Atomic Structure and Periodicity Light and Waves Describing

Atomic Structure and Periodicity

Light and Waves
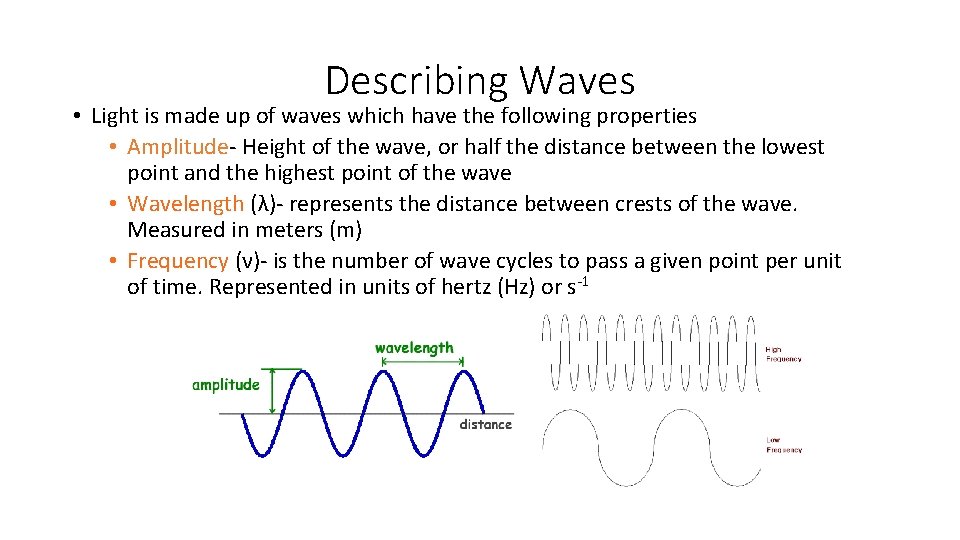
Describing Waves • Light is made up of waves which have the following properties • Amplitude- Height of the wave, or half the distance between the lowest point and the highest point of the wave • Wavelength (λ)- represents the distance between crests of the wave. Measured in meters (m) • Frequency (ν)- is the number of wave cycles to pass a given point per unit of time. Represented in units of hertz (Hz) or s-1

The speed of light! • Frequency and Wavelength in a wave are inversely proportional to each other • As wavelength increases, frequency decreases and as frequency increases wavelength decreases • The Speed of Light = Wavelength x Frequency • c= λν • c=2. 998 x 108 m/s • This is true for ALL electromagnetic waves!!
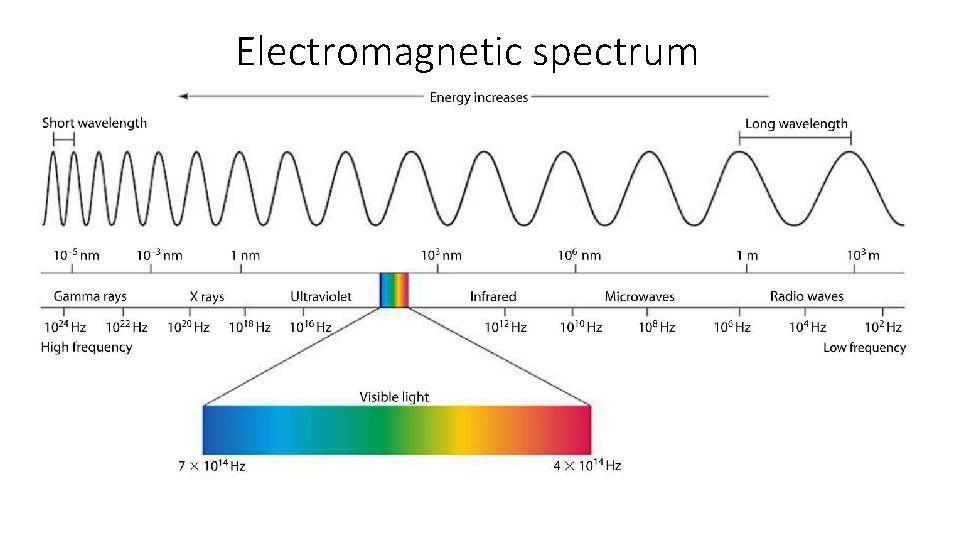
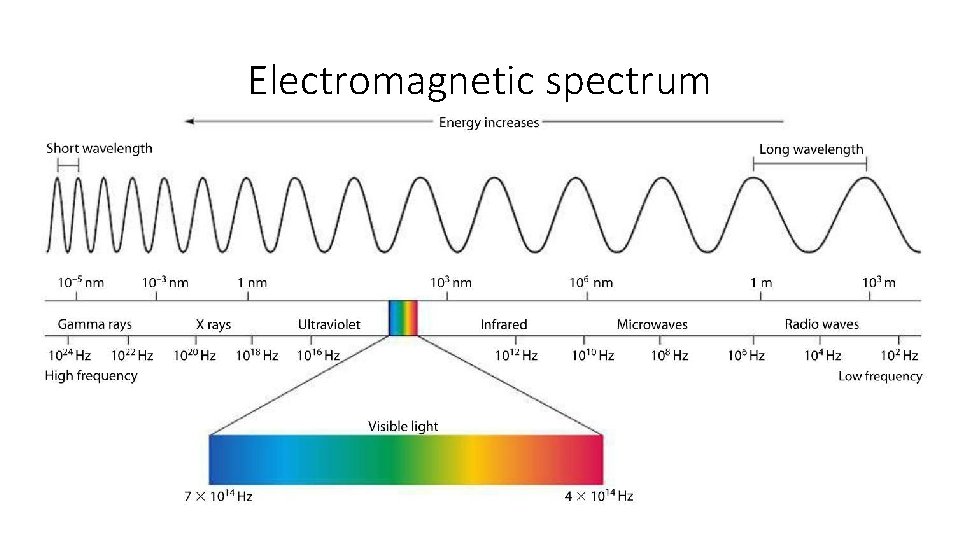
Electromagnetic spectrum

