomic radius of C is smaller than that of B. outer electron of C would experience greater nuclear attraction. Both B & C have the same shielding effect by innershell electrons (because they have the same no. of inner principal quantum shell) As a result, 1 st IE of C > 1 st IE of B.

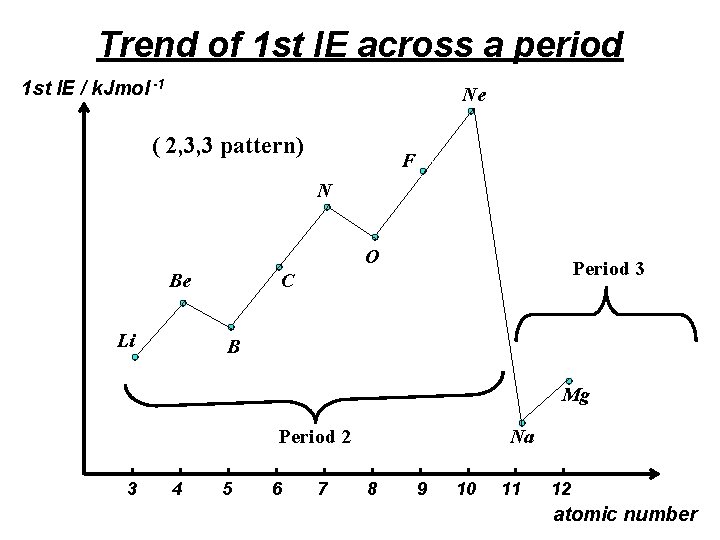
Trend of 1 st IE across a period 1 st IE / k. Jmol -1 Ne ( 2, 3, 3 pattern) F N O Be Li Period 3 C B Mg Period 2 3 4 5 6 7 Na 8 9 10 11 12 atomic number

Across any period, there is a general increase in the 1 st IE due to the increase in the nuclear charge & the decrease in atomic radius across the period. (the shielding effect remains almost the same from one element to the other because electrons are added successively to the same shell)

However, • the 1 st IE of B is lower than that of Be. 2 2 s 2 2 p 1 B : 1 s 5 2 2 s 2 Be : 1 s 4 2 p electron of B is of a higher energy than the 2 s electron of Be. Hence, a lower amount of energy is required to remove the 2 p electron from B than to remove the 2 s electron from Be.

• the 1 st IE of O is lower than that of N. 2 2 s 2 2 p 4 O : 1 s 8 2 2 s 2 2 p 3 N : 1 s 7 The mutual repulsion of the paired electrons in a 2 p orbital of O makes the removal of one electron from that orbital easier compared to a 2 p electron of N which does not experience such repulsion.

Down a group, the 1 st IE decreases. This is due to the increase in atomic radius the increase in shielding effect by inner-shell electrons. These two factors outweigh the effect of the increasing nuclear charge.

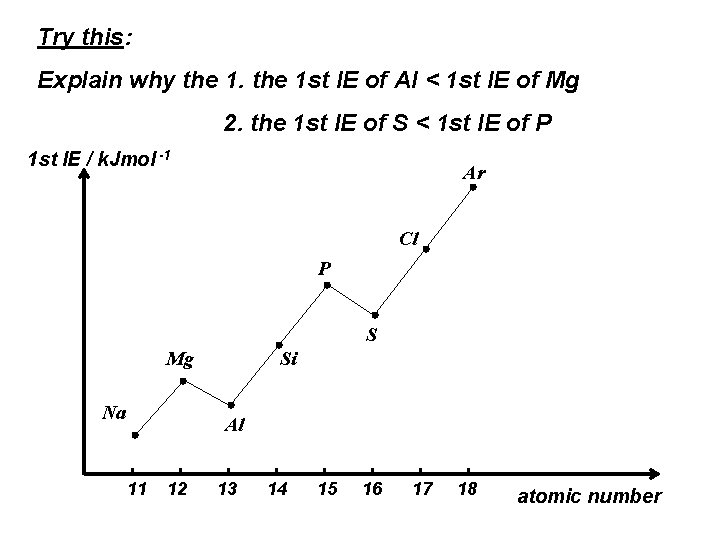
Try this: Explain why the 1 st IE of Al < 1 st IE of Mg 2. the 1 st IE of S < 1 st IE of P 1 st IE / k. Jmol -1 Ar Cl P S Mg Na Si Al 11 12 13 14 15 16 17 18 atomic number

