Atomic Structure Basic Structure n4 n3 n2 n1

Atomic Structure
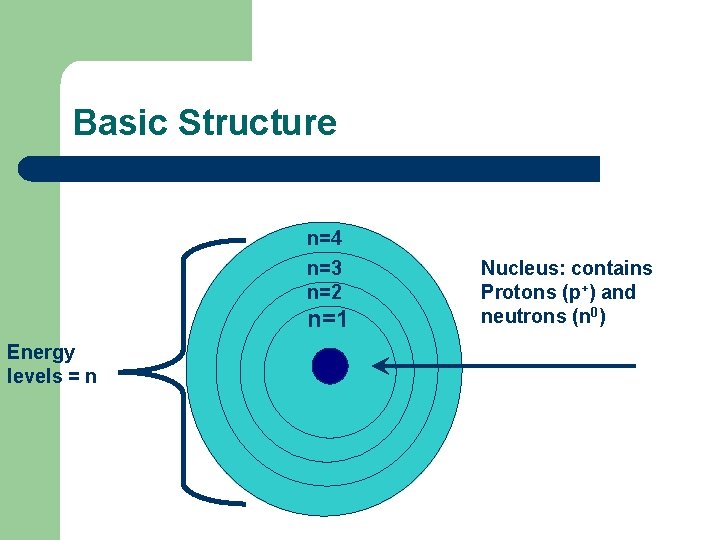
Basic Structure n=4 n=3 n=2 n=1 Energy levels = n Nucleus: contains Protons (p+) and neutrons (n 0)
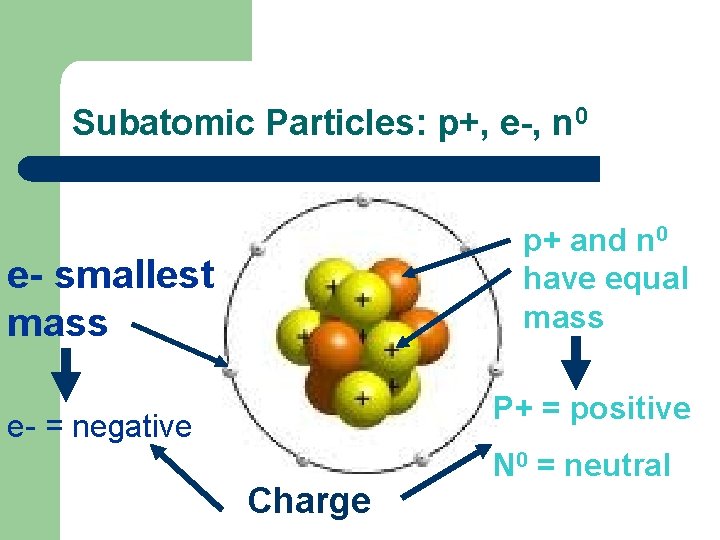
Subatomic Particles: p+, e-, n 0 p+ and n 0 have equal mass e- smallest mass P+ = positive e- = negative Charge N 0 = neutral

Calculating the number of electrons in an energy level. 2 n 2 n=1 2(1)2 = 2 e- n=2 2(2)2 = 8 e- n=3 2(3)2 = 18 e- n=4 2(4)2 = 32 e-
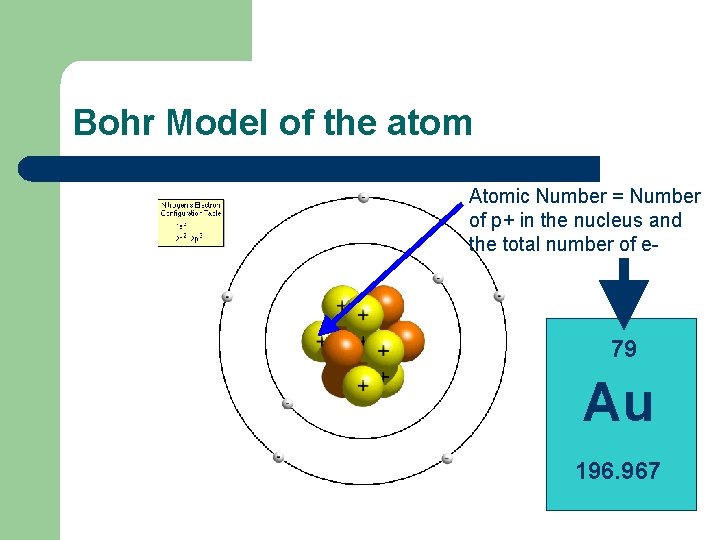
Bohr Model of the atom Atomic Number = Number of p+ in the nucleus and the total number of e- 79 Au 196. 967
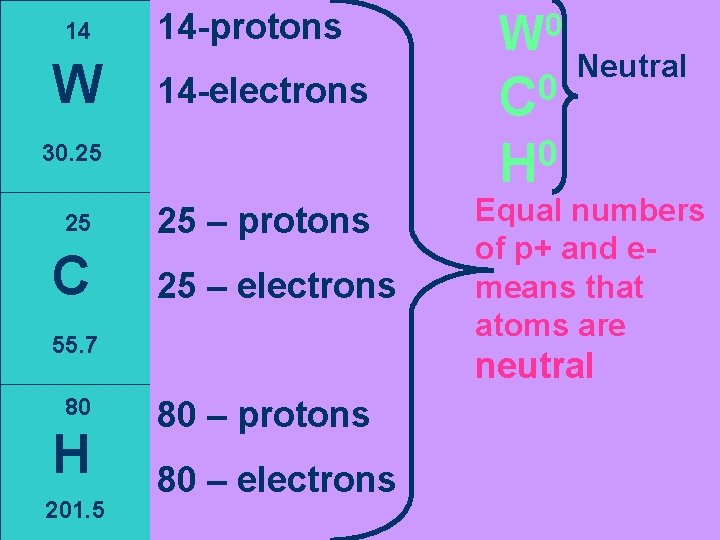
14 W 14 -protons 14 -electrons C 25 – protons 25 – electrons 55. 7 80 H 201. 5 0 C Neutral 0 H 30. 25 25 0 W Equal numbers of p+ and emeans that atoms are neutral 80 – protons 80 – electrons
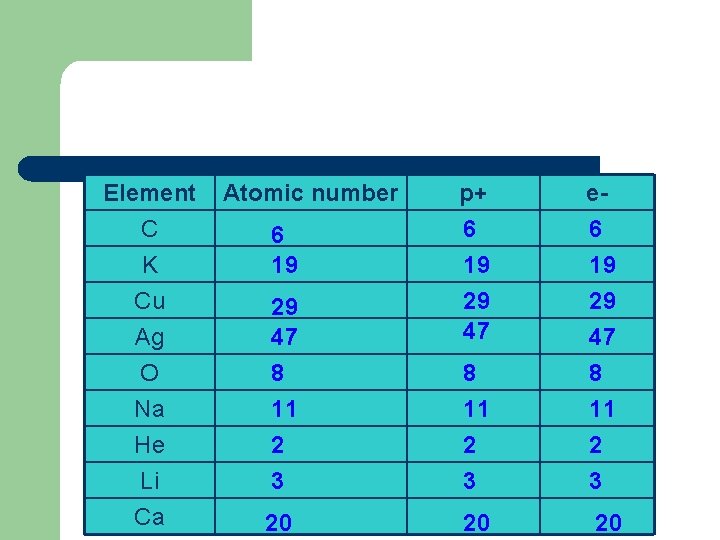
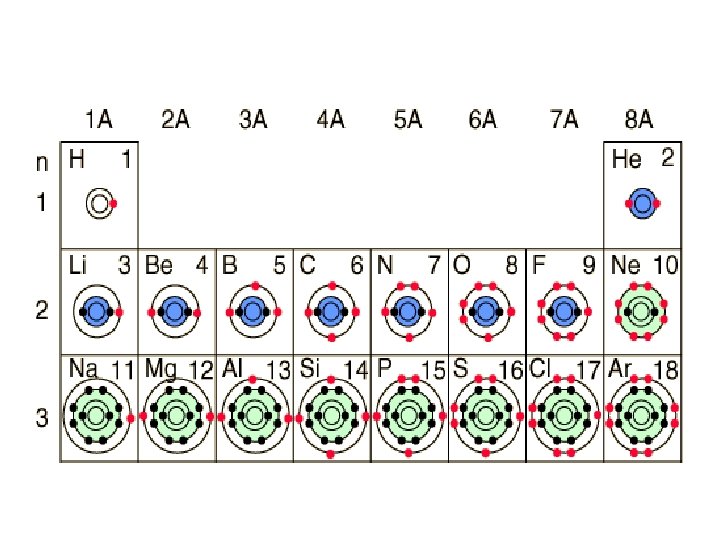
Element C K Cu Ag O Na He Li Ca Atomic number 6 19 29 47 8 11 2 3 20 p+ e- 6 19 29 47 8 11 2 3 20 20

