Atomic Orbitals sorbitals porbitals dorbitals Chemical Bonding Overlap

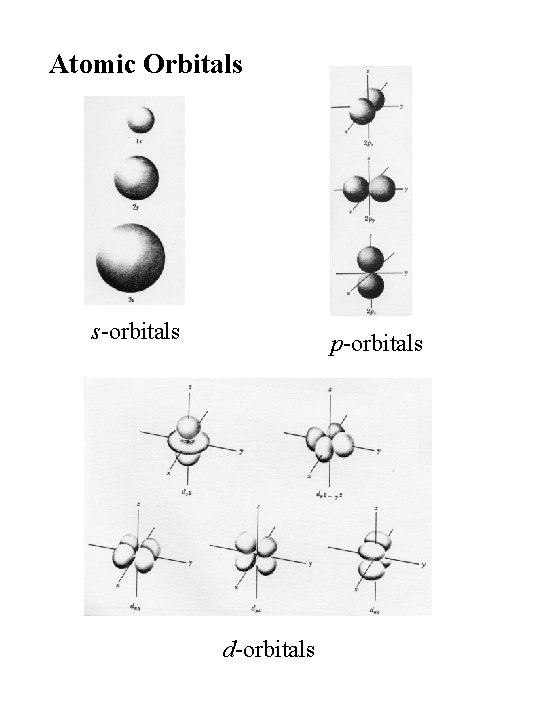
Atomic Orbitals s-orbitals p-orbitals d-orbitals
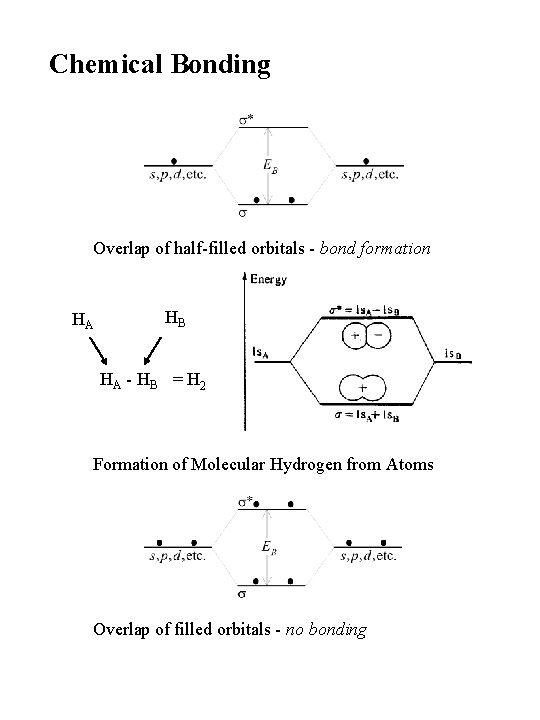
Chemical Bonding Overlap of half-filled orbitals - bond formation HA HB HA - HB = H 2 Formation of Molecular Hydrogen from Atoms Overlap of filled orbitals - no bonding
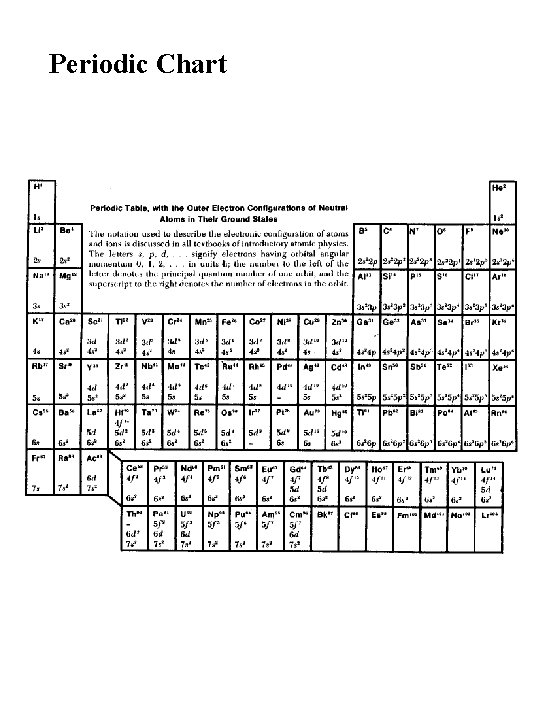
Periodic Chart
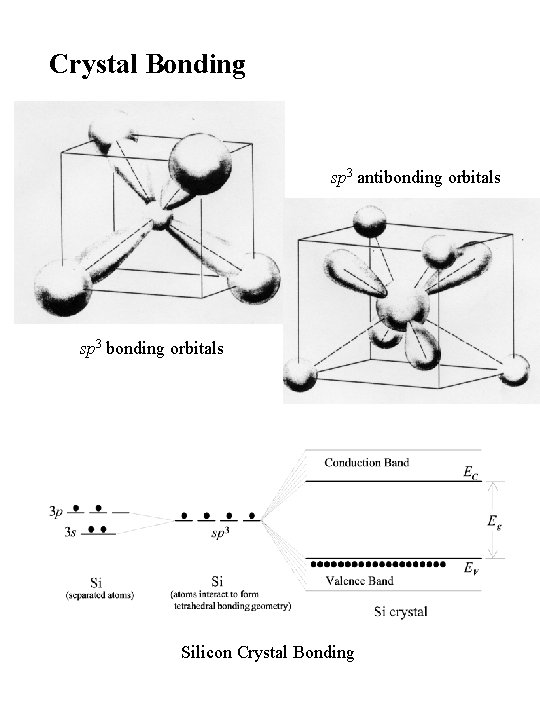