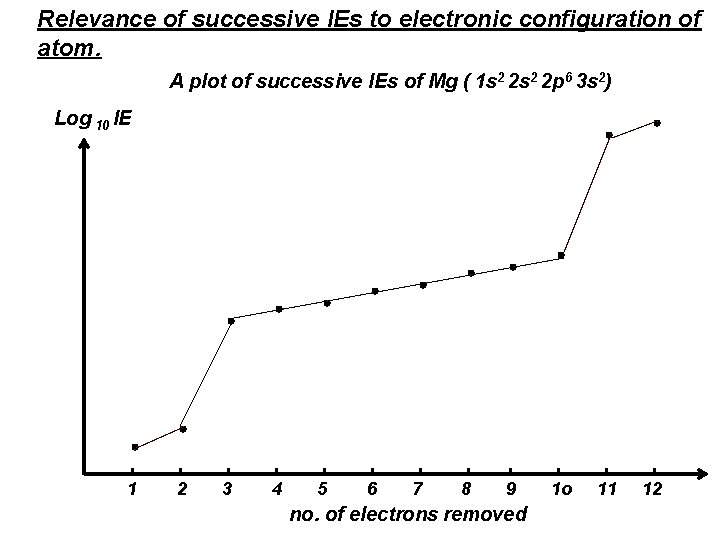
Relevance of successive IEs to electronic configuration of atom. A plot of successive IEs of Mg ( 1 s 2 2 p 6 3 s 2) Log 10 IE 1 2 3 4 5 6 7 8 9 no. of electrons removed 1 o 11 12

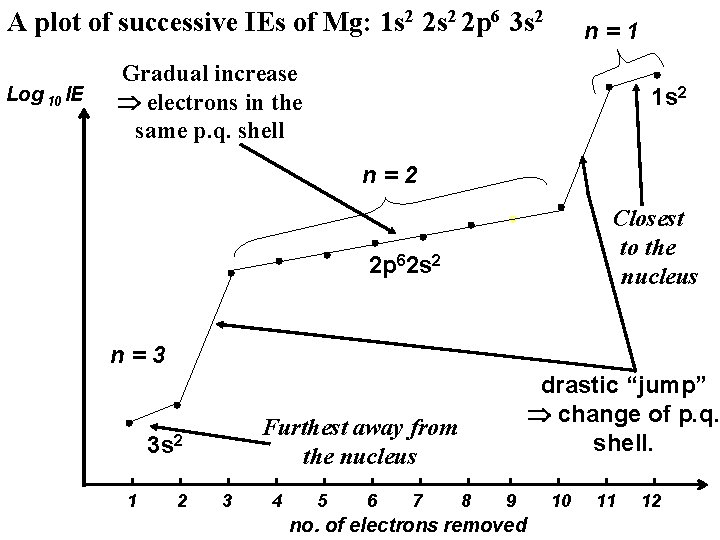
A plot of successive IEs of Mg: 1 s 2 2 p 6 3 s 2 Log 10 IE n=1 Gradual increase electrons in the same p. q. shell 1 s 2 n=2 Closest to the nucleus 2 p 62 s 2 n=3 Furthest away from the nucleus 3 s 2 1 2 drastic “jump” change of p. q. shell. 3 4 5 6 7 8 9 no. of electrons removed 10 11 12

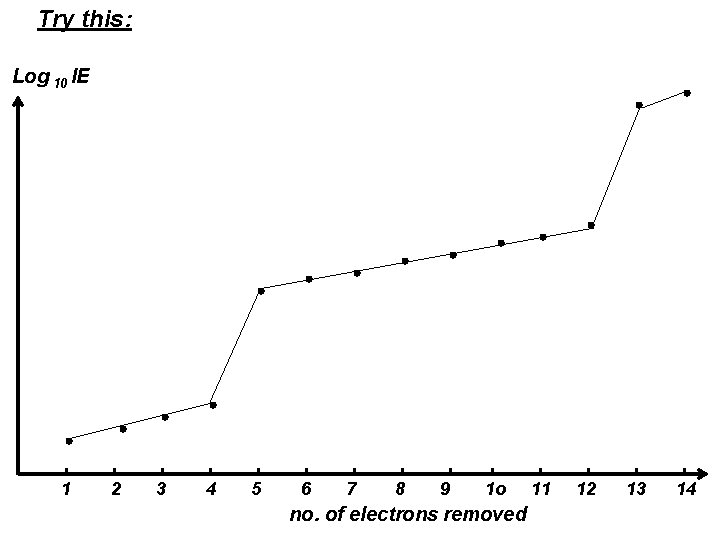
Try this: Log 10 IE 1 2 3 4 5 6 7 8 9 1 o no. of electrons removed 11 12 13 14