Relative mass or Mass number 79 Au 196 79 Au 197 Mass number = 196 amu How many p+ How many e. How many n 0 79 79 117 Mass # = p+ + n 0 196 = 79 + n 0 n= 117 Mass number = 197 amu How many p+ 79 How many e- 79 118 How many n 0 Mass # = p+ + n 0 197 = 79 + n 0 n= 118
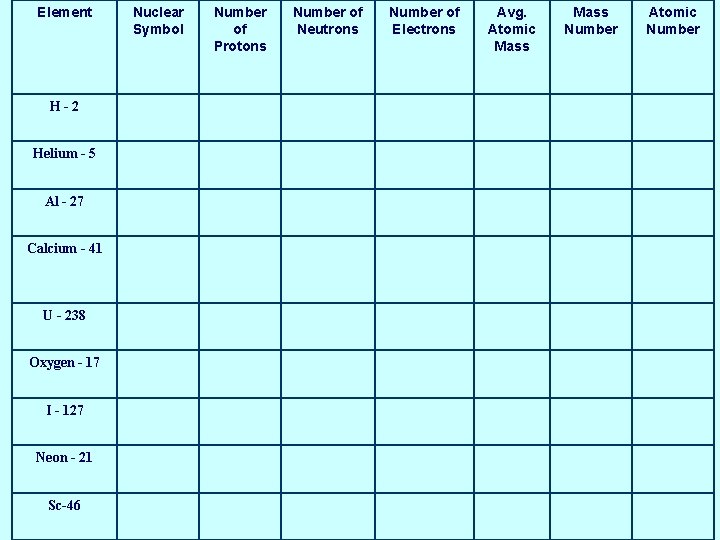
Element H-2 Helium - 5 Al - 27 Calcium - 41 U - 238 Oxygen - 17 I - 127 Neon - 21 Sc-46 Nuclear Symbol Number of Protons Number of Neutrons Number of Electrons Avg. Atomic Mass Number Atomic Number
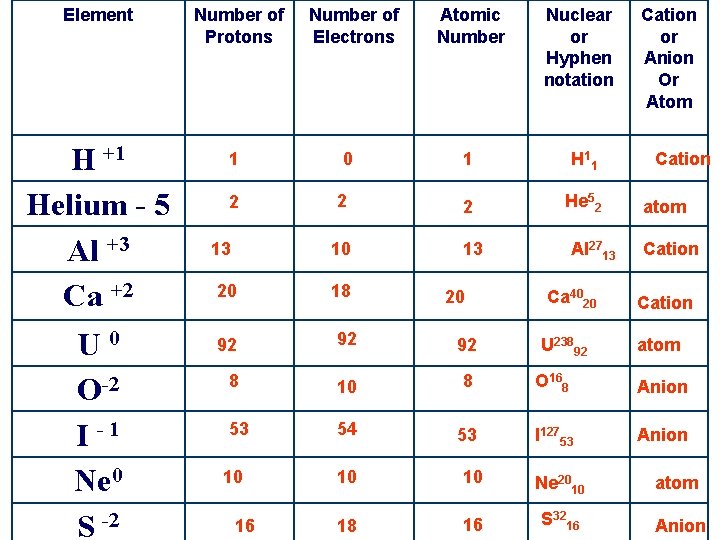
Element Number of Protons H +1 Helium - 5 Al +3 Ca +2 1 U 0 O-2 I -1 Ne 0 S -2 2 13 20 Number of Electrons Atomic Number Nuclear or Hyphen notation 1 H 1 1 2 2 He 52 10 13 0 18 20 Al 2713 Ca 4020 Cation or Anion Or Atom Cation atom Cation 92 92 92 8 10 8 O 168 Anion 53 54 53 I 12753 Anion 10 16 U 23892 atom 10 10 Ne 2010 atom 18 16 S 3216 Anion
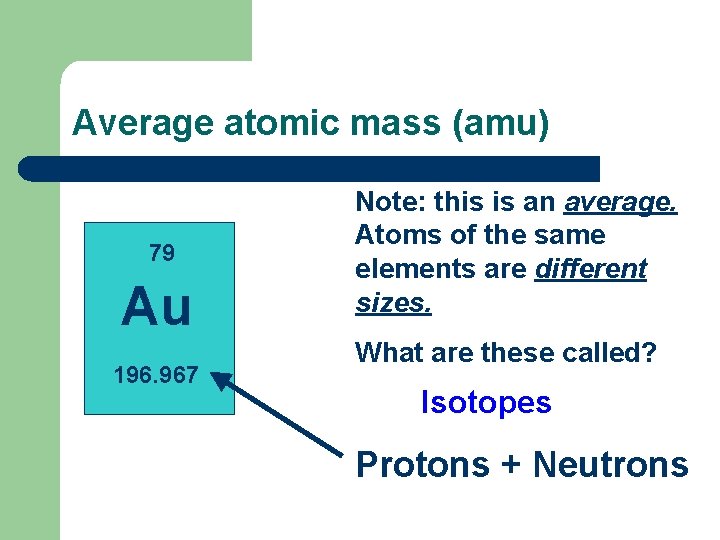
Average atomic mass (amu) 79 Au 196. 967 Note: this is an average. Atoms of the same elements are different sizes. What are these called? Isotopes Protons + Neutrons

