ATOMIC STRUCTURE Chemistry 11 Atomic Theory 2 SUBATOMIC

ATOMIC STRUCTURE Chemistry 11 – Atomic Theory 2

SUBATOMIC PARTICLES: Particles which make up the atom are called subatomic particles. Electron: negative charge, almost no mass Proton: positive charge, has a mass of 1 atomic mass unit (amu) Neutron: no charge, has a mass of 1 atomic mass unit (amu). Responsible for the stability of the atom

ATOMIC NUMBER: Atomic Number is the number of protons in the nucleus. Each element has a unique atomic number.
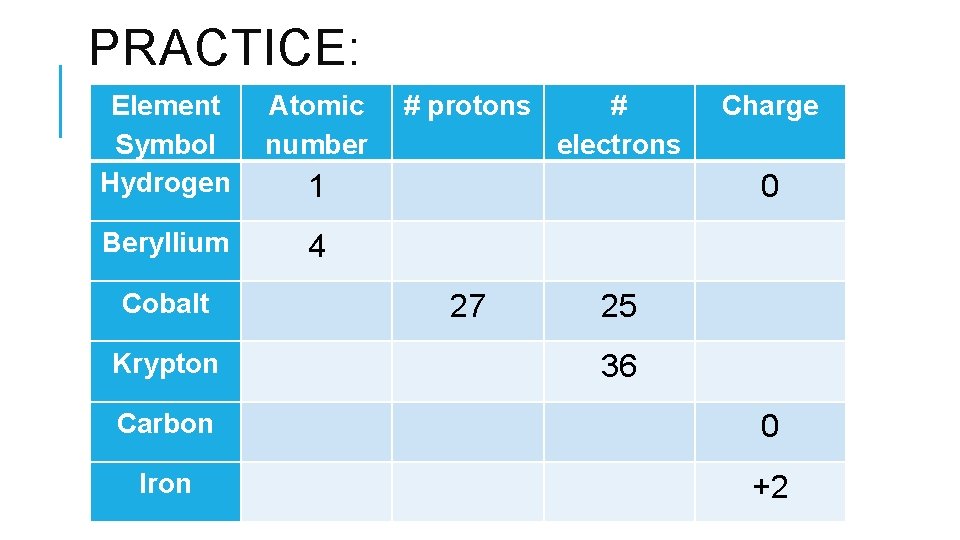
PRACTICE: Element Symbol Hydrogen Atomic number # protons # electrons Charge 1 0 Beryllium 4 Cobalt 27 25 Krypton 36 Carbon 0 Iron +2
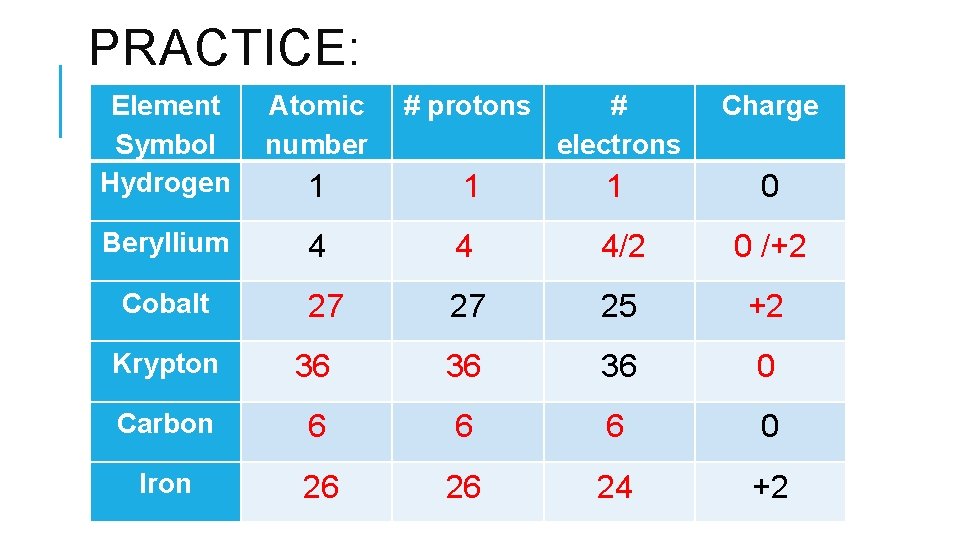
PRACTICE: Element Symbol Hydrogen Atomic number # protons # electrons Charge 1 1 1 0 Beryllium 4 4 4/2 0 /+2 Cobalt 27 27 25 +2 Krypton 36 36 0 Carbon 6 6 6 0 Iron 26 26 24 +2