Crystal Bonding sp 3 antibonding orbitals sp 3 bonding orbitals Silicon Crystal Bonding
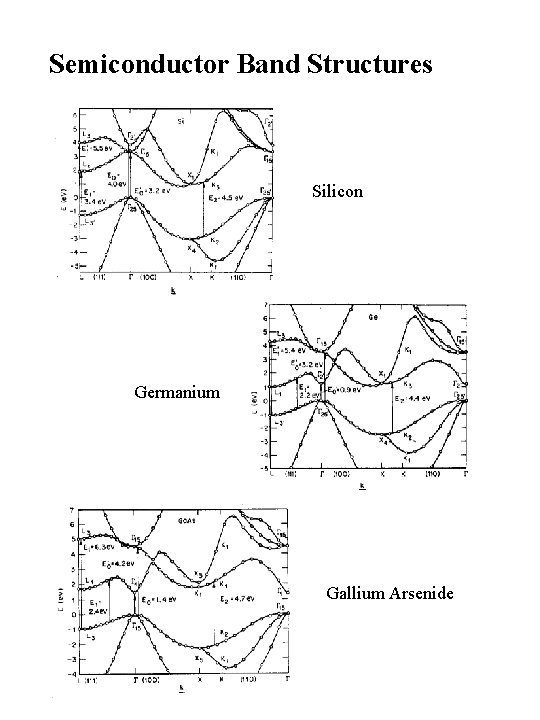
Semiconductor Band Structures Silicon Germanium Gallium Arsenide
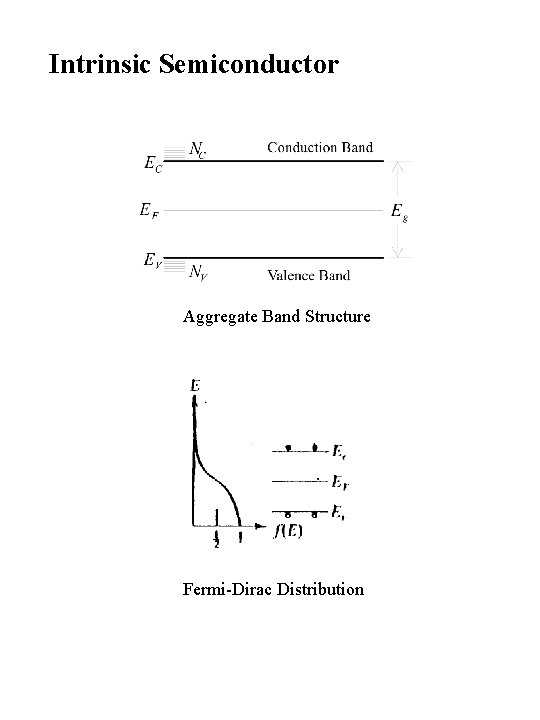
Intrinsic Semiconductor Aggregate Band Structure Fermi-Dirac Distribution
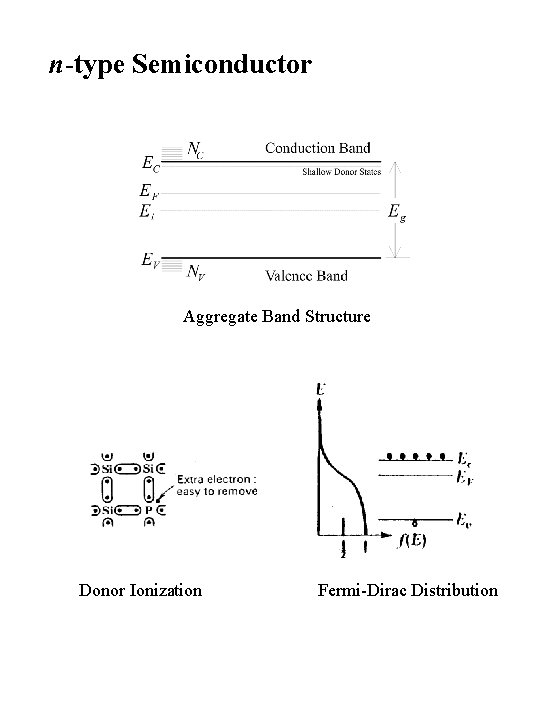
n-type Semiconductor Aggregate Band Structure Donor Ionization Fermi-Dirac Distribution