Atomic Model
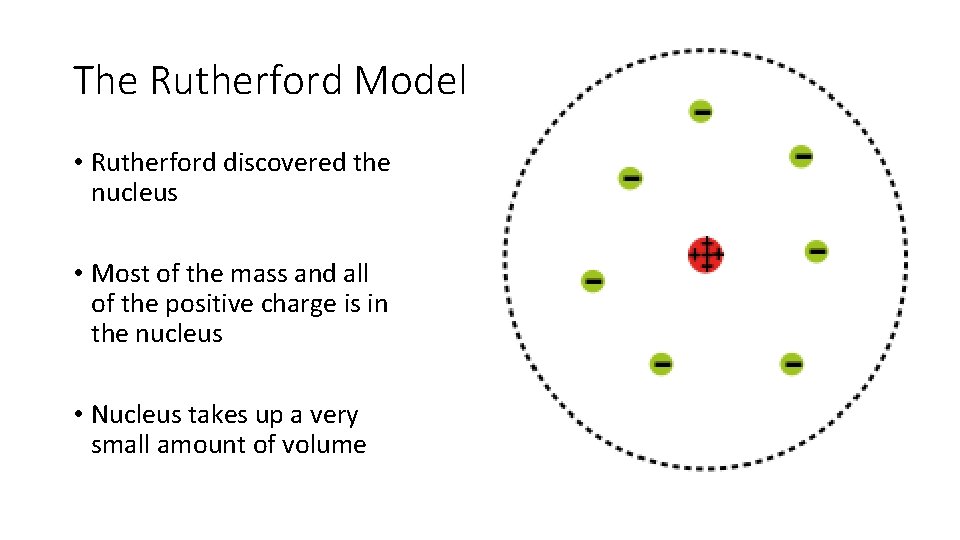
The Rutherford Model • Rutherford discovered the nucleus • Most of the mass and all of the positive charge is in the nucleus • Nucleus takes up a very small amount of volume
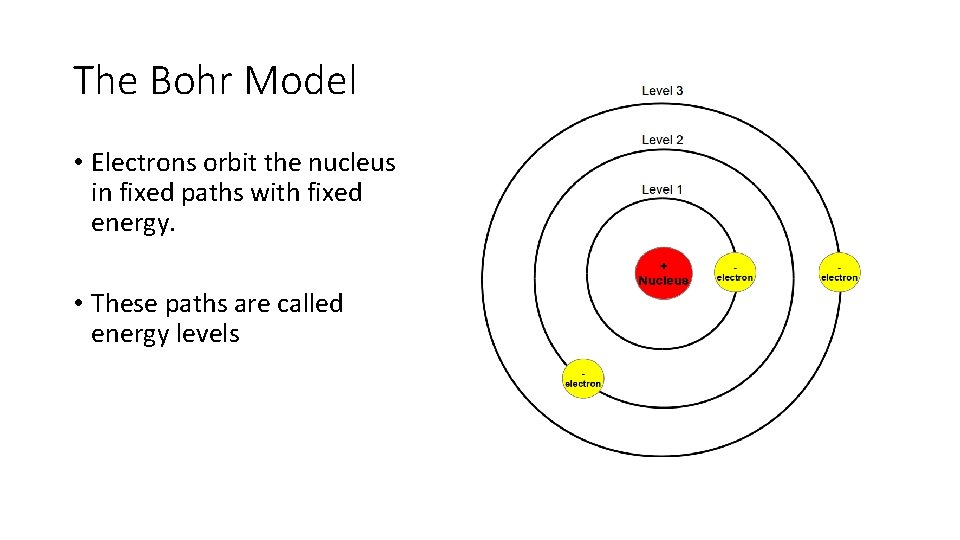
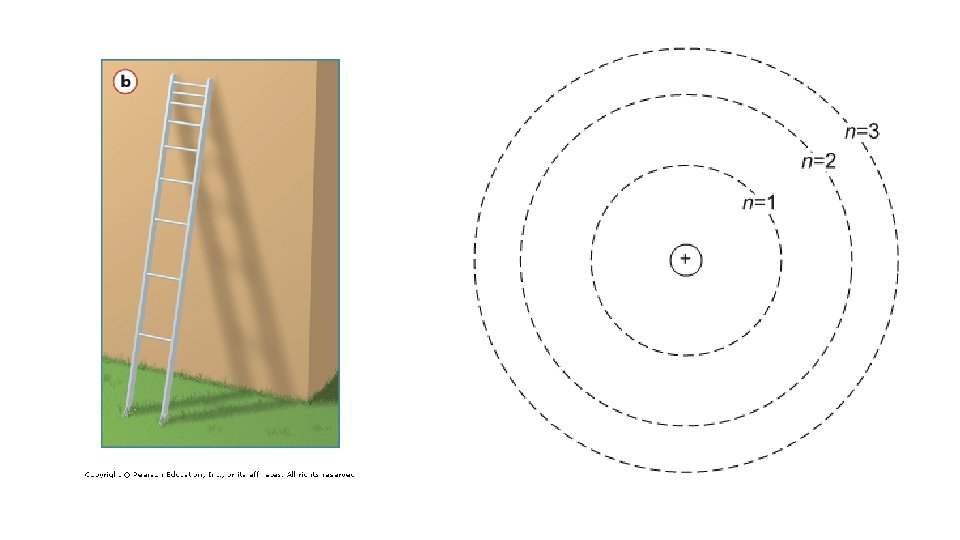
The Bohr Model • Electrons orbit the nucleus in fixed paths with fixed energy. • These paths are called energy levels