Comparison of Isotopes Atomic mass is the average of all the naturally isotopes of that element. Carbon = 12. 011 Isotope Symbol Composition of % in nature the nucleus 12 C Carbon-12 6 protons 98. 89% 6 neutrons Carbon-13 13 C 6 protons 7 neutrons 1. 11% Carbon-14 14 C 6 protons 8 neutrons <0. 01%
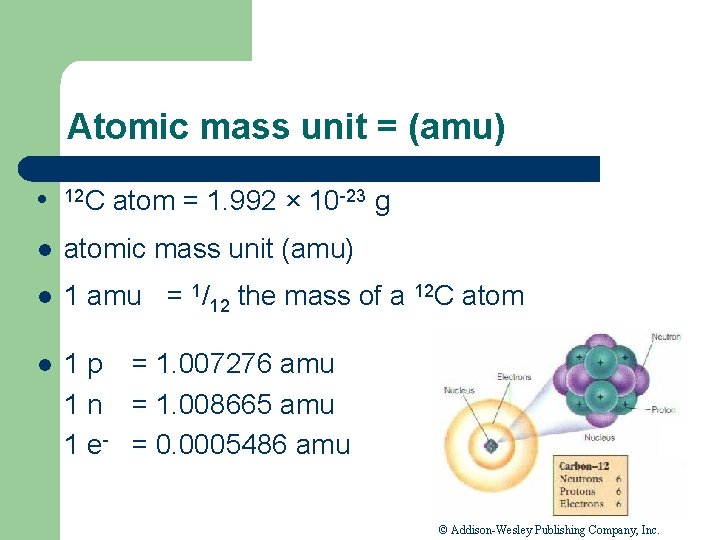
Atomic mass unit = (amu) l 12 C atom = 1. 992 × 10 -23 g l atomic mass unit (amu) l 1 amu = 1/12 the mass of a 12 C atom l 1 p = 1. 007276 amu 1 n = 1. 008665 amu 1 e- = 0. 0005486 amu © Addison-Wesley Publishing Company, Inc.

Average Atomic Mass l weighted average of all isotopes on the Periodic Table Avg. Atomic Mass

D. Average Atomic Mass l EX: Calculate the avg. atomic mass of oxygen if its abundance in nature is 99. 76% 16 O, 0. 04% 17 O, and 0. 20% 18 O. Avg. Atomic Mass 16. 00 amu

D. Average Atomic Mass l EX: Calculate the avg. atomic mass of oxygen if its abundance in nature is 55. 32% 25 X, 0. 94% 23 X, and 43. 74% 24 X. Avg. Atomic Mass 24. 54 amu

D. Average Atomic Mass l EX: Calculate the avg. atomic mass of oxygen if its abundance in nature is 99. 97% 1 H, 0. 02% 2 H, and 0. 01% 3 H. Avg. Atomic Mass 16. 00 amu

Average Atomic Mass l EX: Calculate the avg. atomic mass of oxygen if its abundance in nature is 50% 197 Au, 50% 198 Au. Avg. Atomic Mass (197)(50)+ (198)(50) = = 197. 5 amu 100
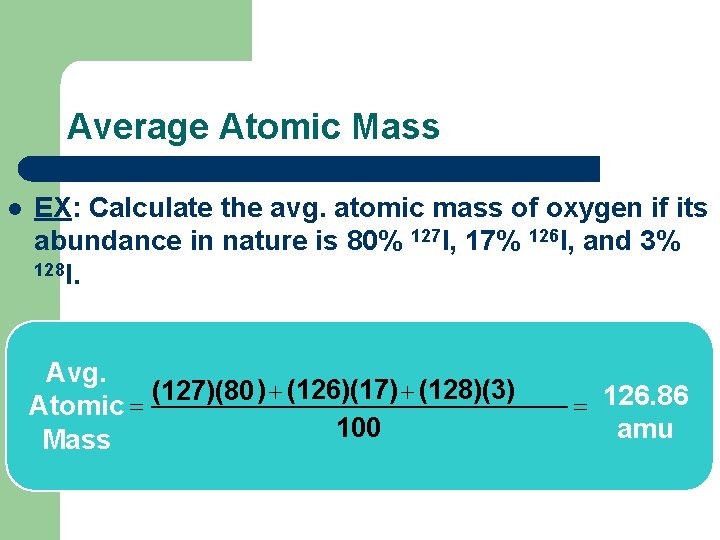
Average Atomic Mass l EX: Calculate the avg. atomic mass of oxygen if its abundance in nature is 80% 127 I, 17% 126 I, and 3% 128 I. Avg. (127)(80 ) + (126)(17) + (128)(3) Atomic = 100 Mass = 126. 86 amu

Average Atomic Mass l EX: Find chlorine’s average atomic mass if approximately 8 of every 10 atoms are chlorine-35 and 2 are chlorine-37. Avg. Atomic Mass 35. 40 amu