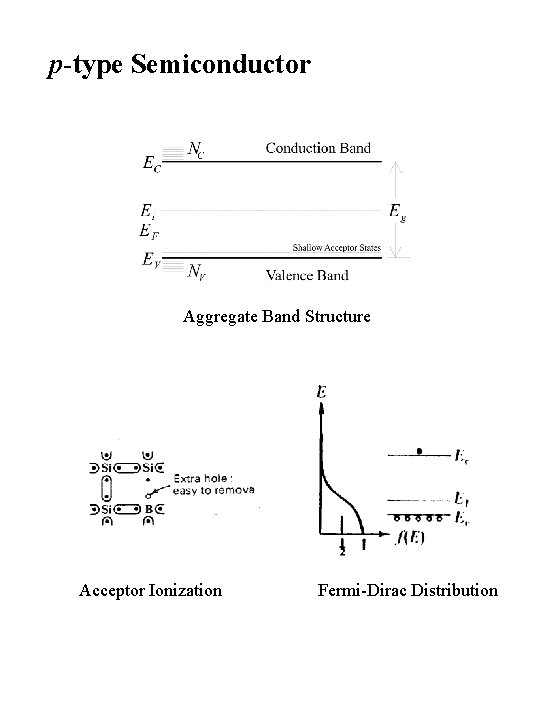
p-type Semiconductor Aggregate Band Structure Acceptor Ionization Fermi-Dirac Distribution
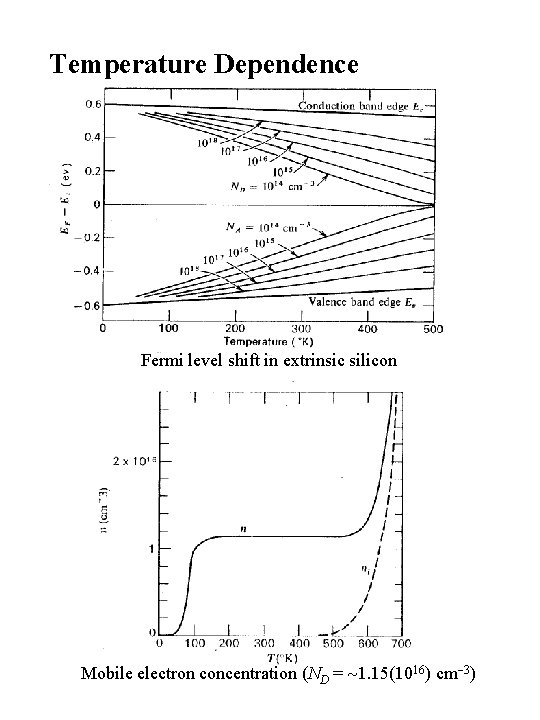
Temperature Dependence Fermi level shift in extrinsic silicon Mobile electron concentration (ND = 1. 15(1016) cm 3)
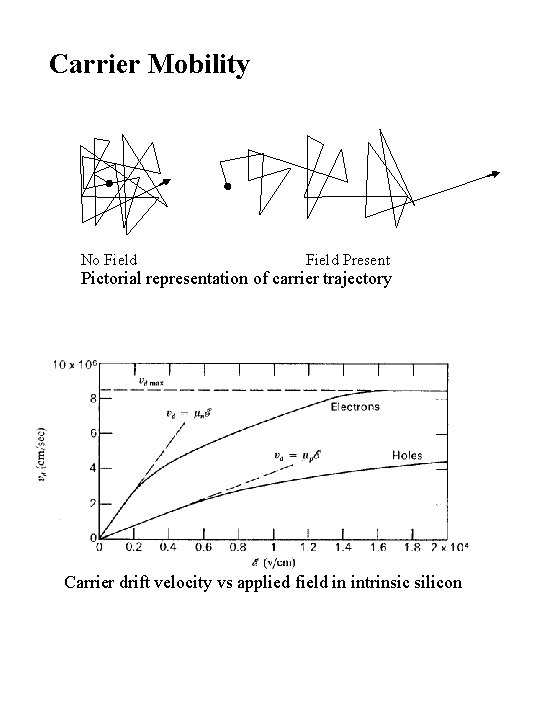
Carrier Mobility No Field Present Pictorial representation of carrier trajectory Carrier drift velocity vs applied field in intrinsic silicon
