Atomic Structure and Electron Configuration Quantum Mechanical Model


















- Slides: 29

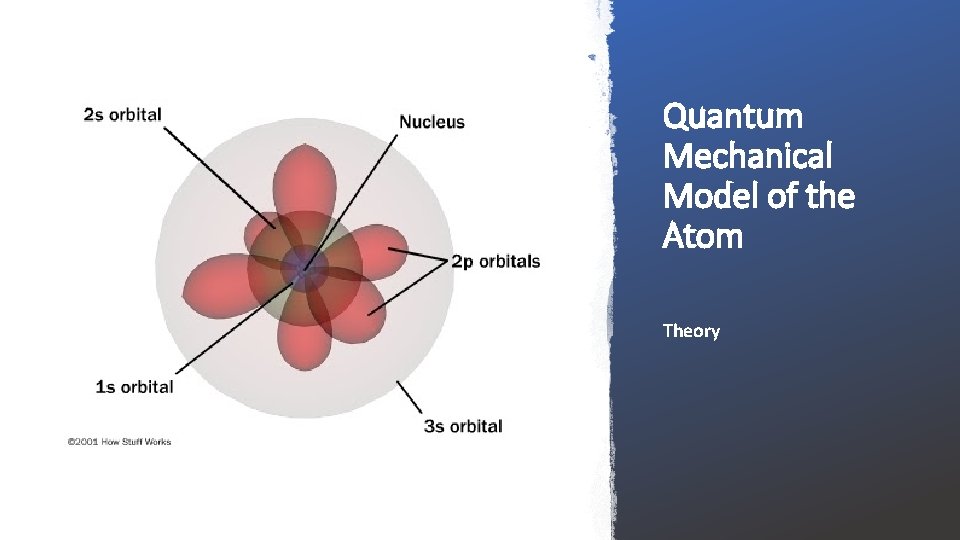
Atomic Structure and Electron Configuration Quantum Mechanical Model of the Atom

The Bohr-Rutherford model of the atom, while easy to understand use, is not the current model of the atom. Developed shortly after the Bohr model, the quantum mechanical model has been the accepted model. However, due to its abstract nature, it is studied in later chemistry courses. We will: 1. look at the historical development of this model 2. use it to show electron arrangements called electron configurations 3. model the electron configuration by constructing orbital diagrams 4. go into the specifics of the model to explain some chemical and physical phenomena of the elements as well as periodic trends

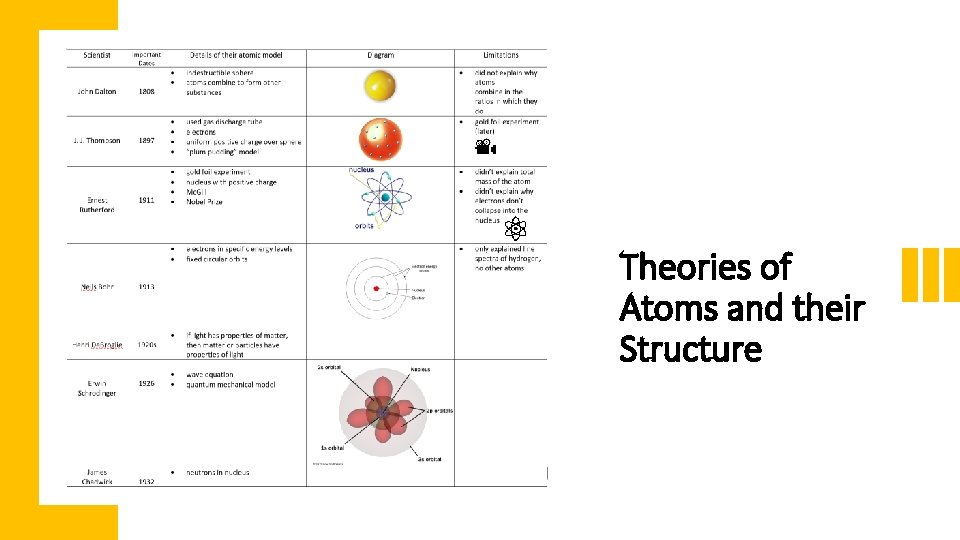
Theories of Atoms and their Structure

Back: Auguste Piccard, Émile Henriot, Paul Ehrenfest, Édouard Herzen, Théophile de Donder, Erwin Schrödinger, JE Verschaffelt, Wolfgang Pauli, Werner Heisenberg, Ralph Fowler, Léon Brillouin. Middle: Peter Debye, Martin Knudsen, William Lawrence Bragg, Hendrik Anthony Kramers, Paul Dirac, Arthur Compton, Louis de Broglie, Max Born, Niels Bohr. Front: Irving Langmuir, Max Planck, Marie Curie, Hendrik Lorentz, Albert Einstein, Paul Langevin, Charles-Eugène Guye, CTR Wilson, Owen Richardson.