Average Atomic Mass l EX: Find element Y’s average atomic mass if approximately 5 of every 10 atoms are Y- 64 and 2 are Y- 61 and 3 are Y - 60 Avg. Atomic Mass 62. 2 amu
A 1000 B Jb U 17 N D A 1000 N Jb Jb Jb 10 20 30 50% 30% C E 20%
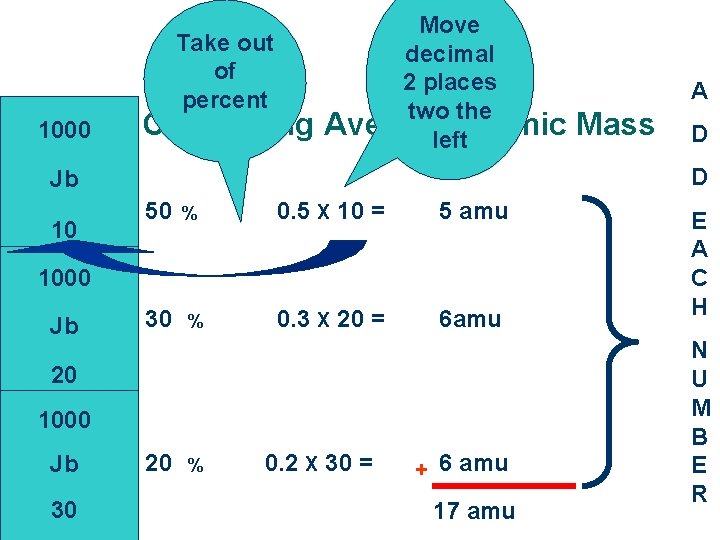
Take out of percent 1000 Calculating Move decimal 2 places two the Average Atomic left 50 % 0. 5 X 10 = 5 amu 0. 3 X 20 = 6 amu 1000 Jb 30 % 20 1000 Jb 30 Mass D D Jb 10 A 20 % 0. 2 X 30 = + 6 amu 17 amu E A C H N U M B E R
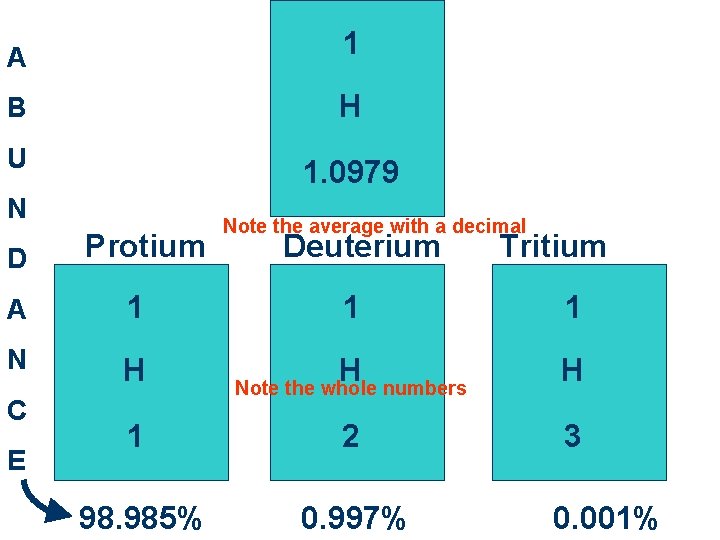
A 1 B H U 1. 0979 N D Protium A 1 N H C E Note the average with a decimal Deuterium Tritium 1 1 H H 1 2 3 98. 985% 0. 997% Note the whole numbers 0. 001%
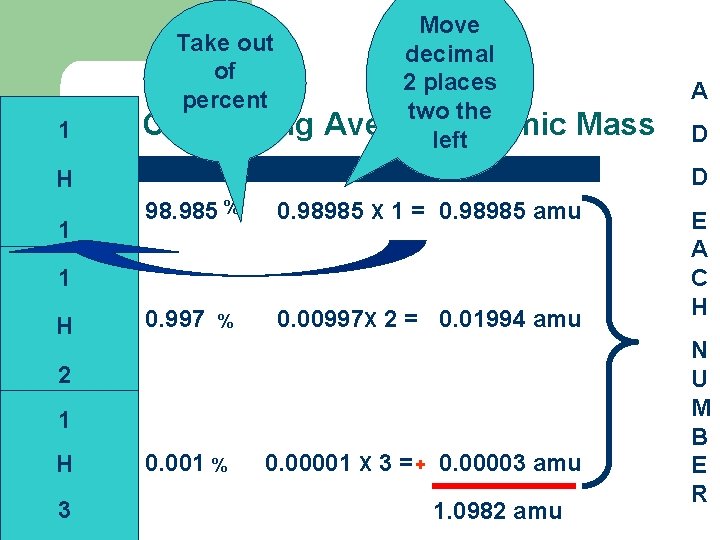
Take out of percent 1 Calculating Move decimal 2 places two the Average Atomic left A Mass D H 1 98. 985 % 0. 98985 X 1 = 0. 98985 amu 0. 997 0. 00997 X 2 = 0. 01994 amu 1 H % 2 1 H 3 D 0. 001 % 0. 00001 X 3 = + 0. 00003 amu 1. 0982 amu E A C H N U M B E R
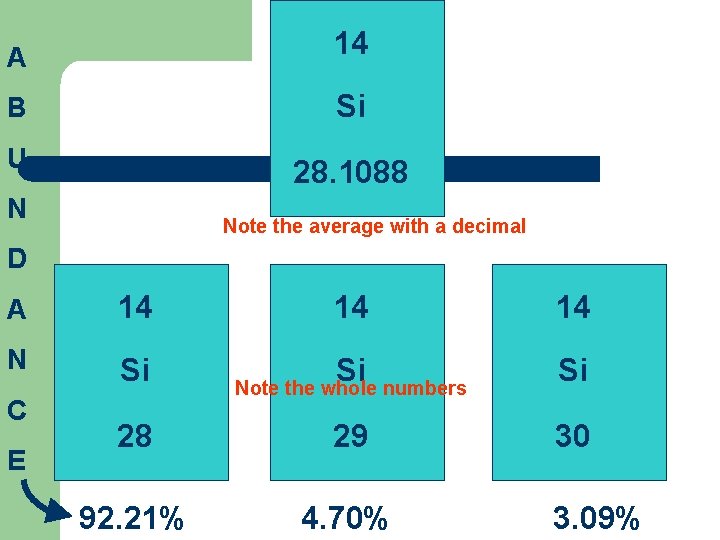
A 14 B Si U 28. 1088 N Note the average with a decimal D A 14 N Si C E 14 14 Si Si 28 29 30 92. 21% 4. 70% Note the whole numbers 3. 09%