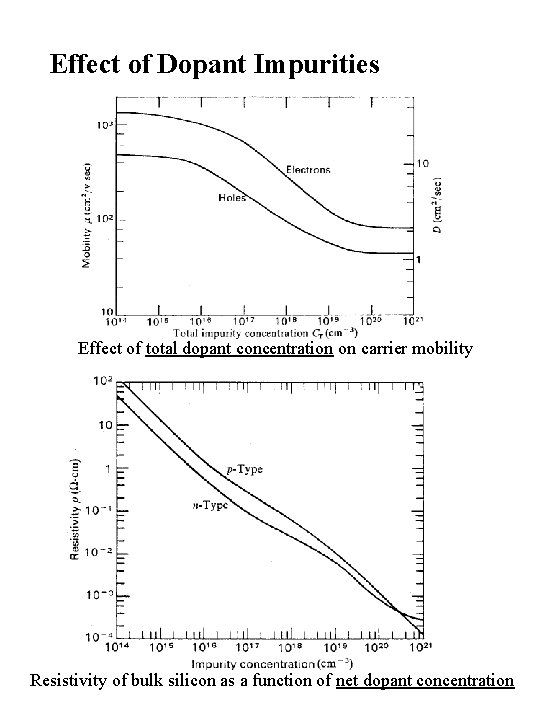
Effect of Dopant Impurities Effect of total dopant concentration on carrier mobility Resistivity of bulk silicon as a function of net dopant concentration

The Seven Crystal Systems
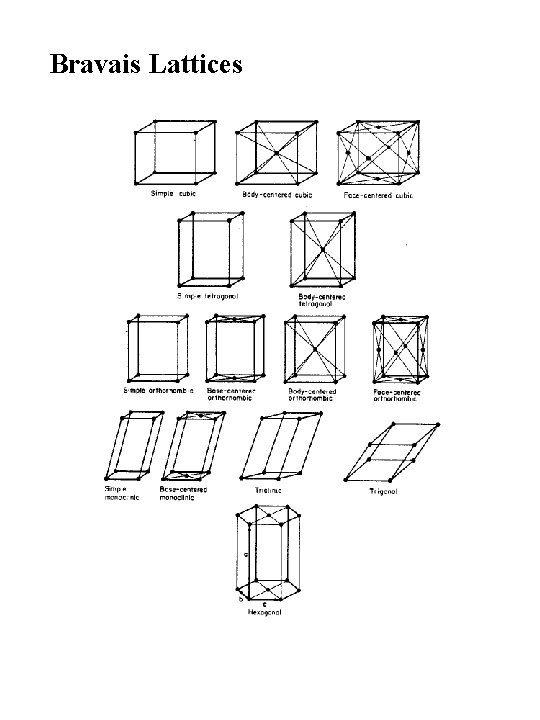
Bravais Lattices
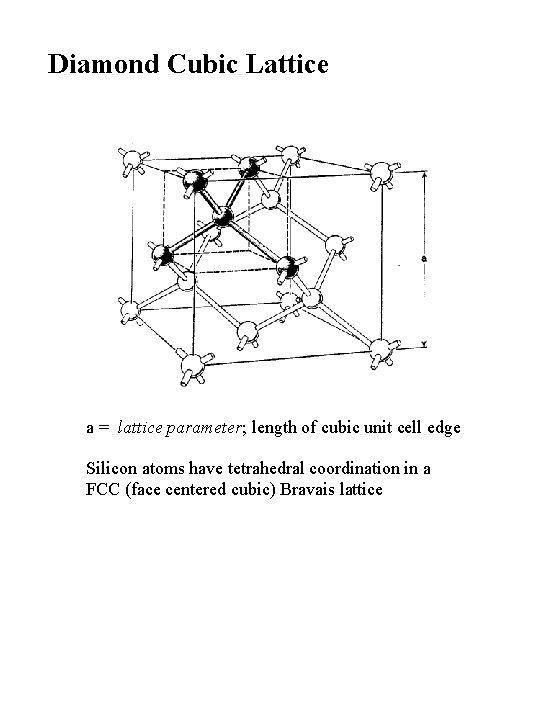
Diamond Cubic Lattice a = lattice parameter; length of cubic unit cell edge Silicon atoms have tetrahedral coordination in a FCC (face centered cubic) Bravais lattice