Erwin Schrödinger Said electrons move around the nucleus, but depending on their energy level the probability of their location can be determined. He developed an equation to predict location.

Quantum Mechanics v. Classical Mechanics • Deals with physical bodies on the microscale • Deals with physical bodies on the macro-scale • Atoms, molecules, subatomic particles, waves of light • Things you can see with the naked eye, people, planes, planets, stars

Quantum • Electrons have fixed amounts of energy depending on which energy level they occupy. • The minimum amount of change in energy is called a quantum. • Energy is quantized meaning it can only be certain amounts.

Quantum Numbers

Quantum Numbers • A set of numbers that give acceptable solutions to the Schrödinger wave function • Each electrons has four quantum numbers

Principle Quantum Number • Abbreviation : n • Tells the energy level of an electron • Values n=1, 2, 3…. 7

Angular Quantum Number • Abbreviation: ℓ • Tells the sublevel an electron is located in and its shape • Values: ℓ = 0, 1, 2… n-1 • Often called s, p, d, f
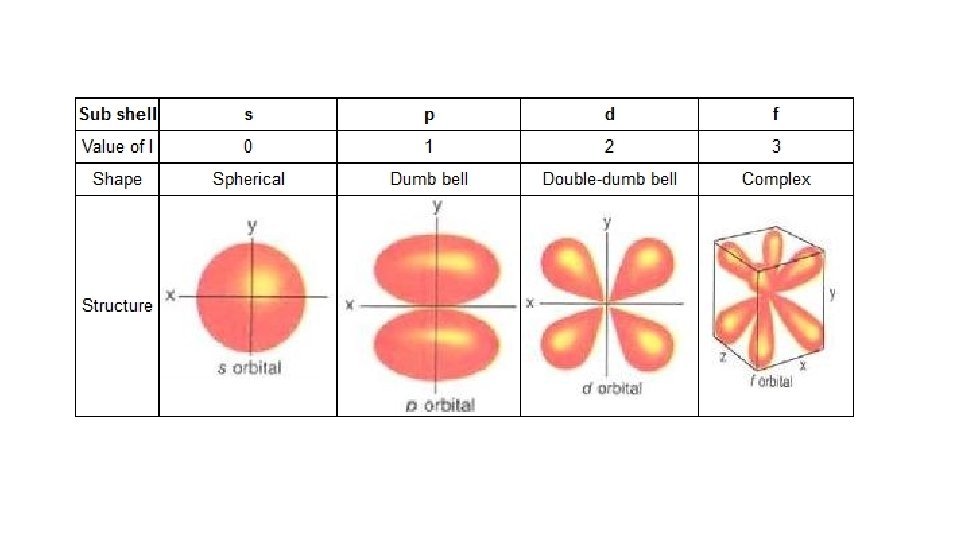

Magnetic Quantum Number • Abbreviation: m • Tells the specific orbital an electron • Values: m= - ℓ… 0… ℓ with all integer values in between • Tells you how many sublevels an energy level can have


Spin Quantum Number • Abbreviation: s • Tells the spin an electron has in an orbital • s= ±½ • Only 2 electrons fit in an orbital and they must have opposite spin
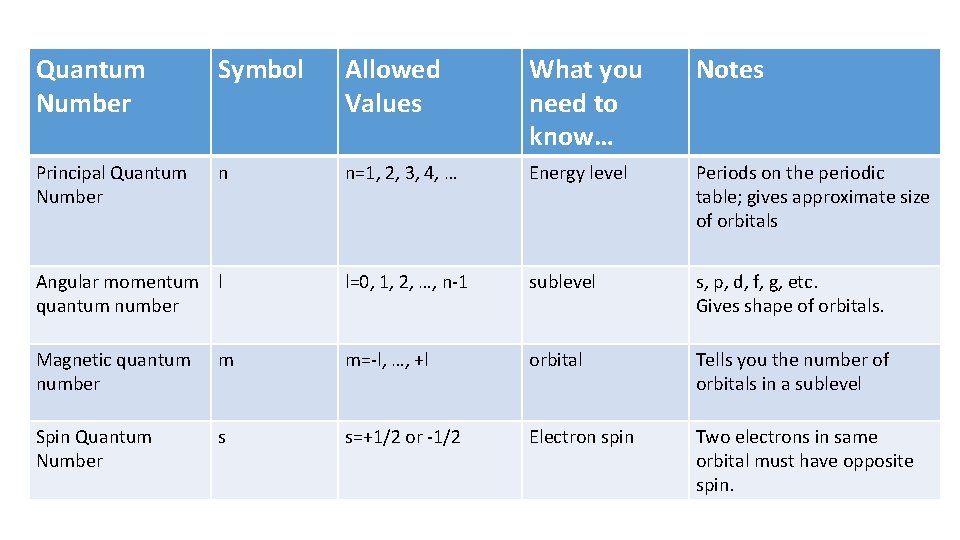
Quantum Number Symbol Allowed Values What you need to know… Notes Principal Quantum Number n n=1, 2, 3, 4, … Energy level Periods on the periodic table; gives approximate size of orbitals Angular momentum l quantum number l=0, 1, 2, …, n-1 sublevel s, p, d, f, g, etc. Gives shape of orbitals. Magnetic quantum number m m=-l, …, +l orbital Tells you the number of orbitals in a sublevel Spin Quantum Number s s=+1/2 or -1/2 Electron spin Two electrons in same orbital must have opposite spin.