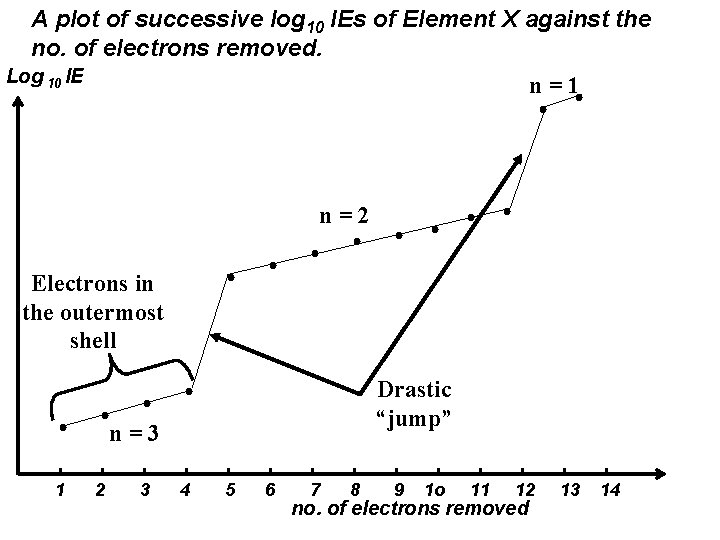
A plot of successive log 10 IEs of Element X against the no. of electrons removed. Log 10 IE n=1 n=2 Electrons in the outermost shell Drastic “jump” n=3 1 2 3 4 5 6 7 8 9 1 o 11 12 no. of electrons removed 13 14

Element X belongs to Group IV because it has 4 valence electrons. It has 3 principal quantum shells and its electronic configuration is 1 s 2 2 p 6 3 s 2 3 p 2 n=1 n=2 n=3 Valence electrons

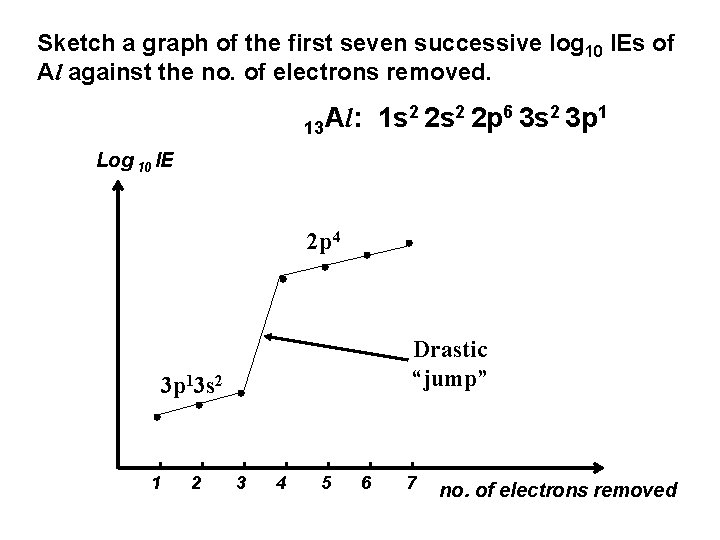
Sketch a graph of the first seven successive log 10 IEs of Al against the no. of electrons removed. 2 2 s 2 2 p 6 3 s 2 3 p 1 Al: 1 s 13 Log 10 IE 2 p 4 Drastic “jump” 3 p 13 s 2 1 2 3 4 5 6 7 no. of electrons removed