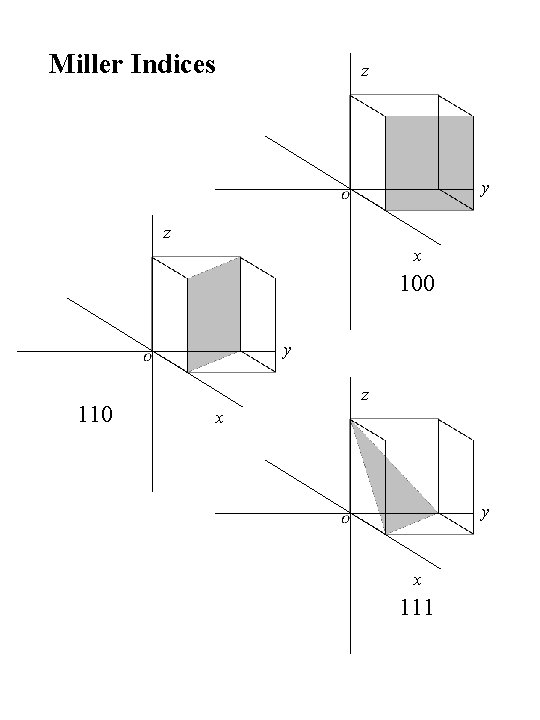
Miller Indices z y z x 100 y 110 z x y x 111
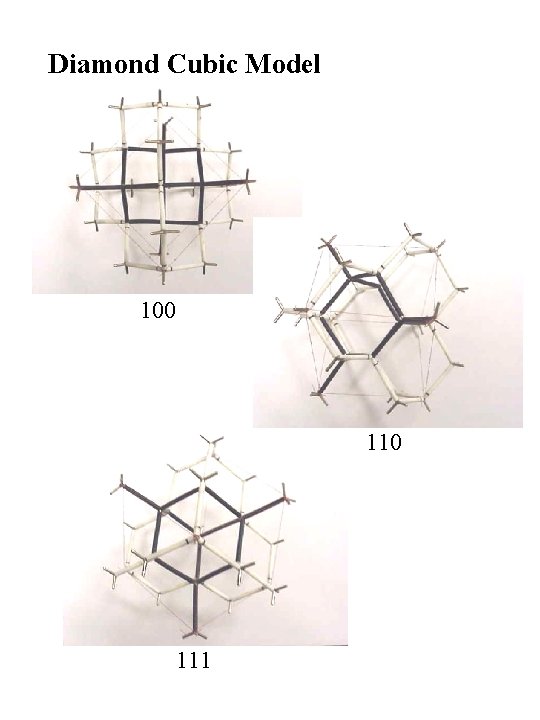
Diamond Cubic Model 100 111
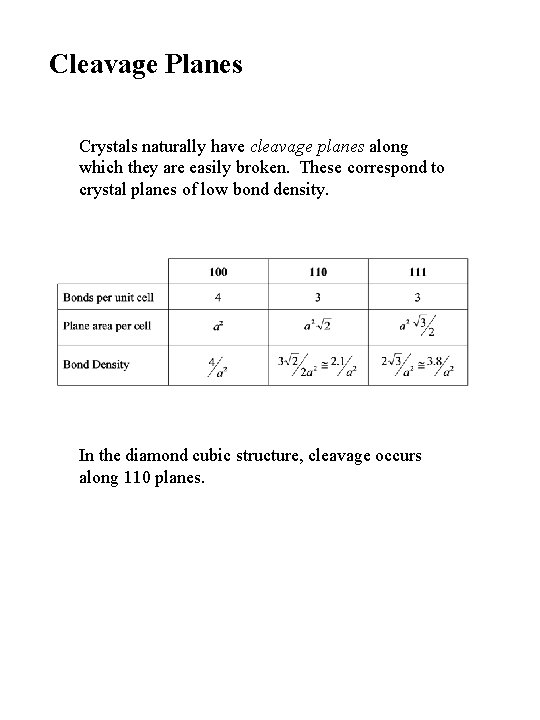
Cleavage Planes Crystals naturally have cleavage planes along which they are easily broken. These correspond to crystal planes of low bond density. In the diamond cubic structure, cleavage occurs along 110 planes.
