Atomic Structure 1 Democritus 400 B C Believed

Atomic Structure 1

Democritus (400 B. C. ) • Believed that matter was composed of tiny particles called atoms • His ideas were rejected by others because he had no evidence 2

John Dalton (1800’s) Dalton’s Atomic Theory • All matter is composed of atoms • All atoms of a given element are identical • Atoms cannot be created, divided or destroyed • Different atoms combine to form compounds • In chemical reactions atoms are rearranged. 3

Thompson (1890’s) • Identified the electron through use of a cathode ray tube. • Determined that electrons carry a negative charge and have an extremely small mass • Created the Plum Pudding Model (chocolate chip cookie dough model) Electrons: charge = -1 symbol = e- 4

Plum Pudding Model Negatively charged electrons were distributed throughout a uniform positive charge 5

Rutherford (1900’s) Gold Foil Experiment • Shot alpha particles (like a laser beam) at a thin piece of gold foil • Expected the alpha particles to go through foil, but found that some particles bounced back 6
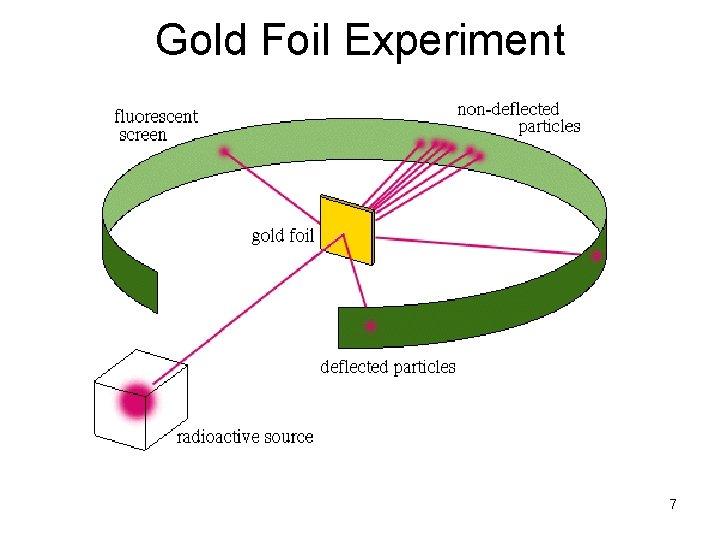
Gold Foil Experiment 7

Rutherford (cont’d) • Discovered the nucleus - tiny dense region which contains all of the atom’s positive charge and almost all its mass • Developed the Nuclear Model – nucleus in center and electrons found in empty space surrounding nucleus 8
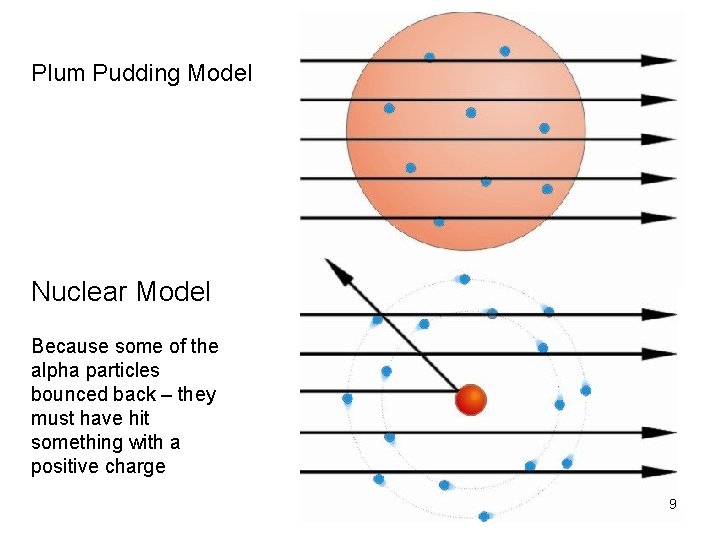
Plum Pudding Model Nuclear Model Because some of the alpha particles bounced back – they must have hit something with a positive charge 9

Inside the nucleus Protons – subatomic particle with a charge equal and opposite to an electron • Discovered by Rutherford Proton: charge = +1 symbol = p+ Neutrons – subatomic particle with a mass almost equal to that of a proton but has no charge (neutral) • Discovered by Chadwick Neutron: charge = 0 symbol = n 0 10

Bohr (1900’s) • Discovered that e- have energy of their own that keep them from being drawn into the nucleus • Believed that e- were found in paths (orbits) around the nucleus • Developed the Planetary Model 11
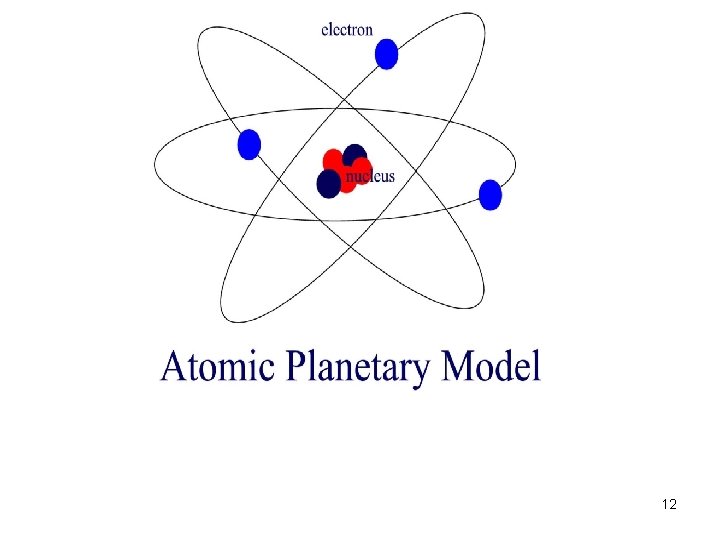
12