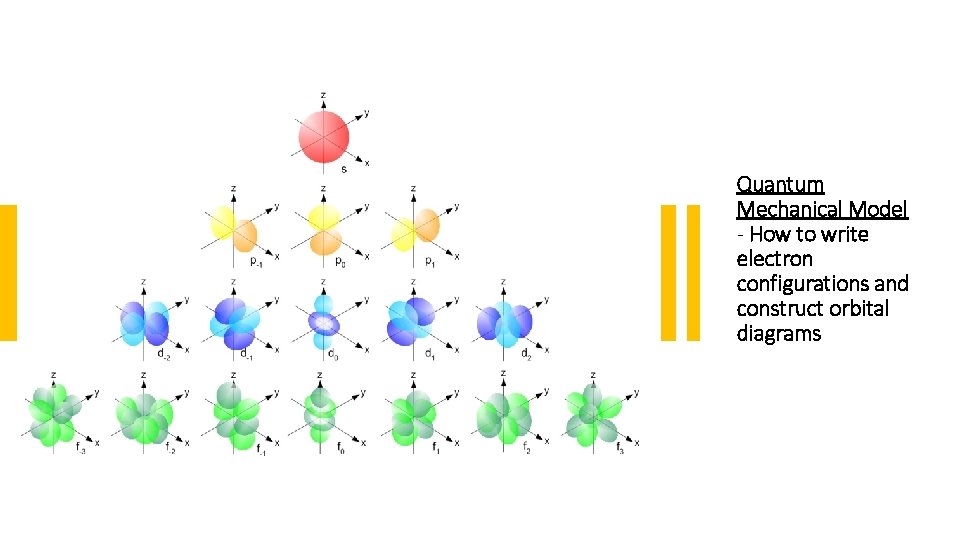
Quantum Mechanical Model - How to write electron configurations and construct orbital diagrams

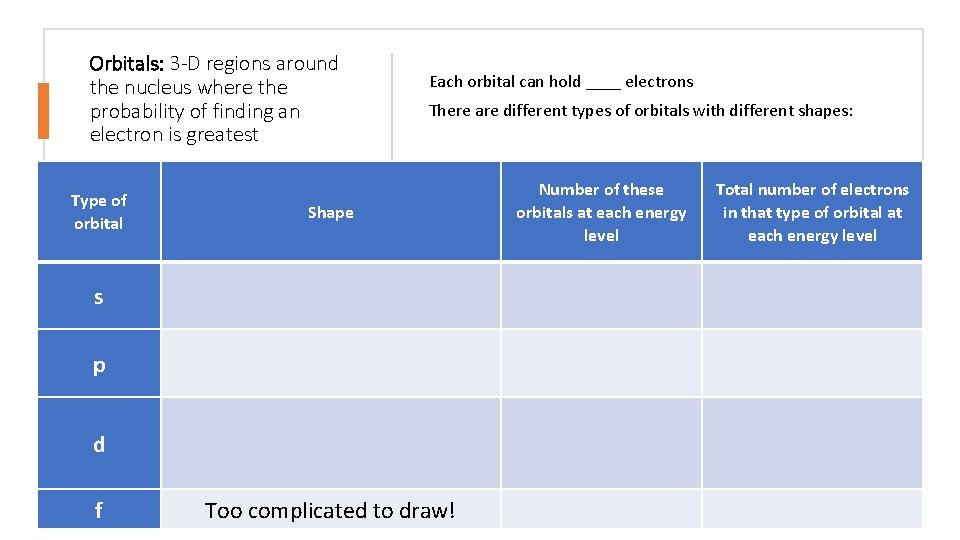
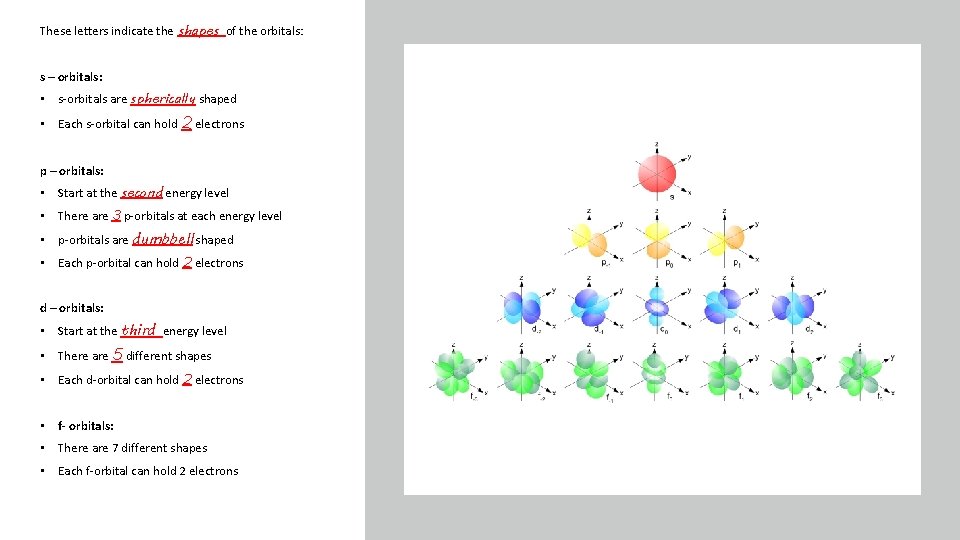
Orbitals: 3 -D regions around the nucleus where the probability of finding an electron is greatest Type of orbital Each orbital can hold ____ electrons There are different types of orbitals with different shapes: Shape s p d f Too complicated to draw! Number of these orbitals at each energy level Total number of electrons in that type of orbital at each energy level