![[100] Orientation [100] Orientation](http://slidetodoc.com/presentation_image_h2/bce8d3e0e892fa88229cd2936227634f/image-19.jpg)
[100] Orientation
![[110] Orientation [110] Orientation](http://slidetodoc.com/presentation_image_h2/bce8d3e0e892fa88229cd2936227634f/image-20.jpg)
[110] Orientation
![[111] Orientation [111] Orientation](http://slidetodoc.com/presentation_image_h2/bce8d3e0e892fa88229cd2936227634f/image-21.jpg)
[111] Orientation
![[100] Cleavage [100] Cleavage](http://slidetodoc.com/presentation_image_h2/bce8d3e0e892fa88229cd2936227634f/image-22.jpg)
[100] Cleavage
![[111] Cleavage [111] Cleavage](http://slidetodoc.com/presentation_image_h2/bce8d3e0e892fa88229cd2936227634f/image-23.jpg)
[111] Cleavage
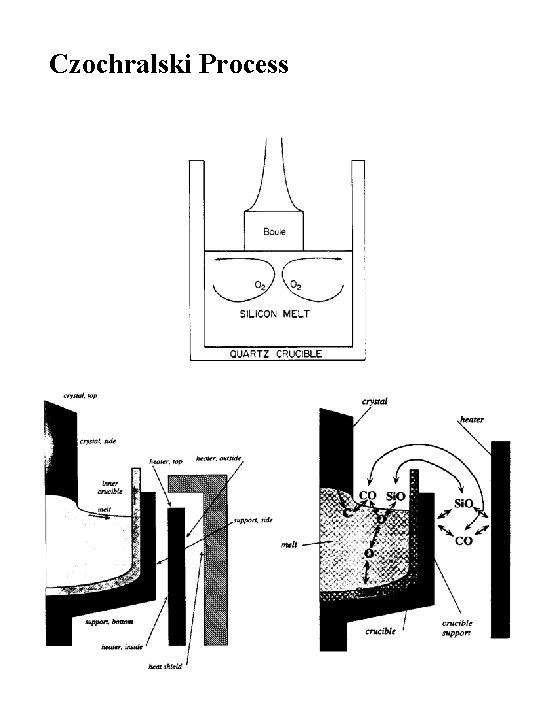
Czochralski Process

Seed Rod (Single Crystal Si) dia. = ~1 cm

Czochralski Process Equipment Image courtesy Microchemicals

Czochralski Factory and Boules

CZ Growth under Rapid Stirring Distribution Coefficients CZ Dopant Profiles under Conditions of Rapid Stirring
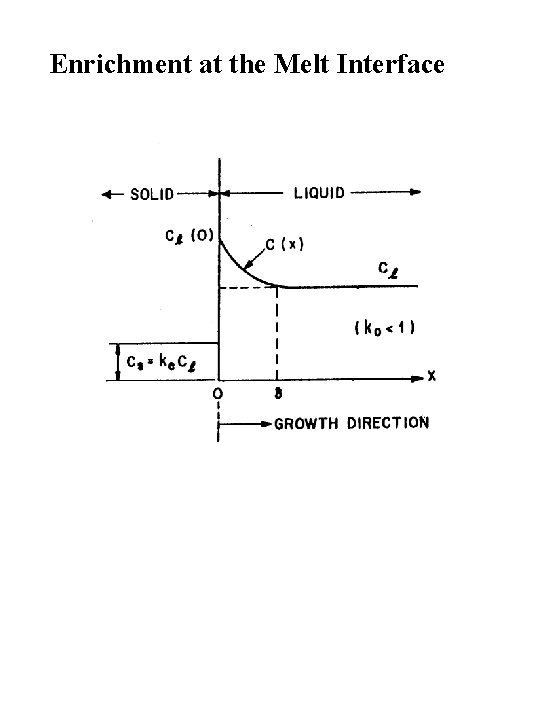
Enrichment at the Melt Interface

Zone Refining Si Ingot Heater Ingot slowly passes through the needle’s eye heater so that the molten zone is “swept” through the ingot from one end to the other
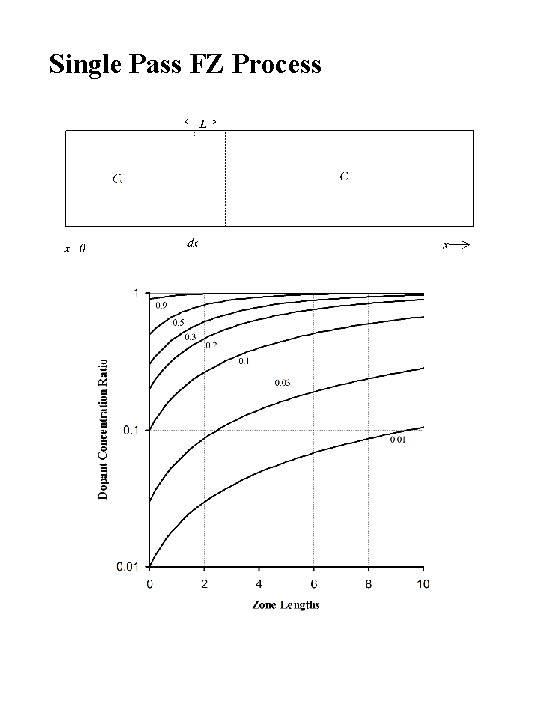
Single Pass FZ Process
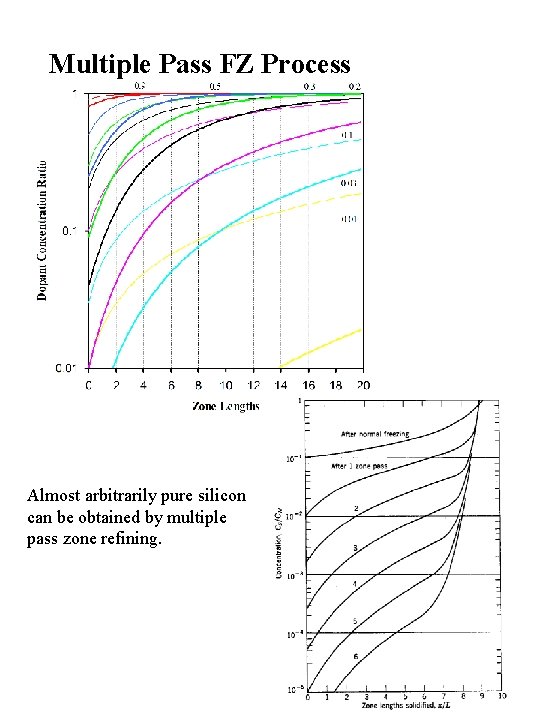
Multiple Pass FZ Process Almost arbitrarily pure silicon can be obtained by multiple pass zone refining.