Electron Configuration

Summary Principle Energy Levels n=1 through n=7 Sub Levels s, p, d, f Orbitals s- 2 electrons p- 6 electrons d- 10 electrons f- 14 electrons

Rules of Electron configuration The Aufbau Principle Pauli Exclusion Principle Hund’s Rule
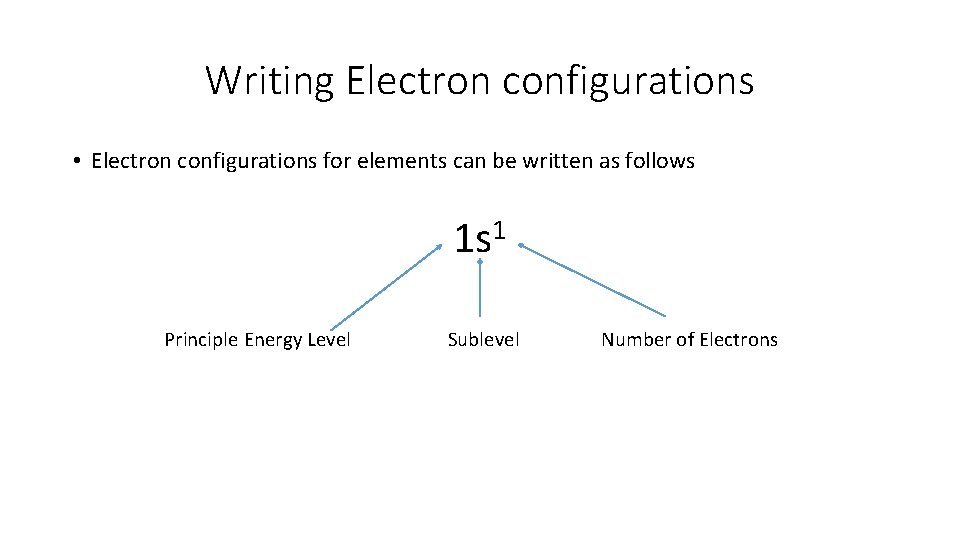
Writing Electron configurations • Electron configurations for elements can be written as follows 1 s 1 Principle Energy Level Sublevel Number of Electrons

