Atomic Structure and Periodic Table Atoms 1 Atoms

Atomic Structure and Periodic Table

Atoms #1 Atoms # 2 Period Table 1 Period Table Organization Higher End 10 10 10 20 20 20 30 30 30 40 40 40 50 50 50


Topic 1 – 10 Points QUESTION: • What are the 3 subatomic parts of an Atom. • A. Proton, Nucleus, Electrons • B. Protons, Neutrons, Atomic Mass • C. Proton, Neutrons, Electrons • D. Nucleus, Electron Cloud and Atomic Mass ANSWER: • C. Protons, Neutrons, Electrons
Topic 1 – 20 Points QUESTION: • The nucleus contains what two subatomic parts? • A. The Electron Cloud and Electrons • B. The Protons and Electrons • C. The Protons and Neutrons • D. The Neutrons and Electrons ANSWER: • C The Protons and Neutrons
Topic 1 – 30 Points QUESTION: • What Acronym do we use to calculate Protons, Neutrons and Electrons? • A. GRAPE • B. APE MAN • C. MAN VERSUS APE • D. APE VERSUS MAN ANSWER: • APE MAN
Topic 1 – 40 Points QUESTION: • Name the two subatomic particles located inside the nucleus of an atom. • A. Protons and Electrons • B. Nucleus and Electron Cloud • C. Neutrons and Electrons • D. Protons and Neutrons ANSWER: • D. Protons and Neutrons
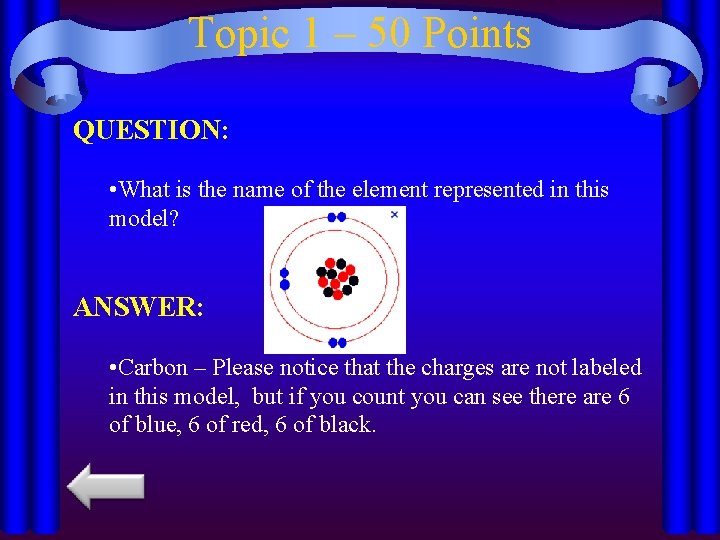
Topic 1 – 50 Points QUESTION: • What is the name of the element represented in this model? ANSWER: • Carbon – Please notice that the charges are not labeled in this model, but if you count you can see there are 6 of blue, 6 of red, 6 of black.

Topic 2 – 10 Points QUESTION: Which of the following is TRUE? A. Protons are Positive and Neutrons are Negative B. Protons are Positive and Electrons are Negative C. Neutrons are Negative and Electrons Positive D. Protons are Negative and Electrons are Positive ANSWER: • B. Protons are Positive – Electrons are negative.

Topic 2 – 20 Points QUESTION: Which pair of elements would have the same number of valence electrons. A. Li and Mg B. Li and Be C. Be and Mg D. Be and Na ANSWER: C. Be and Mg - BOUNUS --- HOW MANY VALENCE ELECTRONS DO THEY HAVE
Topic 2 – 30 Points QUESTION: When moving down a column in the Periodic Table, which of the following increases? A. B. C. D. Number of Valence Electrons Melting point Number of Nucleus Non Metal Properties ANSWER: • Number of Valence Electrons
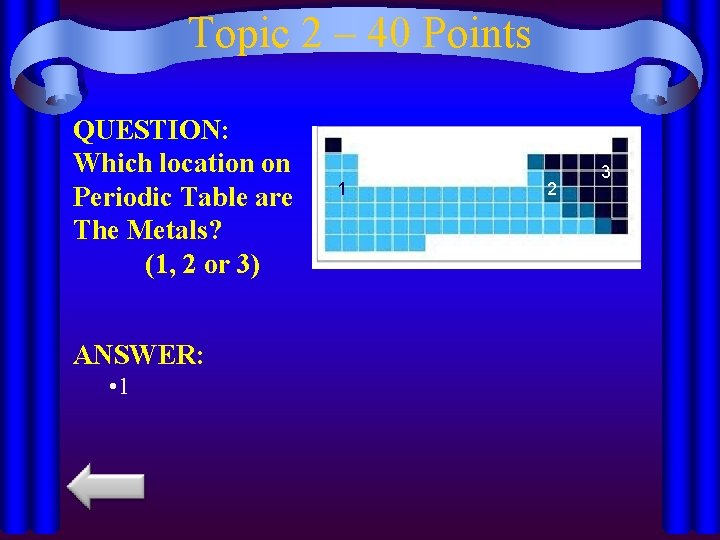
Topic 2 – 40 Points QUESTION: Which location on Periodic Table are The Metals? (1, 2 or 3) ANSWER: • 1 1 2 3