Schrodinger (1920’s) • Found that e- were not in orbits, but instead were found in 3 D clouds (shells) • Developed the Modern Model of the atom 13
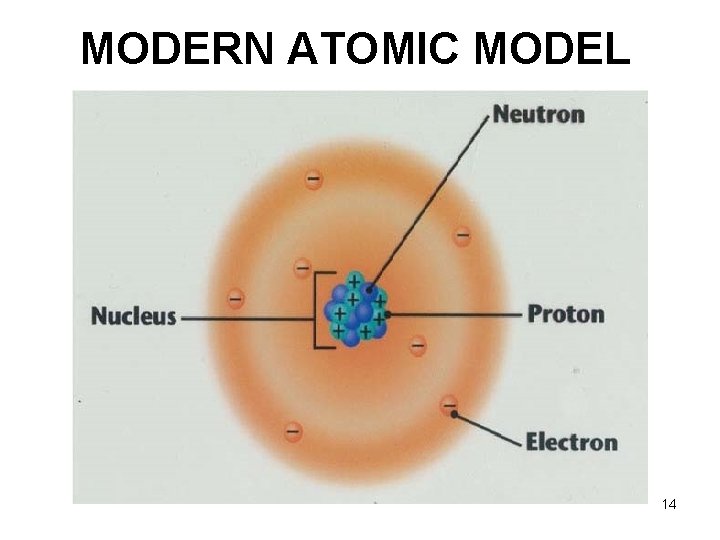
MODERN ATOMIC MODEL 14

How Atoms Differ The number of protons determine the identity of the element • Atomic Number = number of p+ • Atomic Number = number of e(in a neutral atom) • Mass Number = sum of p+ and no 15
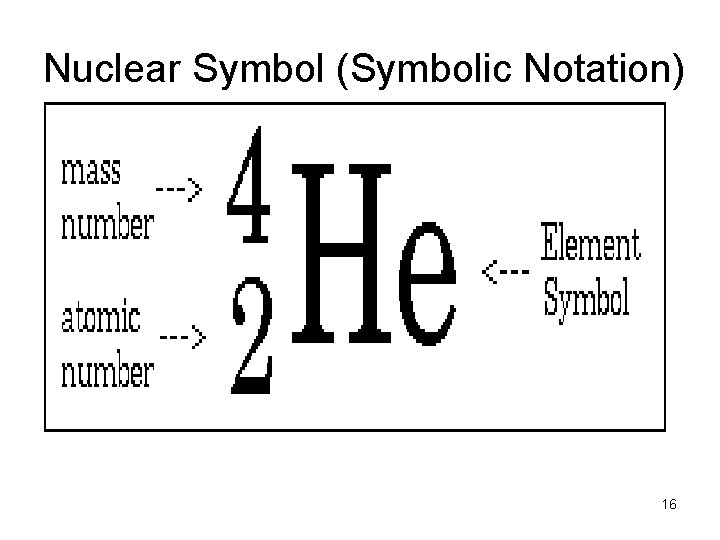
Nuclear Symbol (Symbolic Notation) 16

Examples p+ = e- = no = 17

ISOTOPES • Atoms of the same element that have the same number of protons, but a different number of neutrons. – Same atomic number, different mass number Carbon-12 Carbon-13 Carbon-14 The number after the name is the mass number 18
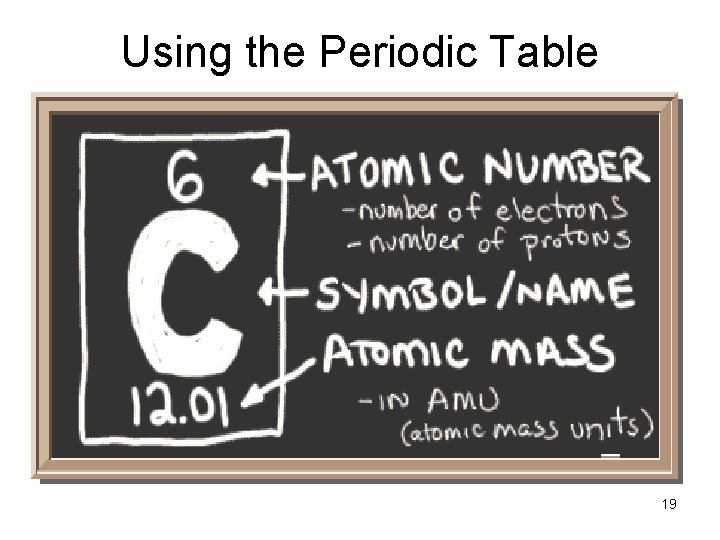
Using the Periodic Table 19

Atomic Mass • Weighted average of the masses for all isotopes of an element • If you round the atomic mass on the periodic table to the nearest whole number, you will get the mass number of the most abundant (common) isotope 20

Calculating Atomic Mass • Example 21
- Slides: 21