ISOTOPES: Mass # Carbon-12 Carbon-13 Carbon-14
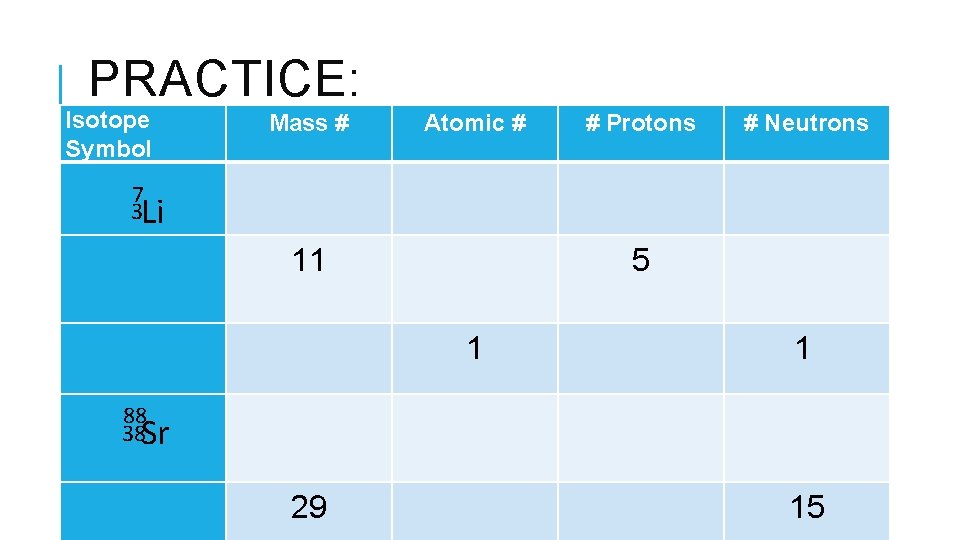
PRACTICE: Isotope Symbol Mass # Atomic # # Protons # Neutrons 11 5 1 1 88 38 Sr 29 7 3 Li 15
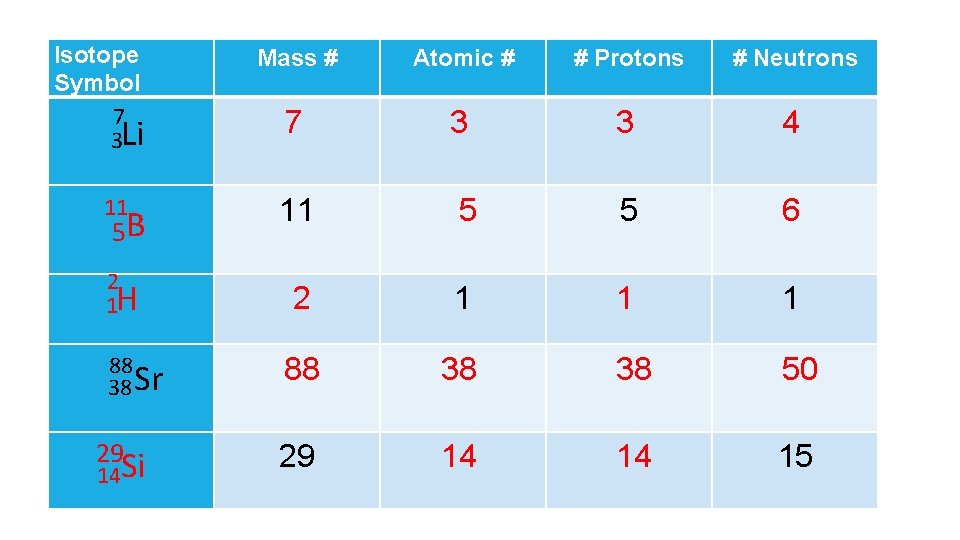
Isotope Symbol 7 3 Li Mass # Atomic # # Protons # Neutrons 7 3 3 11 5 5 2 1 H 2 1 1 4 6 1 88 38 50 29 14 15 5 B 88 38 Sr 29 14 Si

ISOTOPES: The atomic mass listed on the periodic table is a weighted average of all existing isotopes. If we are given the % composition of each isotope, we can figure out the atomic mass: Example If a sample is found to be made up of 90. 48% Ne-20, 0. 27% Ne-21 and 9. 25% Ne-22, then what is the atomic mass of Neon in the sample? (90. 48 x 20) + (0. 27 x 21) + (9. 25 x 22) ÷ 100 = 20. 19

PRACTICE: 1. Assume you have 100 mol of the sample, then find the # moles of each isotope. 2. Multiply each isotope by its molar mass. 3. Add up the mass values, divide by 100 moles. Find the atomic mass of a sample that is made up of 78. 99% Mg-24, 10. 00% Mg-25, and 11. 01% Mg-26. 24. 32

ASSIGNMENT Page 147 #19, 22, 23 a-e, 24
- Slides: 11