Topic 2 – 50 Points QUESTION: Which Group/Family would be the most reactive and Why? A. Group 1 - because they have the most valence electrons. B. Group 1 – because they have the least valence electrons. C. Group 18 - because they have the most valence electrons. D. Group 18 - because they have the least valence electrons. ANSWER: B – Group 1 because they have the least
Topic 3 – 10 Points QUESTION: Identify the element with 47 Protons. ANSWER: • Ag - Silver
Topic 3 – 20 Points QUESTION: • How many protons does Aluminum (Al) have? • A - 13 • B - 15 • C - 27 ANSWER: • 13
Topic 3 – 30 Points QUESTION: How many groups (families) does the periodic table have? A. 7 B. 18 C. 12 ANSWER: • 18
Topic 3 – 40 Points QUESTION: • Name 2 liquids on the periodic table. A. Nitrogen and Neon B. Mercury and Bromine C. Hydrogen and Helium D. Hydrogen and Lithium ANSWER: • B – Mercury and Bromine
Topic 3 – 50 Points QUESTION: • Identify the Noble Gas with two Valence Electrons. • Write In ANSWER: • Helium
Topic 4 – 10 Points QUESTION: • What period is Titanium (Ti) in? • Period 2 • Period 5 • Period 4 ANSWER: • Period 4

Topic 4 – 20 Points QUESTION: • Where are the metals located on the periodic table? • Top • Bottom • Left • Right ANSWER: • Left
Topic 4 – 30 Points QUESTION: • How many neutrons does Fluorine – 9 have ? A. 9 B. 10 C. 18 D. 19 ANSWER: • Mass – Atomic # = Neutrons • 19 - 9 = 10

Topic 4 – 40 Points QUESTION: How many electrons does Phosphorus – P have? A 15 B 30 C 31 D 16 ANSWER: • Phosphorus – P is element # 15 – So it has 15 electrons.
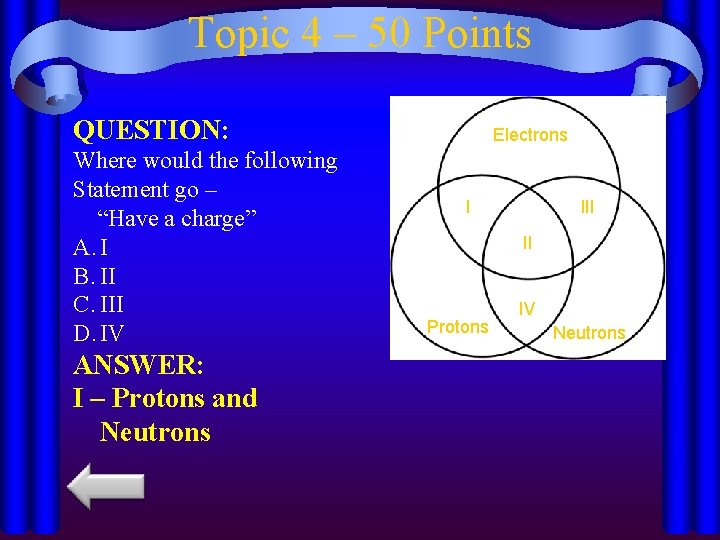
Topic 4 – 50 Points QUESTION: Where would the following Statement go – “Have a charge” A. I B. II C. III D. IV ANSWER: I – Protons and Neutrons Electrons I II Protons IV Neutrons
Topic 5 – 10 Points QUESTION: What are the amount of Electrons allowed on each level? A 2– 8– 2 B 8– 2– 2 C 2– 8– 8 D 8– 8– 8 ANSWER: • C– 2– 8– 8
Topic 5 – 20 Points QUESTION: Where is the majority of mass located in an atom? A. In the Protons B. In the Neutrons C. In the Nucleus D. In the Electrons ANSWER: • C. In the Nucleus (Protons and Neutrons)
Topic 5 – 30 Points QUESTION: • Which two Elements are more reactive? • A. Ne and Ar • B. S and Se • C. Be and Mg • D. H and Li ANSWER: • D. H and Li

Topic 5 – 40 Points QUESTION: • What is an energy Level? A. The amount of mass in the nucleus. B. The ring around the nucleus where the electrons are placed. C. The temp of the Atom. D. The amount of neutrons. ANSWER: • B. The ring around the nucleus where the electrons are placed.
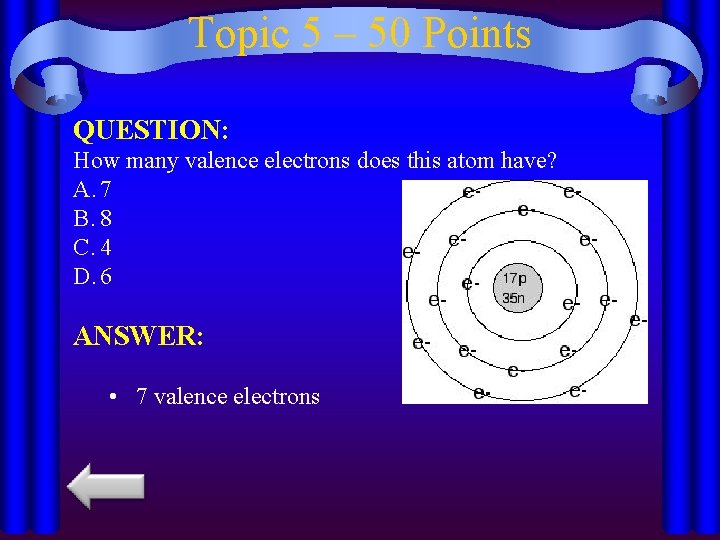
Topic 5 – 50 Points QUESTION: How many valence electrons does this atom have? A. 7 B. 8 C. 4 D. 6 ANSWER: • 7 valence electrons
- Slides: 28