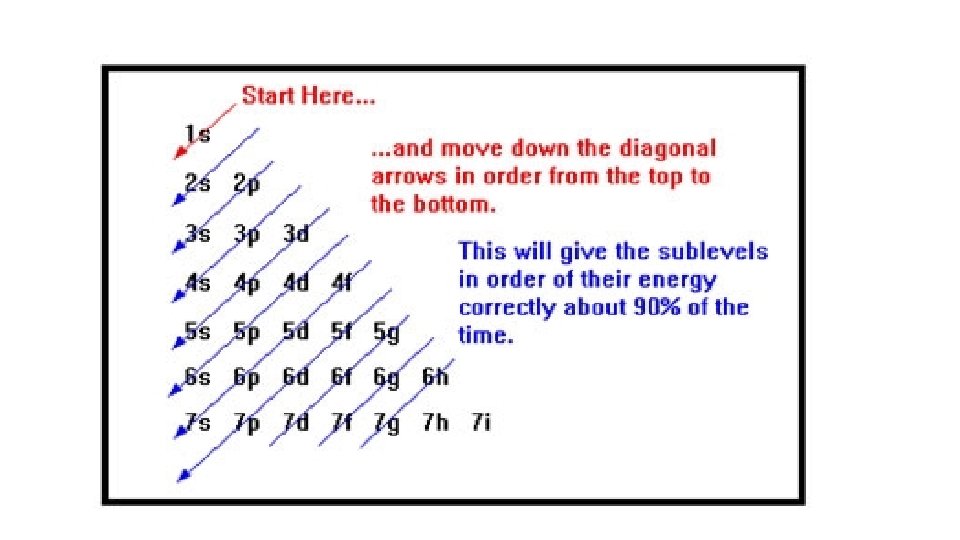
The Aufbau Principle • Electrons occupy the orbitals of the lowest energy first • Within a principle energy level the s sublevel is always the lowest energy level • However the range of energy levels within a principal energy level can overlap the energy levels of another principle level • e. g. 4 s has lower energy than 3 d
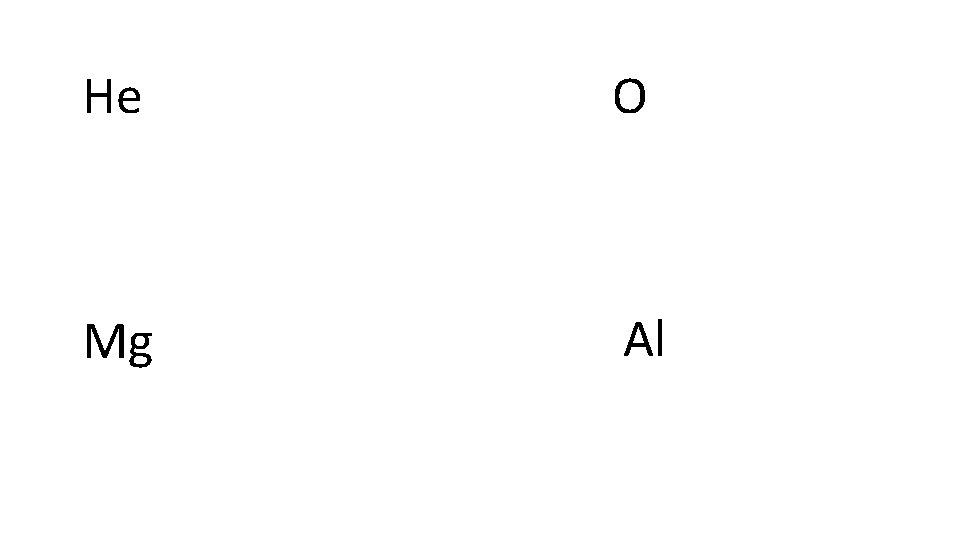
He O Mg Al
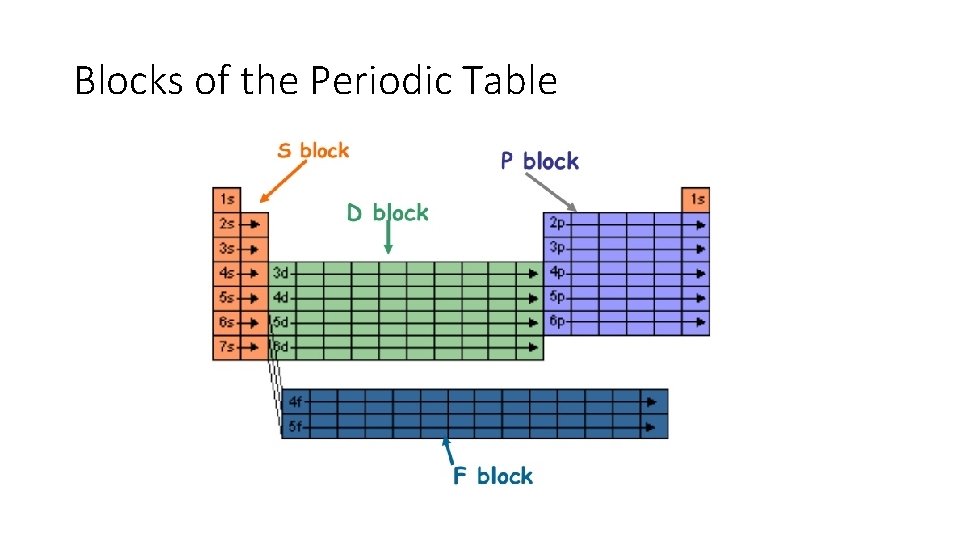
Blocks of the Periodic Table

Noble gas configuration • Electron Configuration for Ne is 1 s 22 p 6 • Electron Configuration for Na is 1 s 22 p 63 s 1 • The symbol in brackets for the last noble gas before the element you’re writing out can be substituted • [Ne] 3 s 1

Exceptions to the Aufbau Principle Copper should have an electron configuration of 1 s 22 p 63 s 23 p 63 d 94 s 2 But instead has an electron configuration of 1 s 22 p 63 s 23 p 63 d 104 s 1 This is because full energy levels are more stable than partially filled ones and because the 3 d and 4 s are so close in energy the tendency to be stable can overcome the small energy difference No exceptions before atomic number 23 Other exceptions: Chromium, Molybdenum, Silver

F Al Na Sr

Orbital Notation • Uses blocks and arrows to show the electron configuration of electrons • Each orbital can only hold 2 electrons s p x, p y , p z
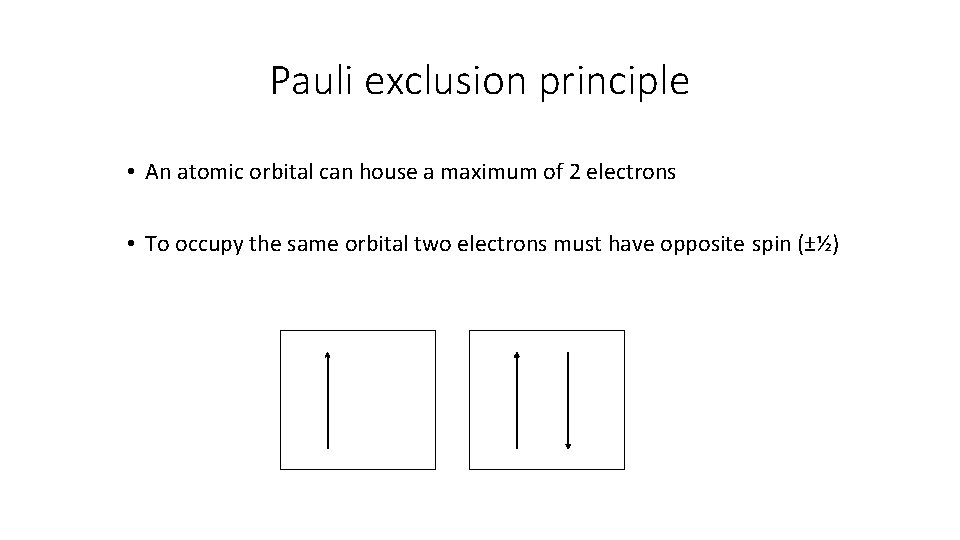
Pauli exclusion principle • An atomic orbital can house a maximum of 2 electrons • To occupy the same orbital two electrons must have opposite spin (±½)