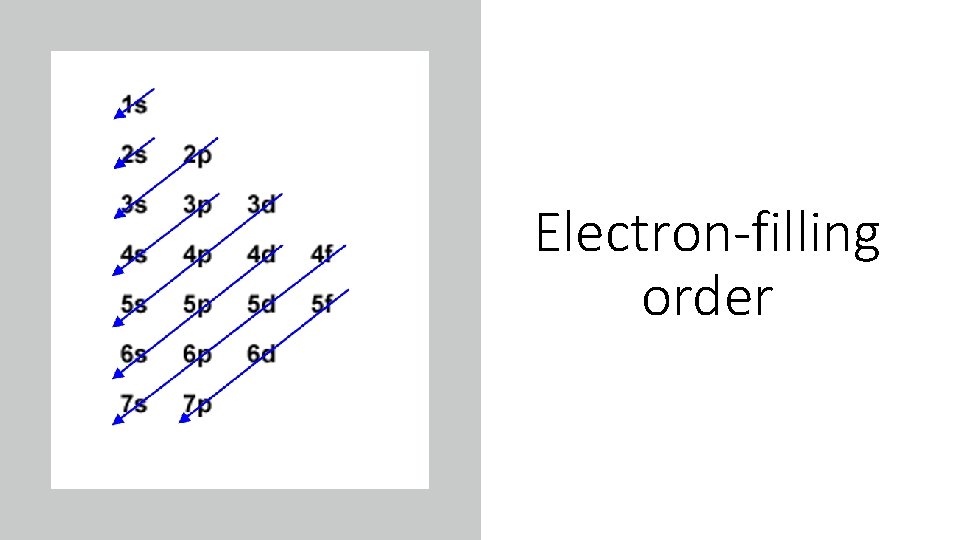
Electron-filling order

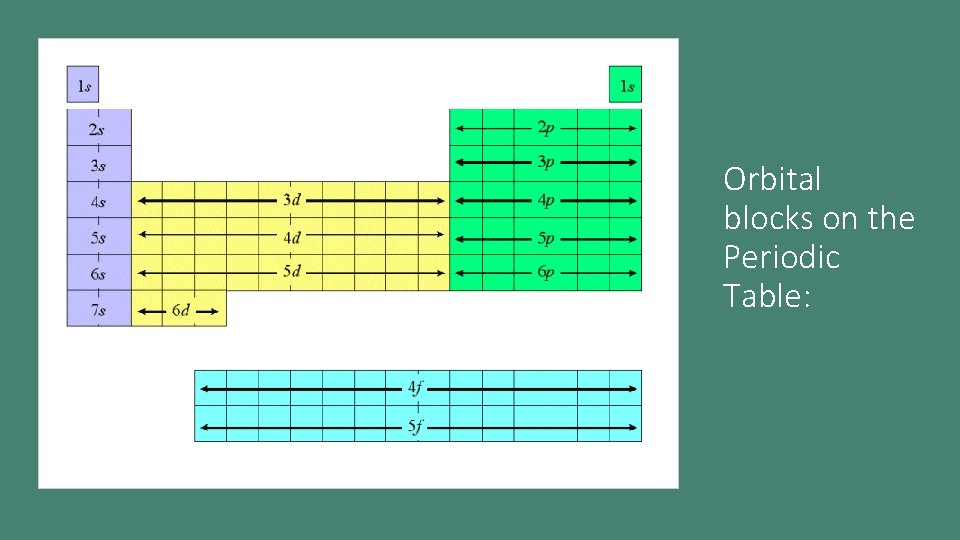
Orbital blocks on the Periodic Table:

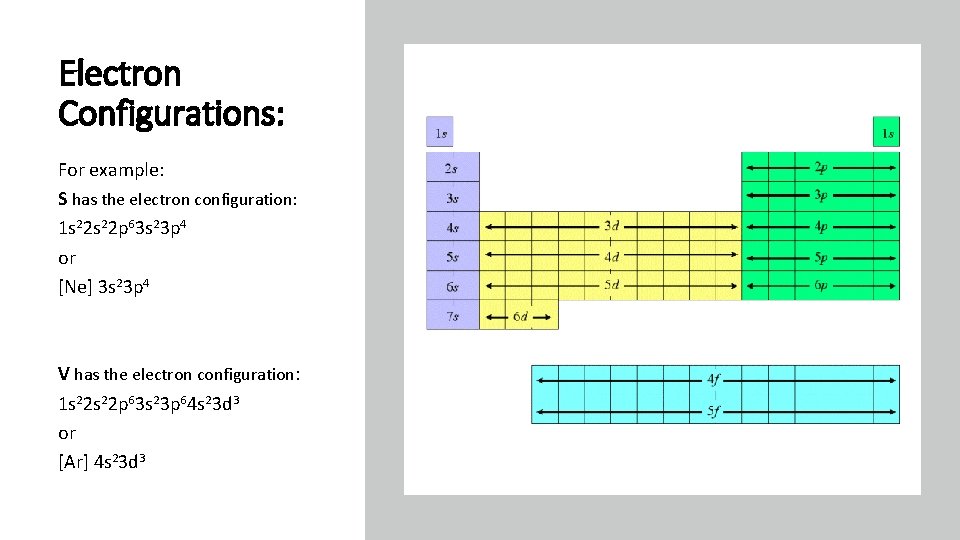
Electron Configurations: For example: S has the electron configuration: 1 s 22 p 63 s 23 p 4 or [Ne] 3 s 23 p 4 V has the electron configuration: 1 s 22 p 63 s 23 p 64 s 23 d 3 or [Ar] 4 s 23 d 3

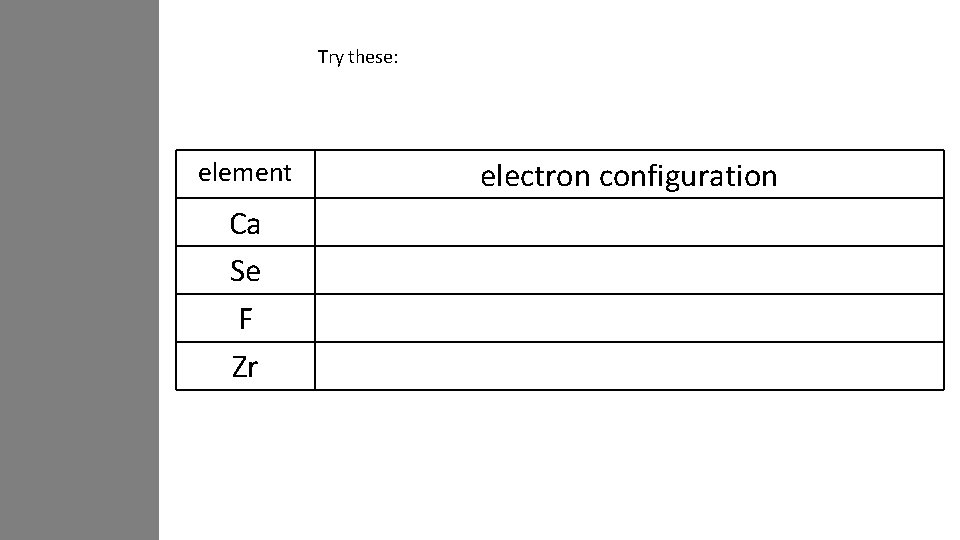
Try these: element Ca Se F Zr electron configuration