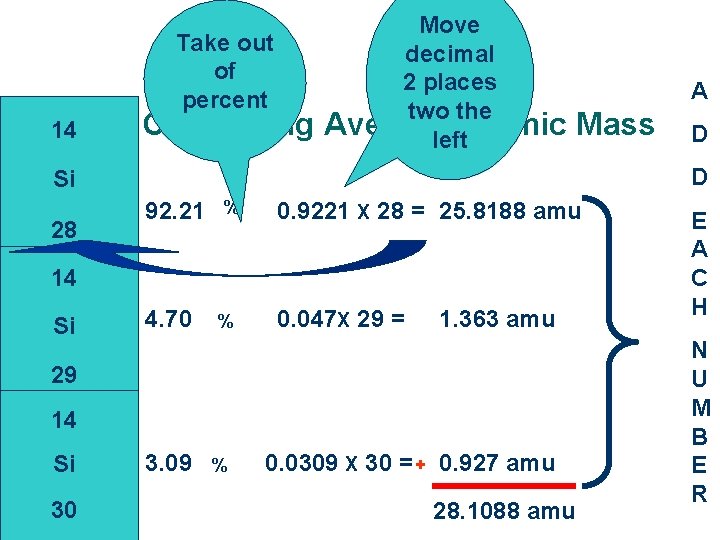
Take out of percent 14 Calculating Move decimal 2 places two the Average Atomic left A Mass D Si 28 92. 21 % 0. 9221 X 28 = 25. 8188 amu 14 Si 4. 70 % 0. 047 X 29 = 1. 363 amu 29 14 Si 30 D 3. 09 % 0. 0309 X 30 = + 0. 927 amu 28. 1088 amu E A C H N U M B E R
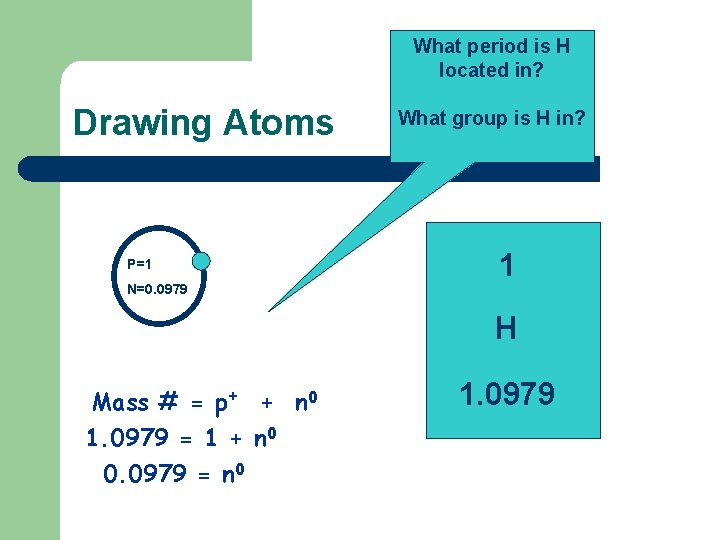
What period is H located in? Drawing Atoms P=1 N=0. 0979 What group is H in? 1 H Mass # = p+ + n 0 1. 0979 = 1 + n 0 0. 0979 = n 0 1. 0979
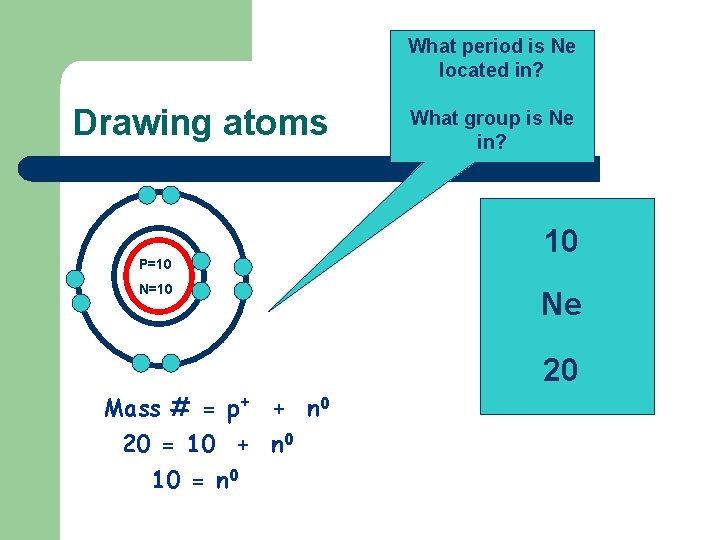
What period is Ne located in? Drawing atoms P=10 N=10 Mass # = p+ + n 0 20 = 10 + n 0 10 = n 0 What group is Ne in? 10 Ne 20
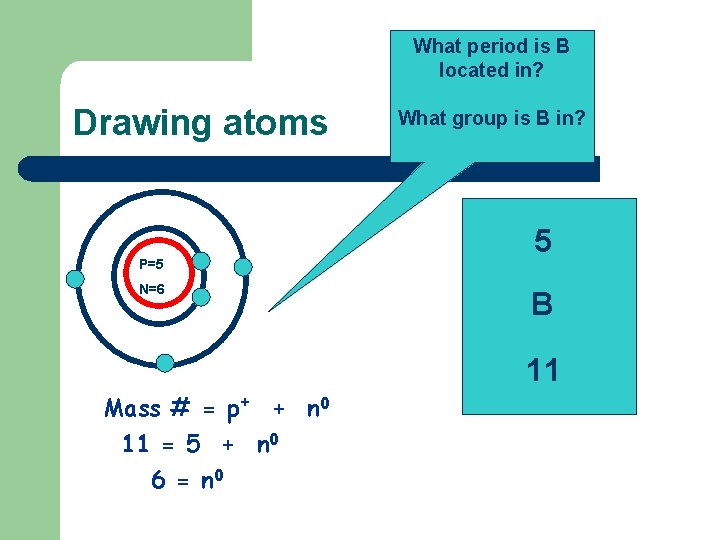
What period is B located in? Drawing atoms P=5 N=6 Mass # = p+ + n 0 11 = 5 + n 0 6 = n 0 What group is B in? 5 B 11
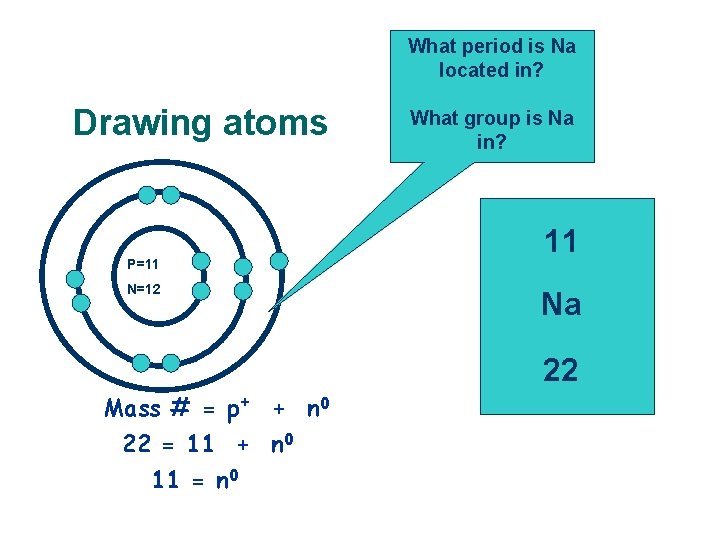
What period is Na located in? Drawing atoms P=11 N=12 Mass # = p+ + n 0 22 = 11 + n 0 11 = n 0 What group is Na in? 11 Na 22
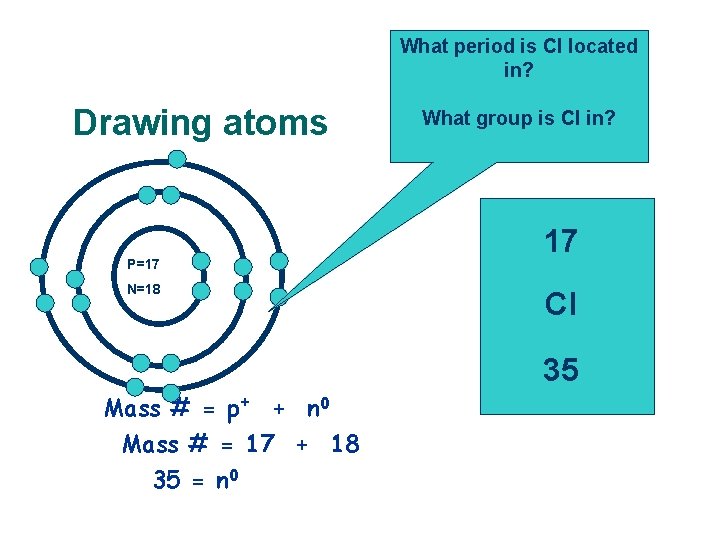
What period is Cl located in? Drawing atoms P=17 N=18 Mass # = p+ + n 0 Mass # = 17 + 18 35 = n 0 What group is Cl in? 17 Cl 35