Hund’s rule • Every orbital in a sublevel is singly occupied before any orbital is doubly occupied. • All of the electrons in singly occupied orbitals have the same spin (to maximize total spin)

He O Mg S
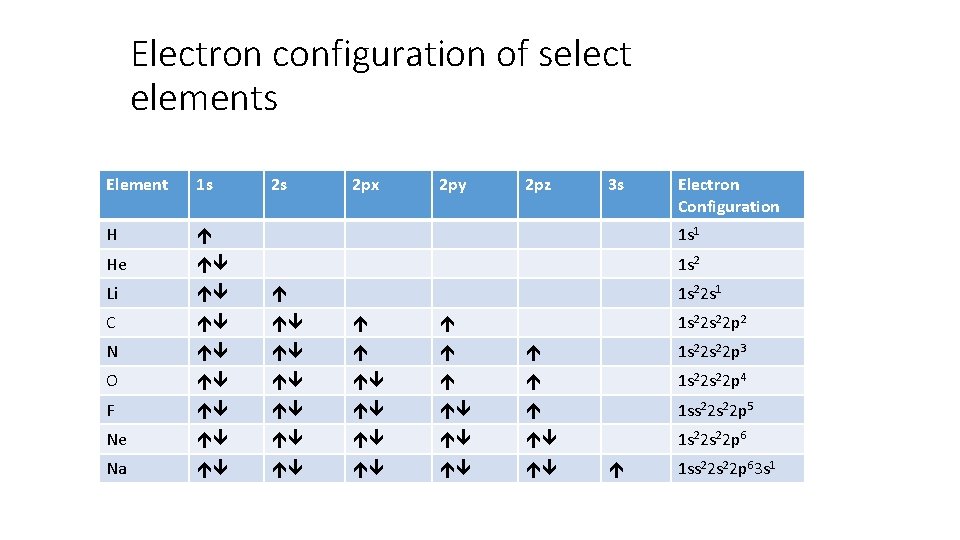
Electron configuration of select elements Element 1 s 2 s 2 px 2 py 2 pz 3 s Electron Configuration H 1 s 1 He 1 s 2 Li C N 1 s 22 p 3 O 1 s 22 p 4 F 1 ss 22 p 5 Ne 1 s 22 p 6 Na 1 s 22 s 1 1 s 22 p 2 1 ss 22 p 63 s 1

Atomic Emission Spectra
Electromagnetic spectrum
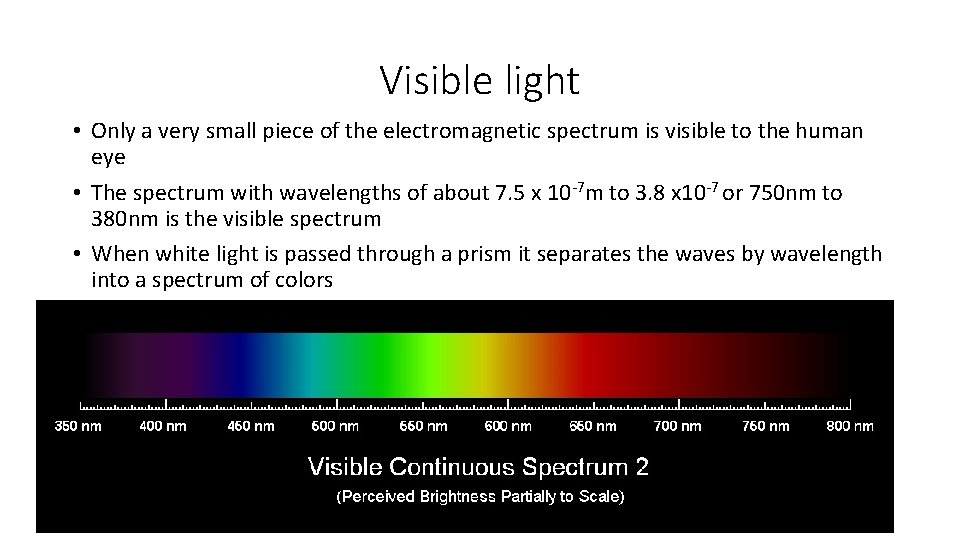
Visible light • Only a very small piece of the electromagnetic spectrum is visible to the human eye • The spectrum with wavelengths of about 7. 5 x 10 -7 m to 3. 8 x 10 -7 or 750 nm to 380 nm is the visible spectrum • When white light is passed through a prism it separates the waves by wavelength into a spectrum of colors

Electrons and Energy • Electrons occupy certain areas (energy levels) based on the amount of energy they contain • If electrons absorb energy they will move up to higher energy levels, their excited state • When electrons fall back down into their normal energy levels they release the energy they absorbed

