Atomic Structure BASIC INFORMATION Objective To be able

Atomic Structure BASIC INFORMATION Objective: To be able to draw the structure of the atom, and given an element tell how many protons, neutrons, and electrons it has.
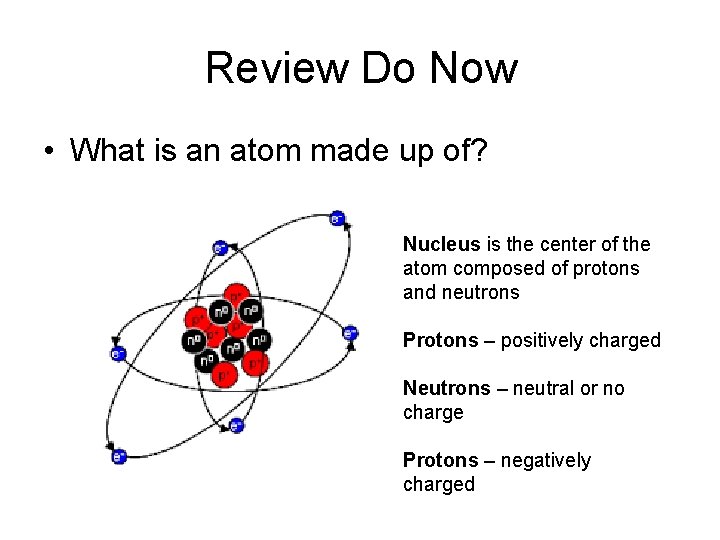
Review Do Now • What is an atom made up of? Nucleus is the center of the atom composed of protons and neutrons Protons – positively charged Neutrons – neutral or no charge Protons – negatively charged
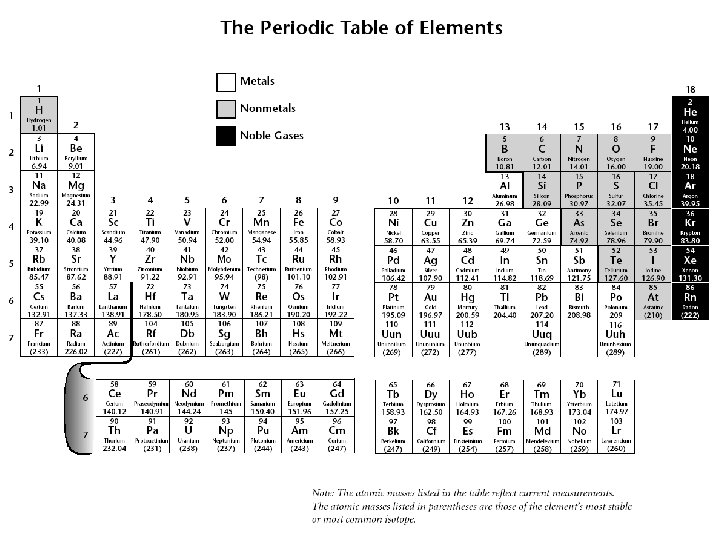
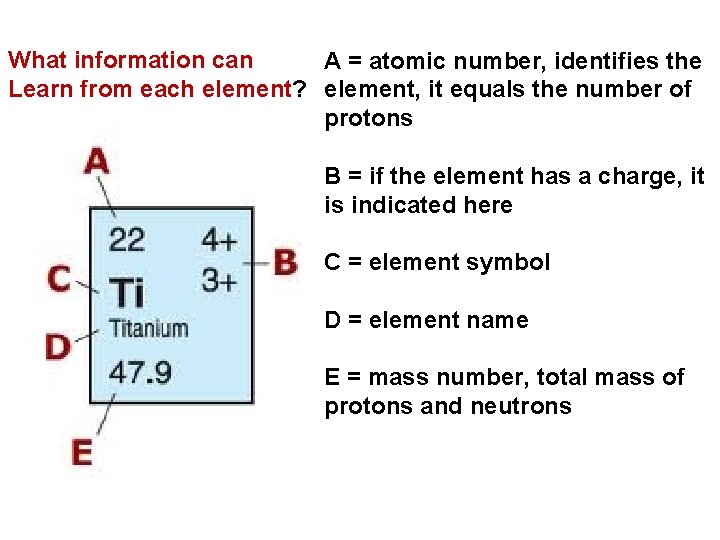
What information can A = atomic number, identifies the Learn from each element? element, it equals the number of protons B = if the element has a charge, it is indicated here C = element symbol D = element name E = mass number, total mass of protons and neutrons
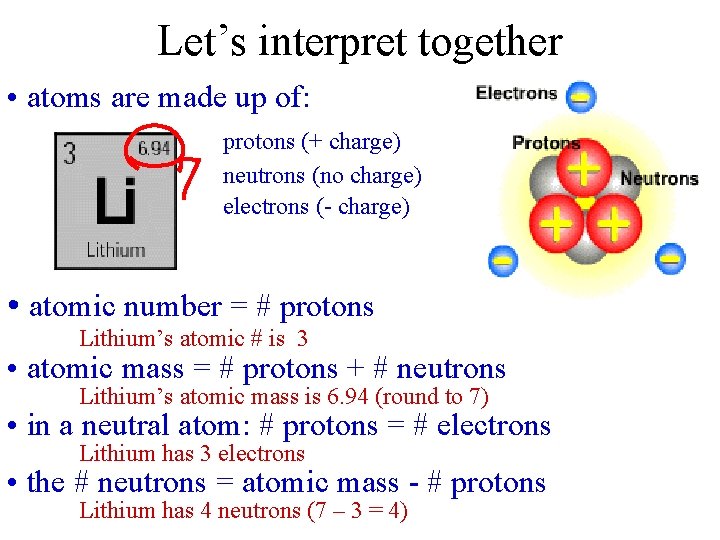
Let’s interpret together • atoms are made up of: protons (+ charge) neutrons (no charge) electrons (- charge) • atomic number = # protons Lithium’s atomic # is 3 • atomic mass = # protons + # neutrons Lithium’s atomic mass is 6. 94 (round to 7) • in a neutral atom: # protons = # electrons Lithium has 3 electrons • the # neutrons = atomic mass - # protons Lithium has 4 neutrons (7 – 3 = 4)
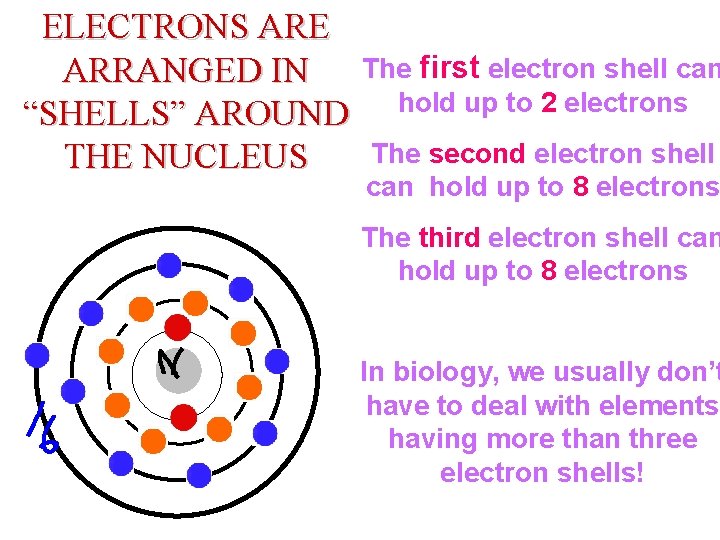
ELECTRONS ARE ARRANGED IN “SHELLS” AROUND THE NUCLEUS Element: Boron = Mass The first electron shell can Symbol: B to 2 electrons #P= hold up # Neutrons = # e = shell The second electron can hold up to 8 electrons # Valence Electrons = The third electron shell can hold up to 8 electrons In biology, we usually don’t have to deal with elements having more than three electron shells!