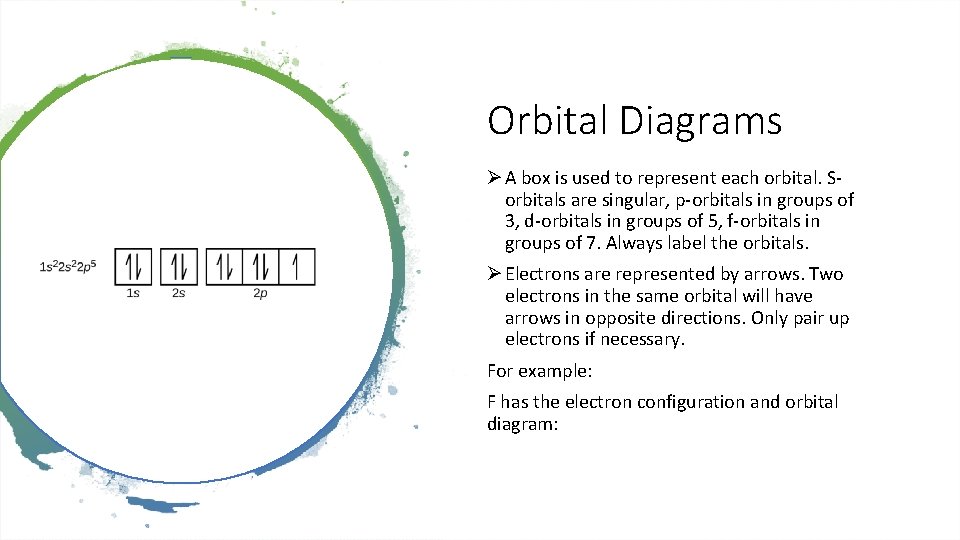
Orbital Diagrams Ø A box is used to represent each orbital. Sorbitals are singular, p-orbitals in groups of 3, d-orbitals in groups of 5, f-orbitals in groups of 7. Always label the orbitals. Ø Electrons are represented by arrows. Two electrons in the same orbital will have arrows in opposite directions. Only pair up electrons if necessary. For example: F has the electron configuration and orbital diagram:

Try these: (use can use the short form for these too) • C • P • V

Quantum Mechanical Model of the Atom Theory

What is an orbital?

An atomic orbital is a mathematical function for an electron in an atom. It is based on its wave-like behaviour. The region in which an electron is most likely to be found around a single atom in a particular energy state can be calculated from this function. The orbital shapes we will look at represent regions around the nucleus where there is a high probability of finding an electron.

Each atomic orbital is identified by three quantum numbers. They are: 1. principle quantum number, n Quantum Numbers 2. angular momentum quantum number, l 3. magnetic quantum number, ml Each electron in an orbital is also given a spin quantum number, ms.

Principle Quantum Number, n • n = 1, 2, 3… • is related to the size and energy of the orbital • as the number increases, the orbital becomes larger and the electrons spend more time further from the nucleus • maximum number of orbitals at that energy level = n 2 • maximum number of electrons at that energy level = 2 n 2

• Identifies the shape of the orbital • l = 0, 1, 2, …, n-1 Angular • when n = 1, l = 0 • when n = 2, l = 0, 1 Momentum • when n = 3, l = 0, 1, 2 (Orbital etc. Shape) • The values of l are most commonly described by letters: Quantum 1 2 3 4 5 Number, l Value of l 0 Letter

These letters indicate the shapes of the orbitals: s – orbitals: • s-orbitals are spherically shaped • Each s-orbital can hold 2 electrons p – orbitals: • Start at the second energy level • There are 3 p-orbitals at each energy level • p-orbitals are dumbbell shaped • Each p-orbital can hold 2 electrons d – orbitals: • Start at the third energy level • There are 5 different shapes • Each d-orbital can hold 2 electrons • f- orbitals: • There are 7 different shapes • Each f-orbital can hold 2 electrons

• Indicates the orbital’s spatial orientation around the nucleus • ml = - l, …, 0, …, l • when: n=1 l=0 ml = 0 Magnetic Quantum Number, ml n=2 l=0 l=1 ml = 0 ml = -1 ml = 0 ml = 1 n=3 l=0 l = 1 ml = 0 ml = -1 ml = 0 ml = 1 ml = -2 ml = -1 ml = 0 ml = 1 ml = 2