Electrons and Energy cont. • The energy released comes out of the atom in the form of an electromagnetic wave • The amount of energy is directly proportional to the frequency of the wave • The frequency of the wave is inversely proportional to the wavelength of the wave

Electrons and Energy cont. • Certain wavelengths of energy are visible to humans • The energy transitions are quantized, meaning only specific amounts of energy can be released • Therefore, only specific colors of light can be emitted

Atomic emission spectra (continued) • When the light emitted by the energized electrons of a gaseous element is passed through a prism the spectrum consists of a limited number of narrow lines of light • These lines of light are called the atomic emission spectrum • The wavelengths of these lights are characteristic of the element

Hydrogen Gas Lamp
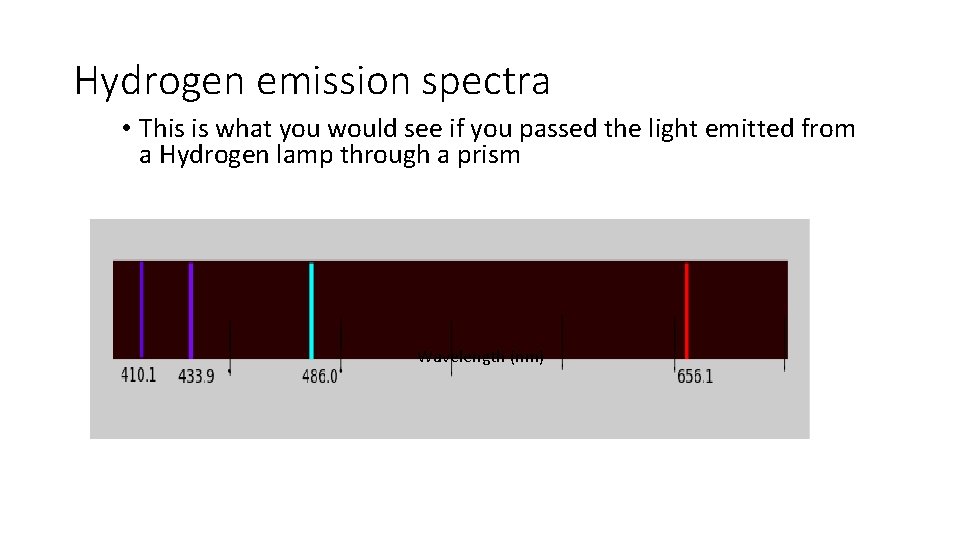
Hydrogen emission spectra • This is what you would see if you passed the light emitted from a Hydrogen lamp through a prism Wavelength (nm)
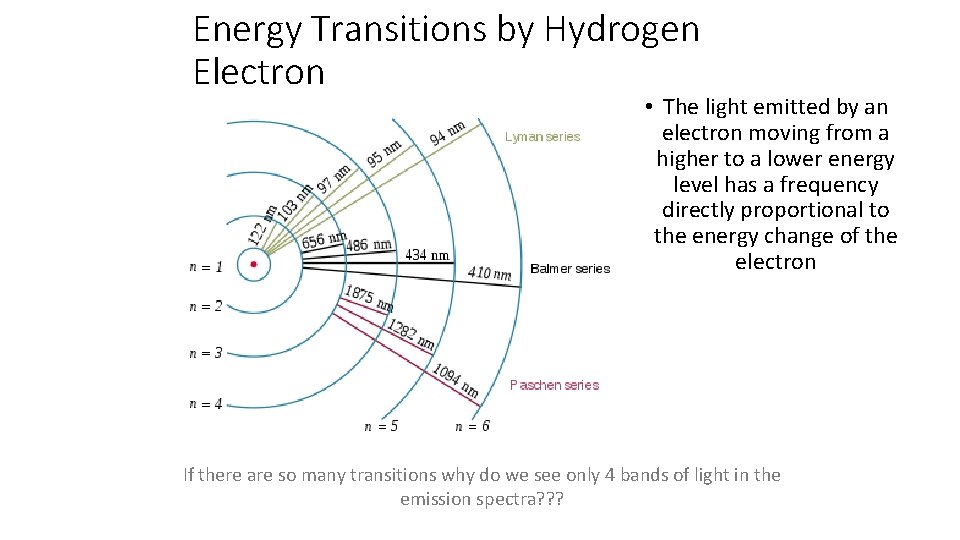
Energy Transitions by Hydrogen Electron • The light emitted by an electron moving from a higher to a lower energy level has a frequency directly proportional to the energy change of the electron If there are so many transitions why do we see only 4 bands of light in the emission spectra? ? ?

Black Body Radiation

Max Planck • In 1900 German physicist Max Planck tried to explain why a “black body” changes colors as temperature is increased • Could only be explained if the energy of the “black body” changed only in small, discrete units called quanta • Proved mathematically the amount of radiant energy of a single quantum absorbed or emitted is directly proportional to the frequency of the radiation E=hν Energy (J) Planck’s Constant (J×s) • Planck’s constant (h) = 6. 626× 10 -34 J×s Frequency (s-1)

Black Body Radiation • Higher Temperature means higher energy • Higher Energy means higher frequency • Higher frequency means lower wavelength