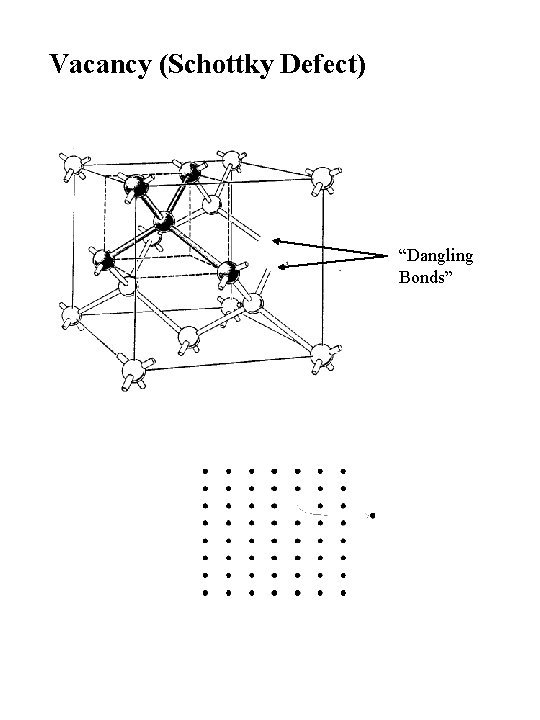
Vacancy (Schottky Defect) “Dangling Bonds”
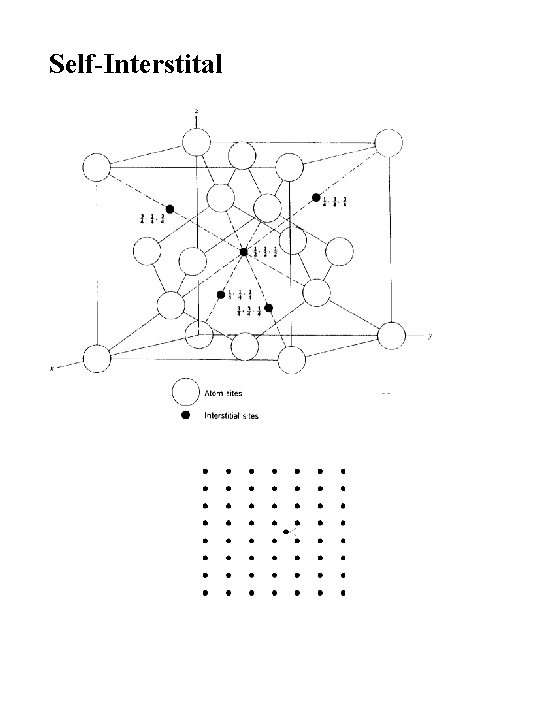
Self-Interstital
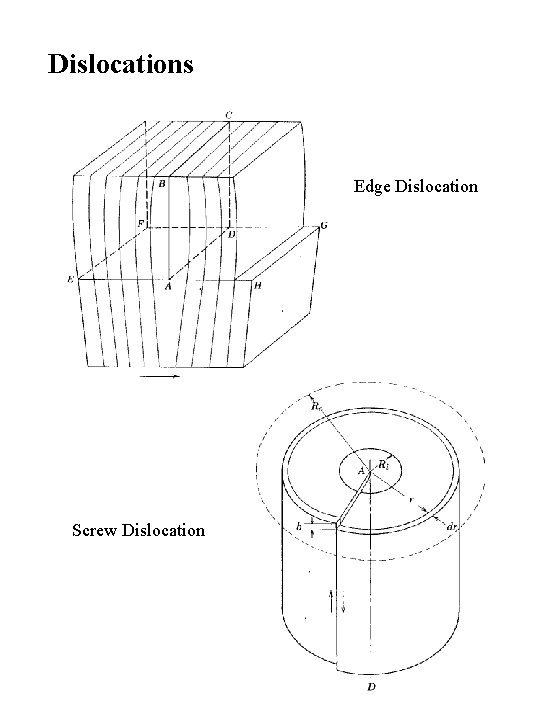
Dislocations Edge Dislocation Screw Dislocation
![Burgers Vector Edge Dislocation Screw Dislocations in Silicon [100] [111] Burgers Vector Edge Dislocation Screw Dislocations in Silicon [100] [111]](http://slidetodoc.com/presentation_image_h2/bce8d3e0e892fa88229cd2936227634f/image-36.jpg)
Burgers Vector Edge Dislocation Screw Dislocations in Silicon [100] [111]

Stacking Faults Intrinsic Stacking Fault Extrinsic Stacking Fault