Which of an atom’s electrons are involved in bonding with other atoms? Ø Only the electrons in its outermost shell! These electrons are called the valence electrons! Hydrogen atoms have 1 valence electron Helium atoms have 2 valence electrons
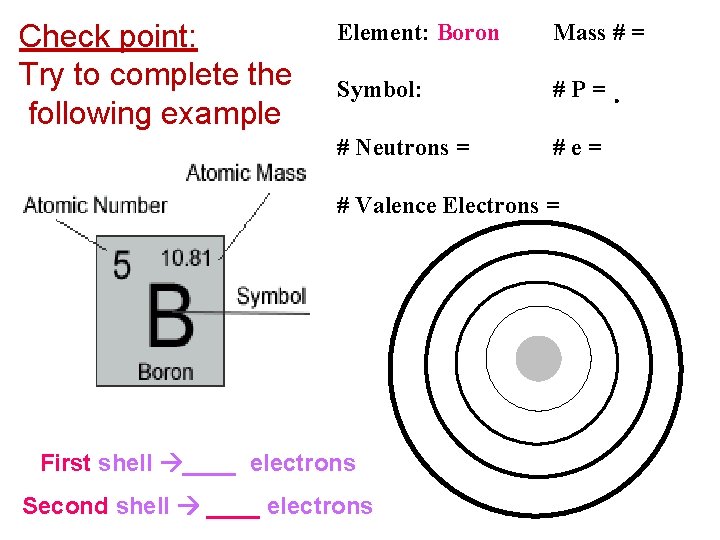
Check point: Try to complete the following example Element: Boron Mass # = Symbol: #P= # Neutrons = #e= # Valence Electrons = First shell ____ electrons Second shell ____ electrons

Now, complete your worksheets !
- Slides: 9