Draw the Bohr structure of atoms 1 -20 odd Label the protons, neutrons and electrons What group is it in? What period is it in?
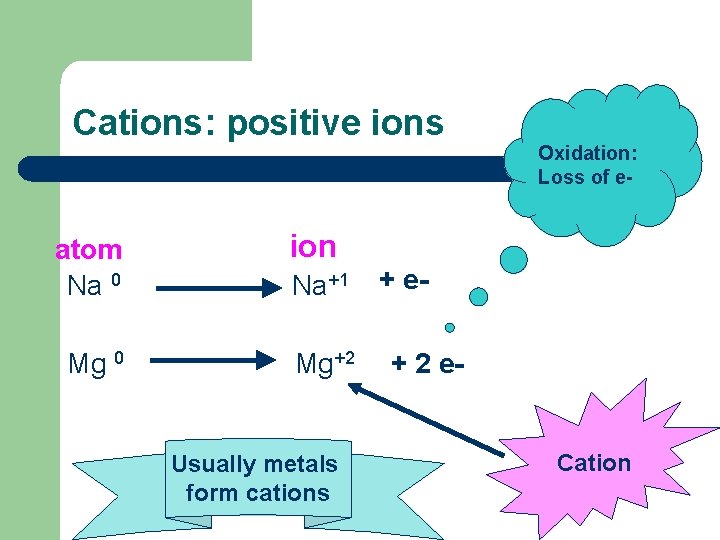
Cations: positive ions atom Na 0 Mg 0 Oxidation: Loss of e- ion Na+1 Mg+2 Usually metals form cations + e+ 2 e- Cation
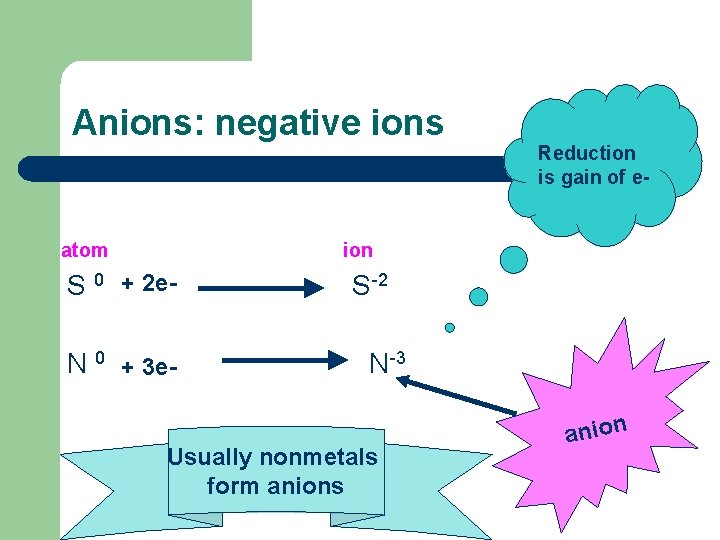
Anions: negative ions atom Reduction is gain of e- ion S 0 + 2 e. N 0 + 3 e- S-2 N-3 Usually nonmetals form anions anion
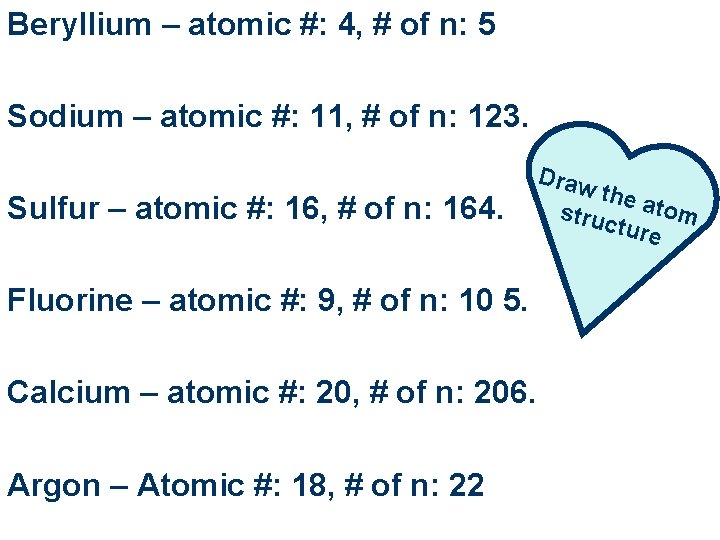
Beryllium – atomic #: 4, # of n: 5 Sodium – atomic #: 11, # of n: 123. Sulfur – atomic #: 16, # of n: 164. Fluorine – atomic #: 9, # of n: 10 5. Calcium – atomic #: 20, # of n: 206. Argon – Atomic #: 18, # of n: 22 Draw the a tom struc ture

