Atomic Properties The Periodic Table Packet 12 ATOMIC

Atomic Properties & The Periodic Table Packet #12

ATOMIC SIZE

Atomic Size I • The sizes of atoms vary. • Atomic size increases as one goes left & down the periodic table. • Atomic size decreases as one goes from right & up on the periodic table.

Atom/Radii Size II • • • As one goes from left to right on the periodic table, the number of protons increase as well as the number of electrons. However, the size of the orbitals, in which the electrons are placed, do not increase in size. This results in the protons drawing the electrons closer to the nucleus—hence resulting in a smaller atom/radii size.

IONIZATION ENERGY/ENERGIES
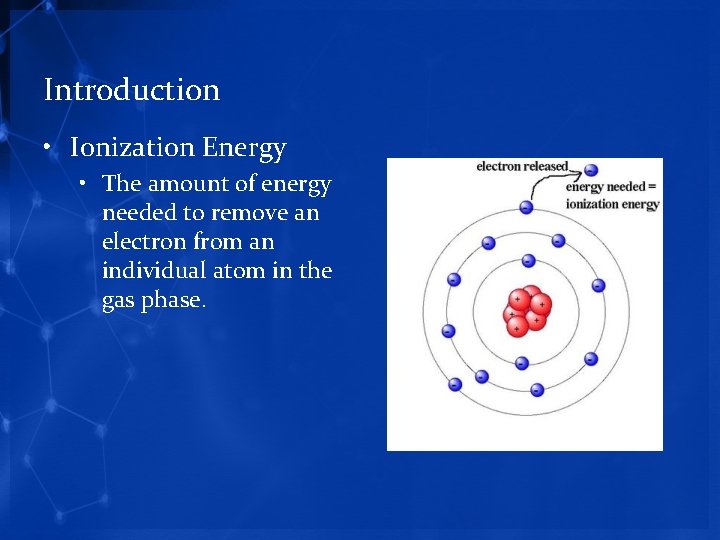
Introduction • Ionization Energy • The amount of energy needed to remove an electron from an individual atom in the gas phase.
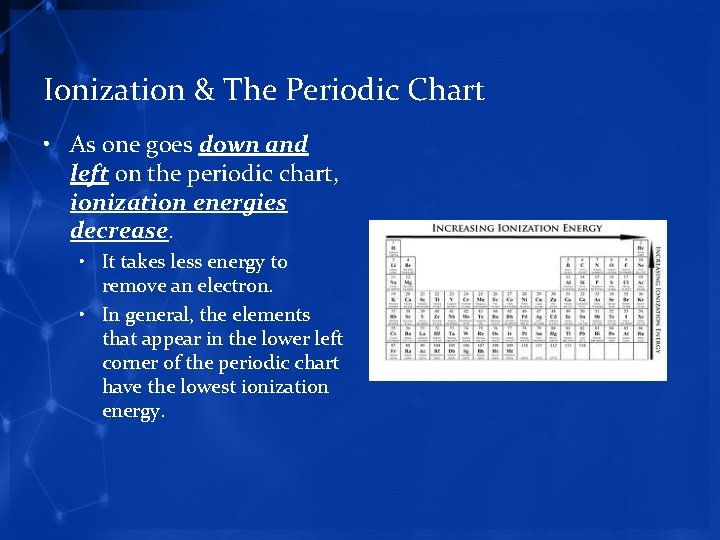
Ionization & The Periodic Chart • As one goes down and left on the periodic chart, ionization energies decrease. • It takes less energy to remove an electron. • In general, the elements that appear in the lower left corner of the periodic chart have the lowest ionization energy.

Special Note • A relatively small amount of energy is necessary to remove an electron from a metal. • The most characteristic chemical property of a metal is losing electrons to nonmetals.

ELECTRONEGATIVITY

Introduction • Electrons, of atoms such as oxygen and nitrogen, surrounding the nucleus of an atom, do not evenly distribute themselves evenly but tend to collect in one position. • As a result, the region where electrons are “collected” will be more negative than the rest of the atom. • Atoms with this uneven distribution of charge is said to be polarized. • This allows atoms to be considered electronegative. • In general, as one goes up and to the right on the periodic chart electronegativities increase. 2/15/2022 5: 32: 52 PM 10

Pauling Electronegativity Scale

MELTING POINTS & BOILING POINTS
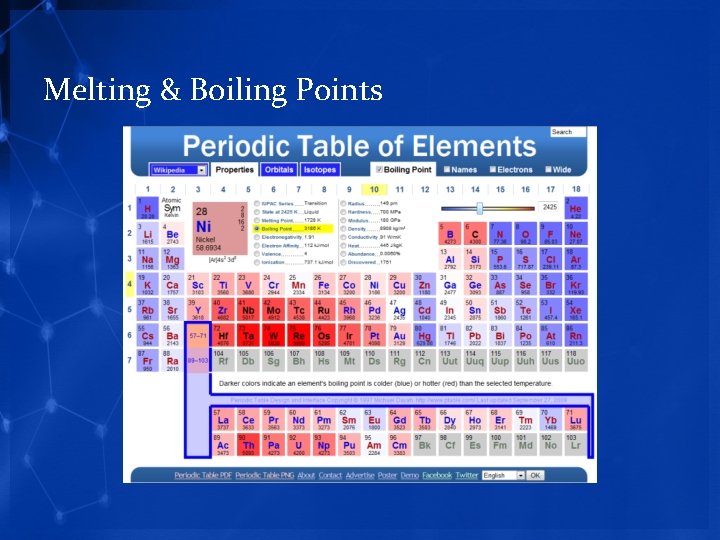
Melting & Boiling Points

REVIEW
- Slides: 14