The Photoelectric Effect
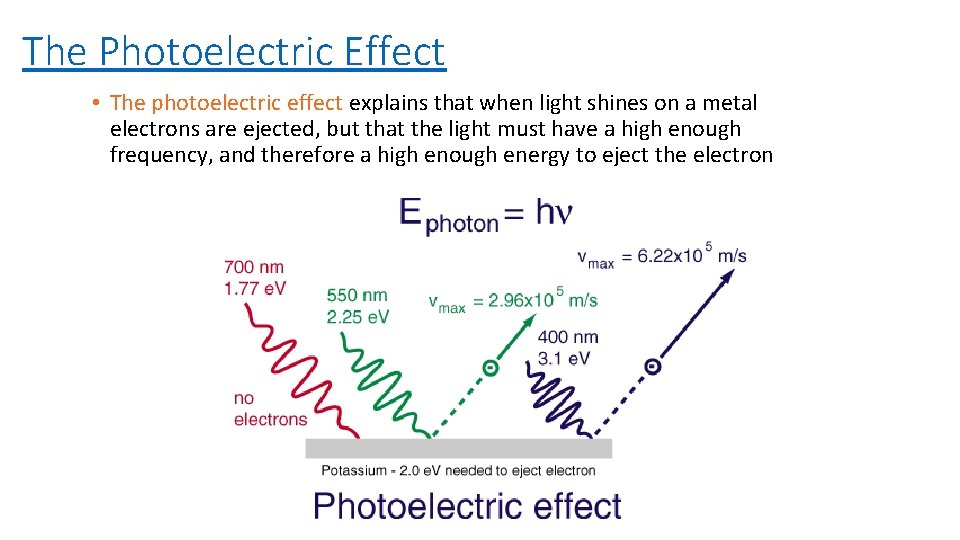
The Photoelectric Effect • The photoelectric effect explains that when light shines on a metal electrons are ejected, but that the light must have a high enough frequency, and therefore a high enough energy to eject the electron

Wave-particle duality • To explain the photoelectric effect Einstein proposed that light could be described a quanta of energy that behave as if they were particles. These light quanta are called photons "It seems as though we must use sometimes the one theory and sometimes the other, while at times we may use either. We are faced with a new kind of difficulty. We have two contradictory pictures of reality; separately neither of them fully explains the phenomena of light, but together they do " -Albert Einstein


Quantum mechanics • de. Broglie hypothesized that if light could be described as both a wave and a stream of particles that all solid objects should be able to be described the same way λ=h/mv Wavelength (m) Planck’s Constant mass(kg) velocity(m/s) Planck’s constant (h) = 6. 626× 10 -34 J×s • Wavelength is inversely proportional to momentum (mass * velocity)
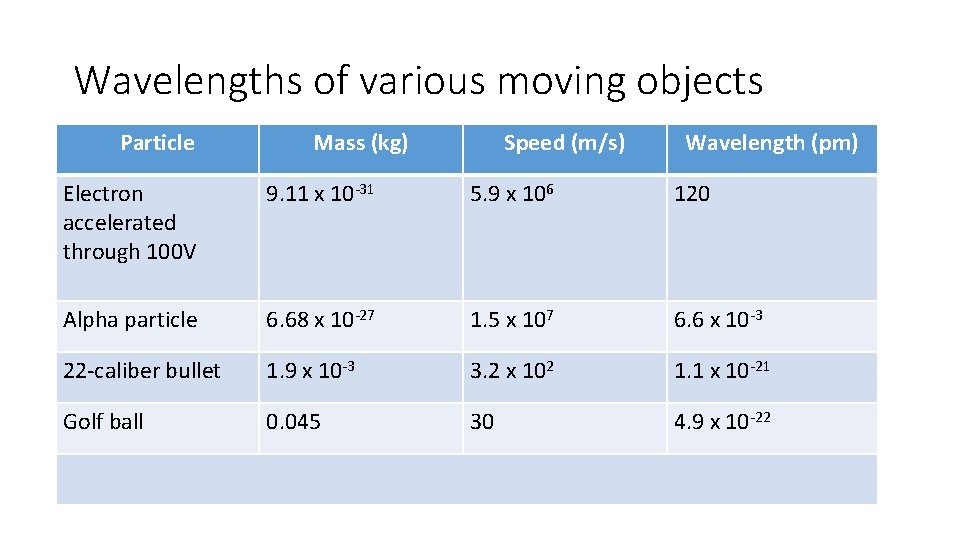
Wavelengths of various moving objects Particle Mass (kg) Speed (m/s) Wavelength (pm) Electron accelerated through 100 V 9. 11 x 10 -31 5. 9 x 106 120 Alpha particle 6. 68 x 10 -27 1. 5 x 107 6. 6 x 10 -3 22 -caliber bullet 1. 9 x 10 -3 3. 2 x 102 1. 1 x 10 -21 Golf ball 0. 045 30 4. 9 x 10 -22

The birth of quantum mechanics • de. Broglie’s prediction that all matter can behave as both a particle and a wave led to a new theory called QUANTUM MECHANICS Classical Mechanics describes the motions of bodies much larger that atoms Quantum Mechanics describes the motion of subatomic particle and atoms as waves

Double Slit Experiment • Was an experiment preformed by Thomas Young in 1803 that proved the wave-like nature of particles! watch!

The Heisenberg uncertainty principle • States that you cannot know the velocity of a particle and the position of a particle at the same time • You can know the position, but not velocity! • You can know velocity but not position!

Quick snapshot we know the balls exact position but not its velocity We can look at the balls path, and calculate its exact velocity but not know its exact position
- Slides: 61