Draw the Bohr structure of atoms 1 -20 odd Label the protons, neutrons and electrons What group is it in? What period is it in?

The Octet Rule Click here l l l The octet rule states that atoms are most stable when they have a full shell of 8 electrons in the outside electron shell. Octet = 8 An atom with eight electrons in the outer shell is more stable than an atom which has fewer electrons in the outer shell. The exception to this is Helium (atomic number 2) which only has two electrons in its outer shell. It has a full shell, so it is a stable inert element.

Group number and oxidation states What do I do with this electron to become and ion? e
What to do? How will I ever get to 8 e- in my outter energy What are level. 8 e- By using the my group choices? number gain or on the. Lose, periodic share. table I have octet if IWill am noanlonger I lose an e-? Yes Because the I neutral because 2 nd energy level lost an e-? ? ? Yes!!!! 2 e- Li How already has 8 e- Does this happen with all metals like me? What charge am I now?
I’d say Spastic Atom I am happy atom

Group number and oxidation states Octet Rule or Rule of 8

Predicting Ionic Charges Group 1: Lose 1 electron to form 1+ ions H+ Li+ Na+ K+
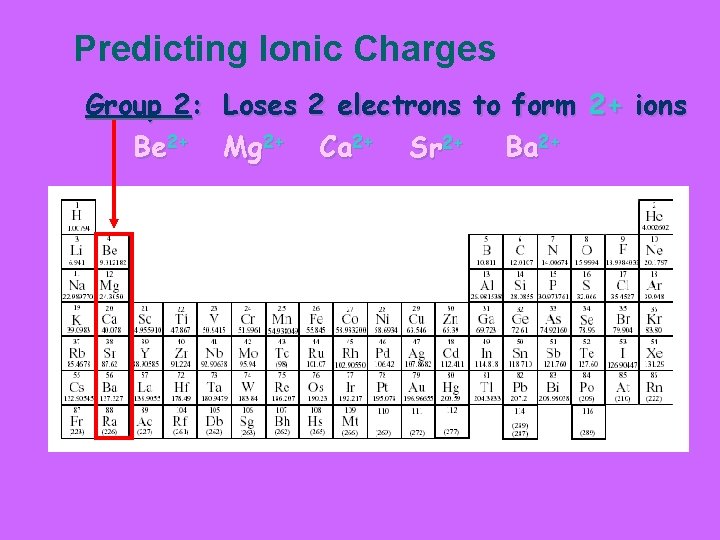
Predicting Ionic Charges Group 2: Loses 2 electrons to form 2+ ions Be 2+ Mg 2+ Ca 2+ Sr 2+ Ba 2+
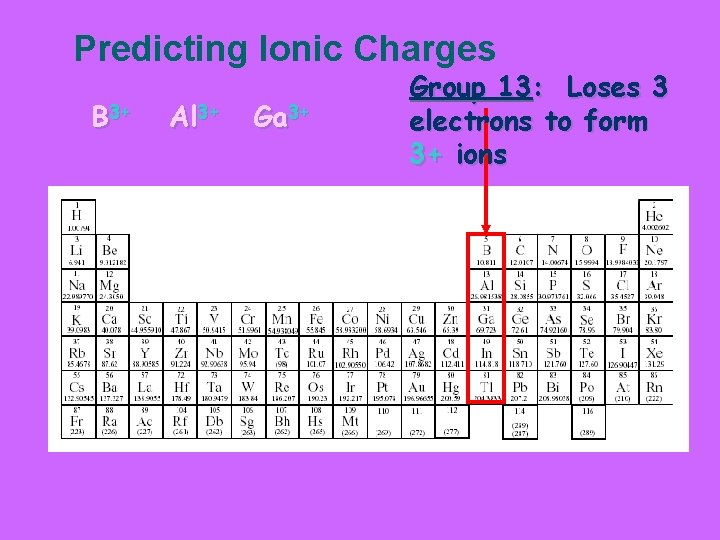
Predicting Ionic Charges B 3+ Al 3+ Ga 3+ Group 13: Loses 3 electrons to form 3+ ions

Predicting Ionic Charges Neither! Group 13 elements rarely form ions. Group 14: Lose 4 electrons or gain 4 electrons?
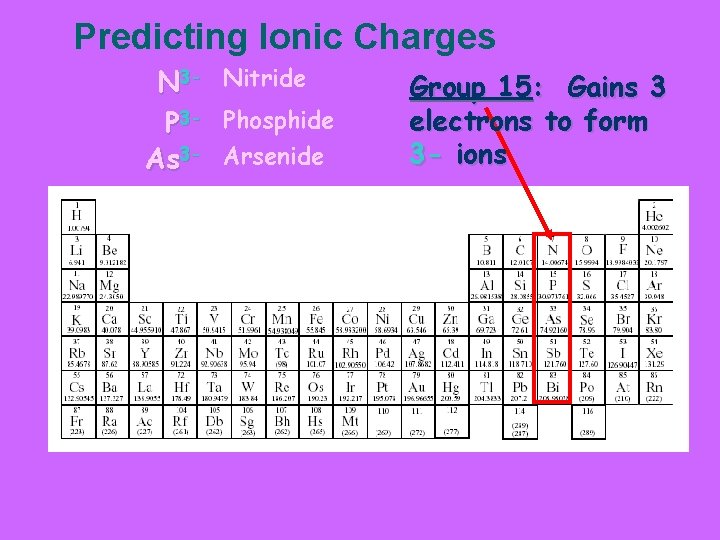
Predicting Ionic Charges N 3 - Nitride P 3 - Phosphide As 3 - Arsenide Group 15: Gains 3 electrons to form 3 - ions
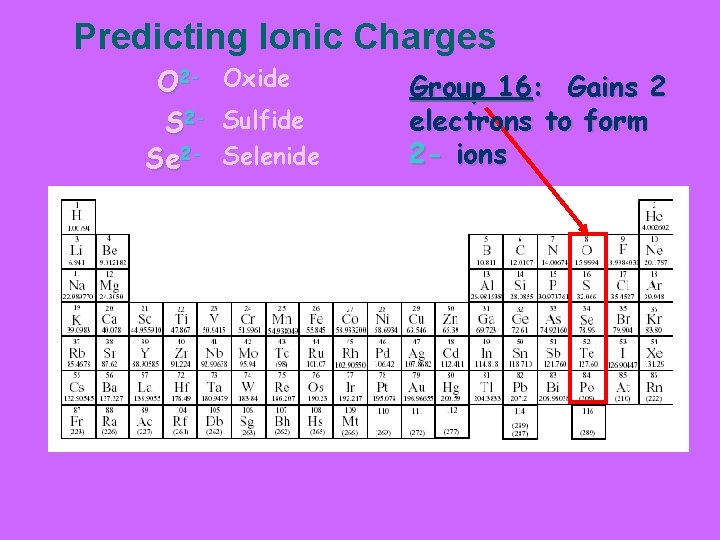
Predicting Ionic Charges O 2 - Oxide S 2 - Sulfide Se 2 - Selenide Group 16: Gains 2 electrons to form 2 - ions
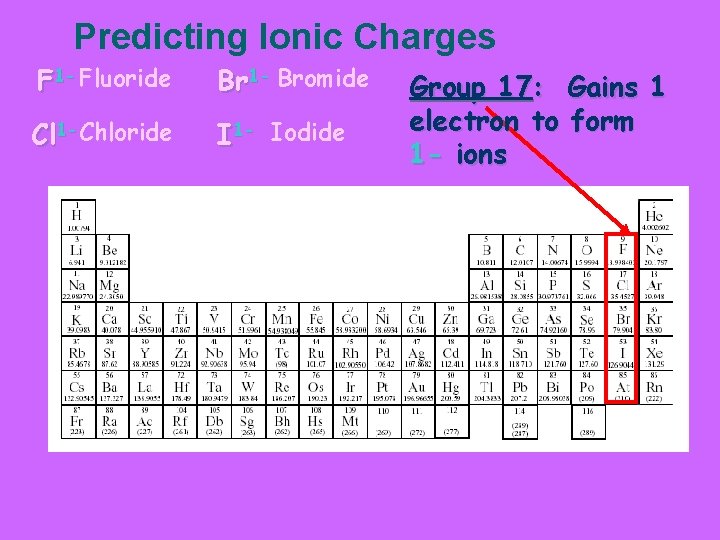
Predicting Ionic Charges F 1 - Fluoride Br 1 - Bromide Cl 1 -Chloride I 1 - Iodide Group 17: Gains 1 electron to form 1 - ions
Predicting Ionic Charges Group 18: Stable Noble gases do not form ions!
Models of Atoms