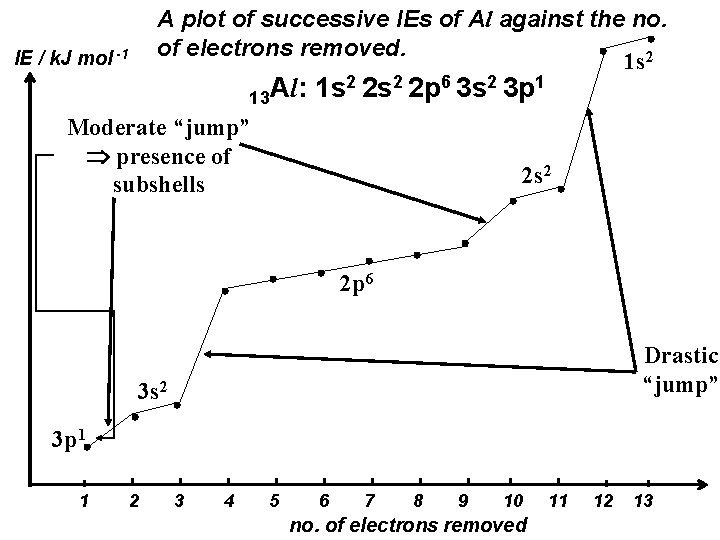
A plot of successive IEs of Al against the no. of electrons removed. 1 s 2 IE / k. J mol -1 13 Al: 1 s 2 2 p 6 3 s 2 3 p 1 Moderate “jump” presence of subshells 2 s 2 2 p 6 Drastic “jump” 3 s 2 3 p 1 1 2 3 4 5 6 7 8 9 10 no. of electrons removed 11 12 13

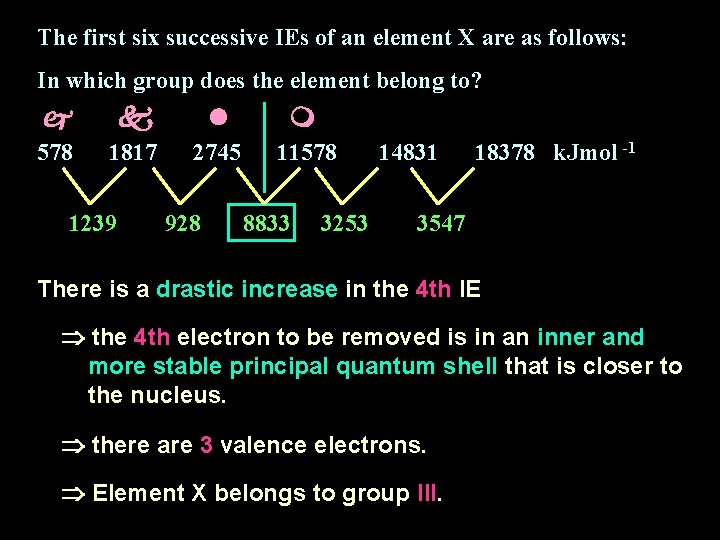
The first six successive IEs of an element X are as follows: In which group does the element belong to? 578 1817 2745 11578 1239 928 8833 3253 14831 18378 k. Jmol -1 3547 There is a drastic increase in the 4 th IE the 4 th electron to be removed is in an inner and more stable principal quantum shell that is closer to the nucleus. there are 3 valence electrons. Element X belongs to group III.

The successive IEs of an element Y are as follows: In which group does element Y belong? 740 760 1500 7700 6200 10500 2800 13600 3100 4400 18000 21700 3700 Ans: Element Y belongs to Group II.

N 92 P 1 Q 1 (a) Write down the electronic configuration of the selenium atom. (b) How would you expect the first ionisation energy of selenium to compare with that of (i) sulphur (ii) bromine Give your reasoning.

(a) 2 2 s 2 2 p 6 3 s 2 3 p 6 3 d 10 4 s 2 4 p 4 Gp VI Se: 1 s 34 (b) 2 2 s 2 2 p 6 3 s 2 3 p 4 Gp VI S: 1 s 16 The 4 p electron of Se to be removed is i) more shielded by inner-shells’ electrons and ii) further away from the nucleus compared with the 3 p electron to be removed from S. Therefore, the 1 st IE of Se is expected to be lower than that of S.

(a) 2 2 s 2 2 p 6 3 s 2 3 p 6 3 d 10 4 s 2 4 p 4 Gp VI Se: 1 s 34 (b) 2 2 s 2 2 p 6 3 s 2 3 p 6 3 d 10 4 s 2 4 p 5 Gp VII Br: 1 s 35 same period Se has a i) smaller nuclear charge and ii) larger atomic radius than Br Therefore, the 1 st IE of Se is expected to be lower than that of Br.