ATOMIC STABILITY NUCLEAR CHEMISTRY ELECTRONIC STABILITY Stability within

ATOMIC STABILITY & NUCLEAR CHEMISTRY
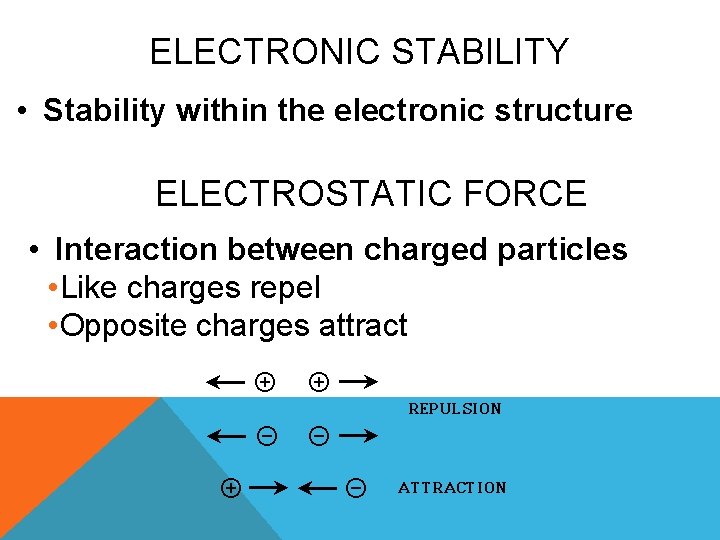
ELECTRONIC STABILITY • Stability within the electronic structure ELECTROSTATIC FORCE • Interaction between charged particles • Like charges repel • Opposite charges attract

NUCLEAR STABILITY • Stability within the nucleus • Factors: 1. Neutron to proton ratio 2. Number of nucleons
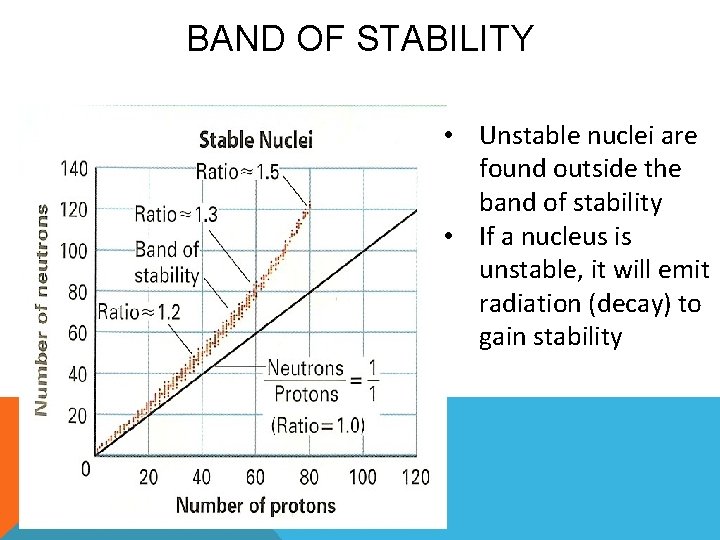
BAND OF STABILITY • Unstable nuclei are found outside the band of stability • If a nucleus is unstable, it will emit radiation (decay) to gain stability

STRONG NUCLEAR FORCE • Attractive force that acts between all nuclear particles that are extremely close together • Keeps the nucleus together • Much stronger than electrostatic force