Dalton Model click here A solid, indivisible sphere

John Dalton click here Click here

Dalton's Atomic Theory l l l All matter is made of atoms ·An atom is indivisible. Atoms of the same element are identical. Atoms of different elements are different. Atoms are rearranged to produce new substances. Atoms combine in whole number ratios.
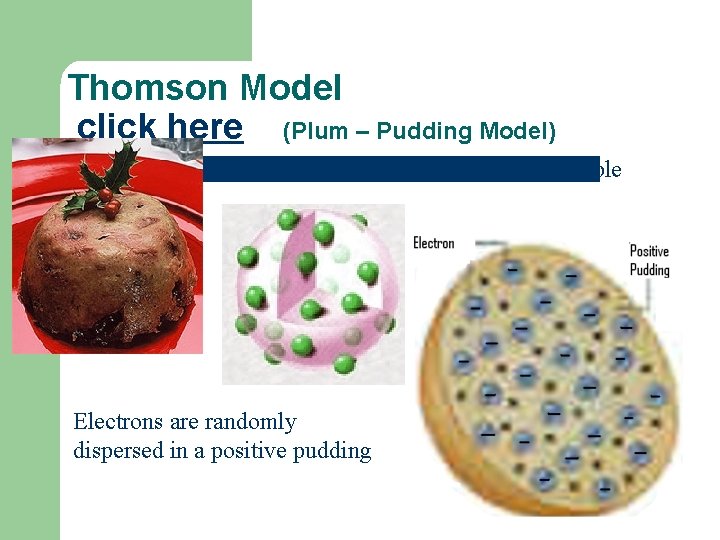
Thomson Model click here (Plum – Pudding Model) Atom no longer considered indivisible Electrons are randomly dispersed in a positive pudding

JJ. Thomson click here

Click here


Mass of the Electron click here 1909 – Robert Millikan determines the mass of the electron. The oil drop apparatus Mass of the electron is 9. 109 x 10 -31 kg
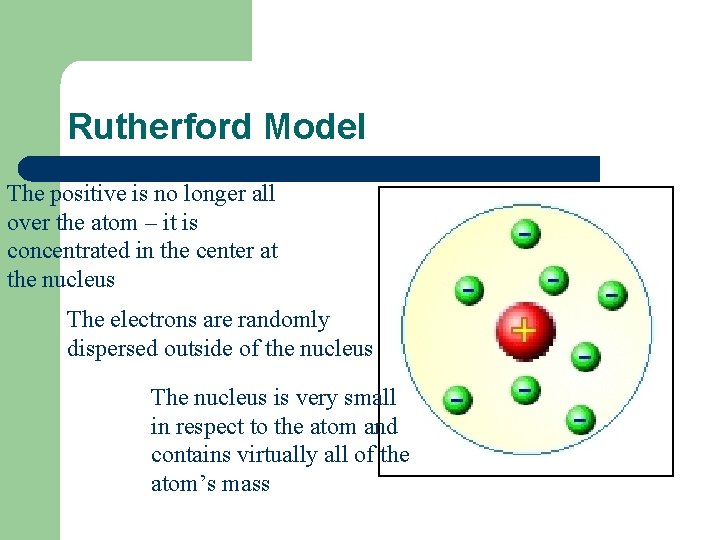
Rutherford Model The positive is no longer all over the atom – it is concentrated in the center at the nucleus The electrons are randomly dispersed outside of the nucleus The nucleus is very small in respect to the atom and contains virtually all of the atom’s mass
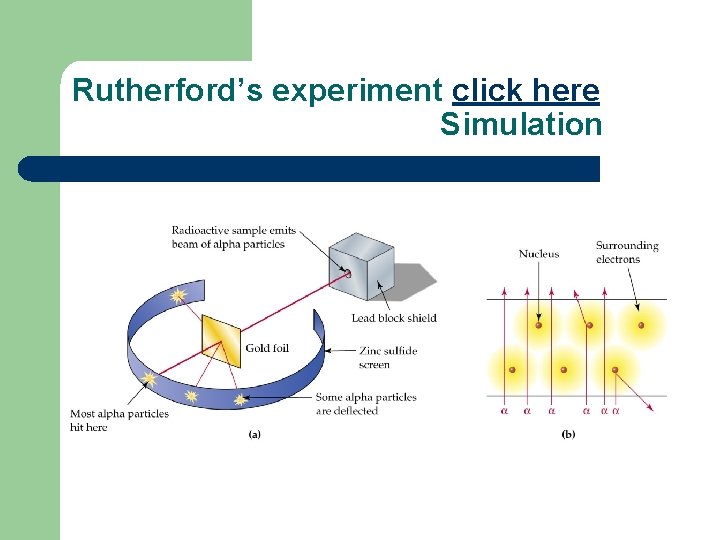
Rutherford’s experiment click here Simulation

Try it Yourself! In the following pictures, there is a target hidden by a cloud. To figure out the shape of the target, we shot some beams into the cloud and recorded where the beams came out. Can you figure out the shape of the target?
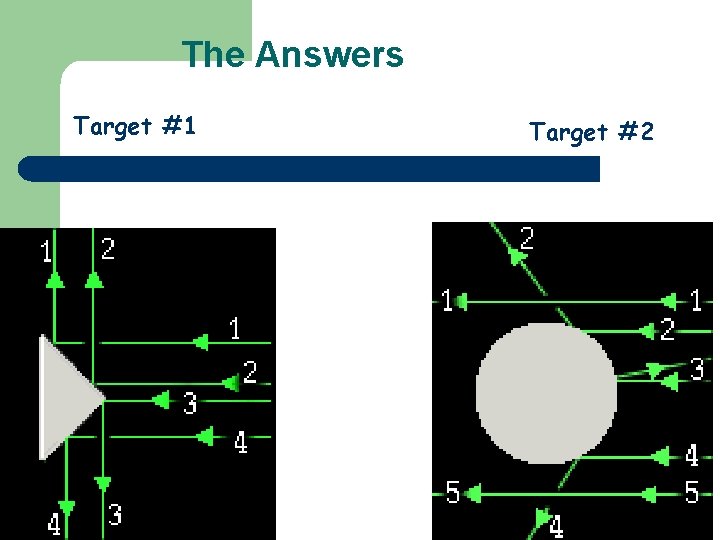
The Answers Target #1 Target #2
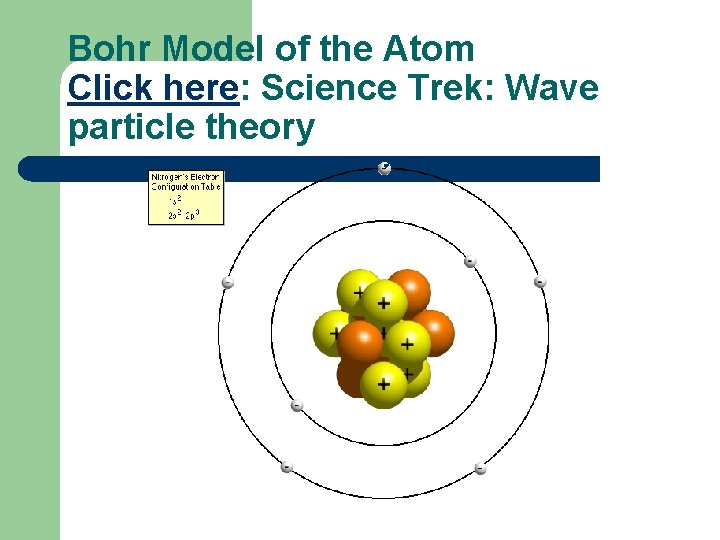
Bohr Model of the Atom Click here: Science Trek: Wave particle theory

Click here How things work: atoms Wave Model – Electron Cloud Model
- Slides: 66