Spin Quantum Number, ms • Describes the electron spin since electrons behave as tiny magnets spinning on their axis in either clockwise or counter-clockwise directon. • ms = +½ or -½

Using Quantum Numbers

We can assign 4 quantum numbers to each of the electrons in all of the atoms on the periodic table For example: • Lithium has 3 electrons: n = 1 l = 0 ms = + ½ n = 1 l = 0 ms = - ½ n = 2 l = 0 ms = + ½ Boron: n = 1 l = 0 ms = + ½ n = 1 l = 0 ms = - ½ n = 2 l = 0 ms = + ½ n = 2 l = 0 ms = - ½ n = 2 l = 1 ml = -1 ms = + ½

Using quantum numbers works well for smaller atoms but can be extremely tedious for larger ones. For example: Mg (12 electrons. . . not even that big an atom – can you imagine mercury with 80 electrons!!!) n = 1 l = 0 ms = + ½ n = 1 l = 0 ms = - ½ n = 2 l = 0 ms = + ½ n = 2 l = 0 ms = - ½ n = 2 l = 1 ml = -1 ms = + ½ n = 2 l = 1 ml = -1 ms = - ½ n = 2 l = 1 ml = 0 ms = + ½ n = 2 l = 1 ml = 0 ms = - ½ n = 2 l = 1 ms = + ½ n = 2 l = 1 ms = - ½ n = 3 l = 0 ms = + ½ n = 3 l = 0 ms = - ½ So we generally use electron configurations and orbital diagrams instead:

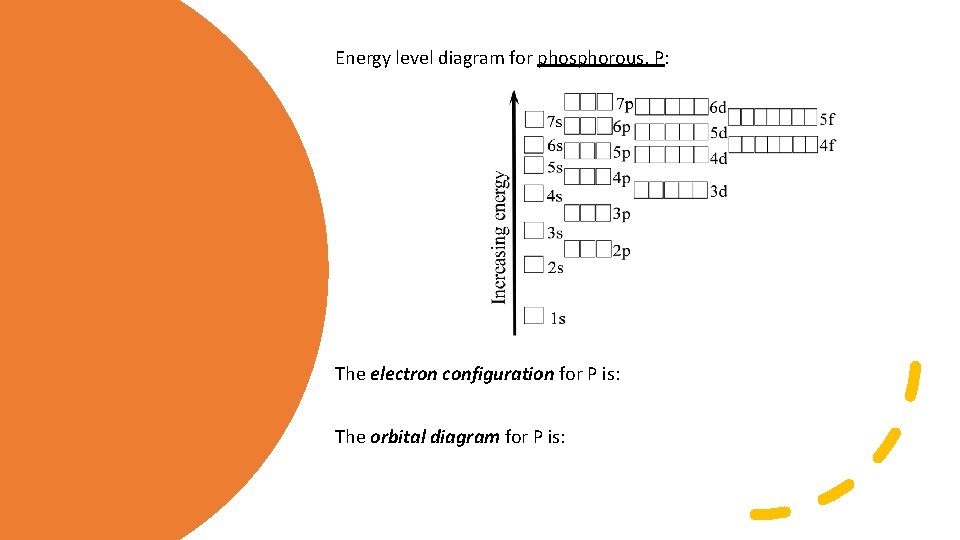
IMPORTANT Rules: • aufbau principle: Energy Level Diagrams For the ground state the orbital boxes are filled with electrons in order of increasing energy 1 s<2 s<2 p<3 s<3 p<4 s<3 d<4 p<5 s< 4 d etc • Pauli exclusion principle: An orbital can accommodate no more than two electrons and these electrons must have opposite spins. • Hund’s rule: In lowest energy electron configuration, electrons in the same energy occupy separate orbitals and have the same spin (this minimizes repulsion).

Energy level diagram for phosphorous, P: The electron configuration for P is: The orbital diagram for P is:

*** half-filled and filled orbital shells have a lower energy*** and lower energy means they are more stable and preferred. Explanation in Periodic Trends section. Look at Chromium, Cr: • What we expect the electron configuration to be: Exceptions to Electron Configuration Rules • What the actual electron configuration is: • Why? Because this gives us ______ half-filled orbital shell.

Look at Copper, Cu: • What we expect the electron configuration to be: • What the actual electron configuration is: Exceptions to Electron Configuration Rules • Why? Because this gives us ______ half-filled and ____ full orbital shell.

Now try the practice AP MC questions on your handout.