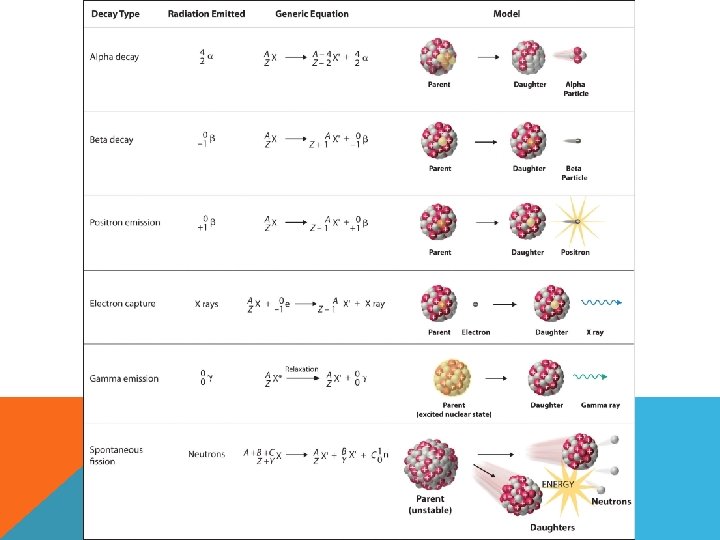
NUCLEAR CHEMISTRY • Study of reactions involving changes in atomic nuclei NUCLEAR REACTIONS • Change in the identity or characteristics of an atomic nucleus • Radioactive decay—spontaneous emission of particles and/or electromagnetic radiation • Nuclear transmutation—way of converting one element/isotope into another element/isotope by bombarding it with an energetic particle (**can result in
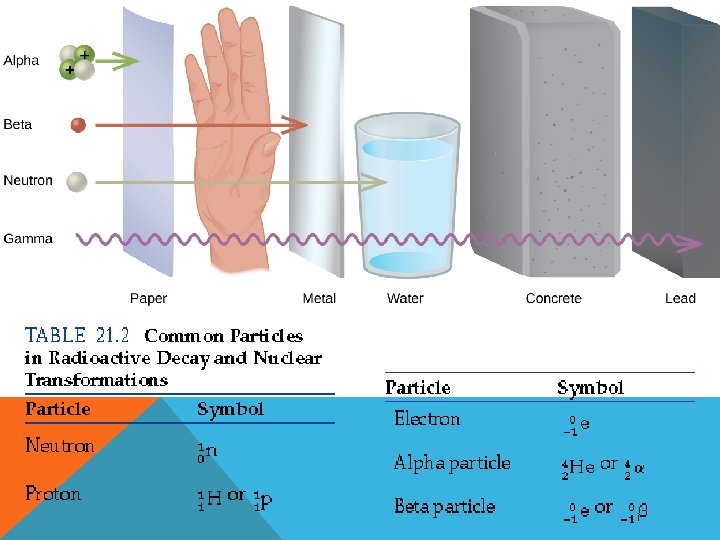
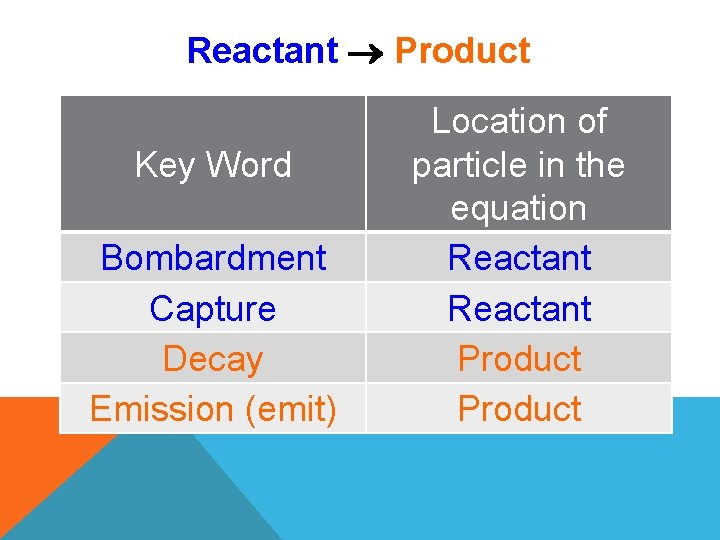
Reactant Product Key Word Bombardment Capture Decay Emission (emit) Location of particle in the equation Reactant Product

RADIOISOTOPE • Isotopes of atoms with unstable nuclei that go through radioactive decay to obtain a more stable nuclei • All elements with an atomic number greater than 83 are radioactive • Transuranium elements—were produced through induced transmutation

CHEMICAL REACTION • Atoms are rearranged by the breaking and forming of chem. bonds • Only e— are involved • Rxns are accompanied by absorption or release of relatively sm. NUCLEAR REACTION • Elements (or isotopes) are converted from one to another • p+, n 0, e—, or other elementary particles may be involved • Rxns are accompanied by absorption or

NUCLEAR EQUATIONS • Symbolic representation of nuclear reactions FUSION • Combining of 2 light atomic nuclei to produce a heavier more stable nucleus • Releases large amounts of energy FISSION • Process where the nucleus of a heavier atom splits into smaller parts (lighter nuclei) • Releases energy
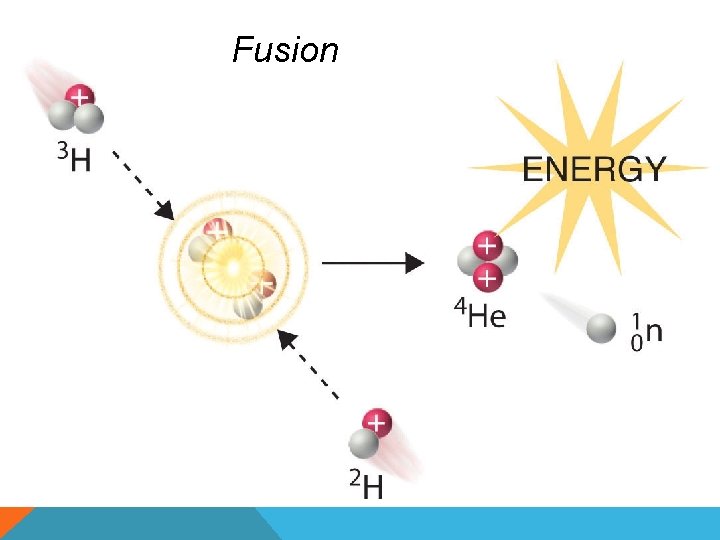
Fusion
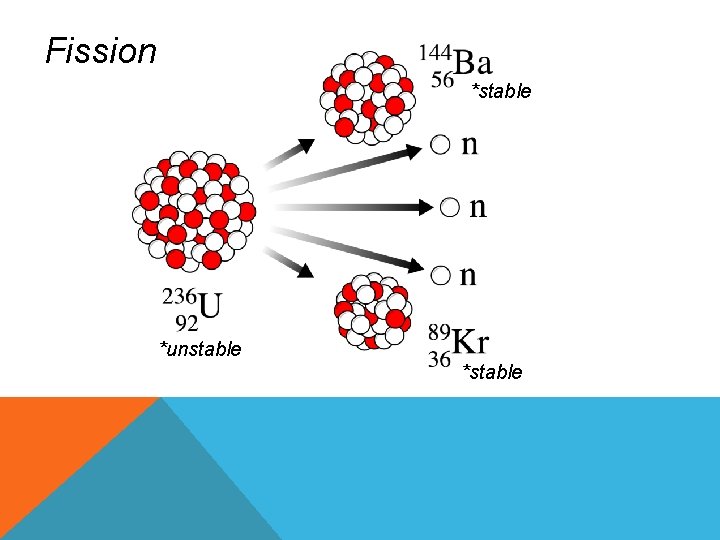
Fission *stable *unstable *stable

1. 97 40 218 84 2. Zr Po 3. ? 4. 47 20 244 96 5. 222 86 0 -1 e + ? 4 2 He + ? 4 2 Rn + Ca -10 Cm 4 2 He e + ? He + ?
- Slides: 15