Vacancy-Interstitial Equilibrium Formation of a Frenkel defect - vacancy-interstitial pair “Chemical” Equilibrium

Thermodynamic Potentials E = Internal Energy H = Enthalpy (heat content) A = Helmholtz Free Energy G = Gibbs Free Energy For condensed phases: E and H are equivalent = internal energy (total system energy) A and G are equivalent = free energy (energy available for work) T = Absolute Temperature S = Entropy (disorder) Boltzmann’s relation
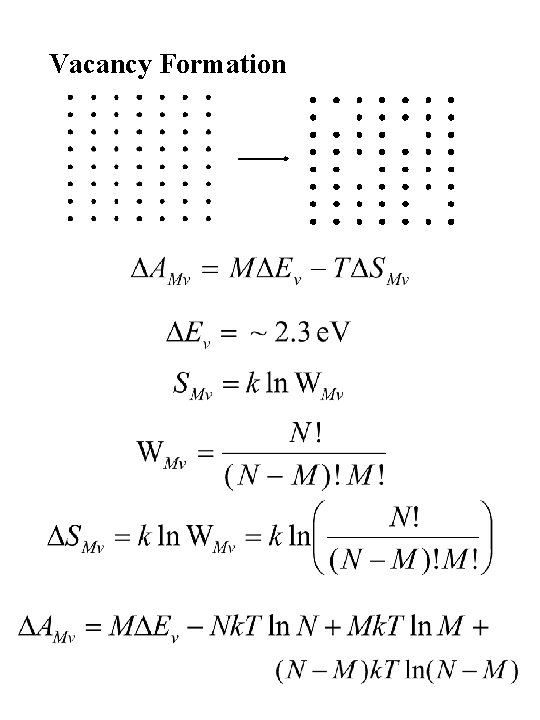
Vacancy Formation
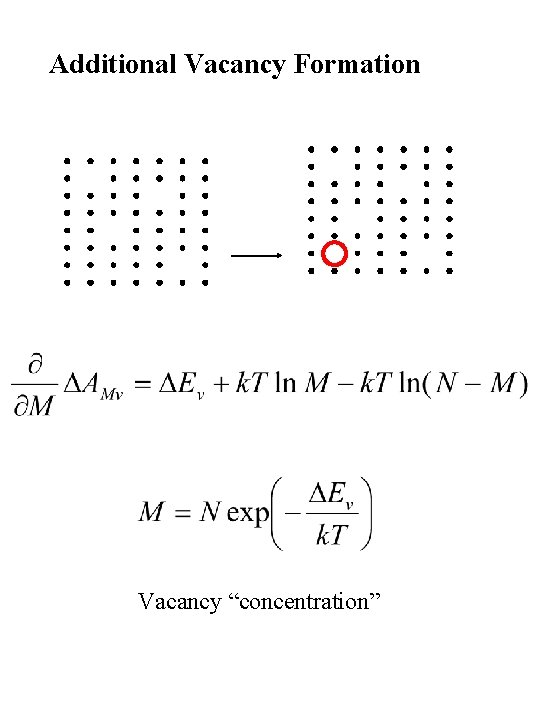
Additional Vacancy Formation Vacancy “concentration”
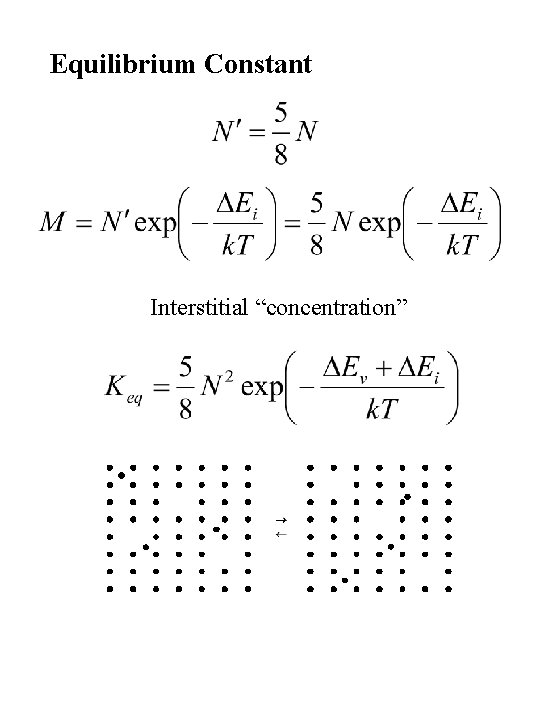
Equilibrium Constant Interstitial “concentration”
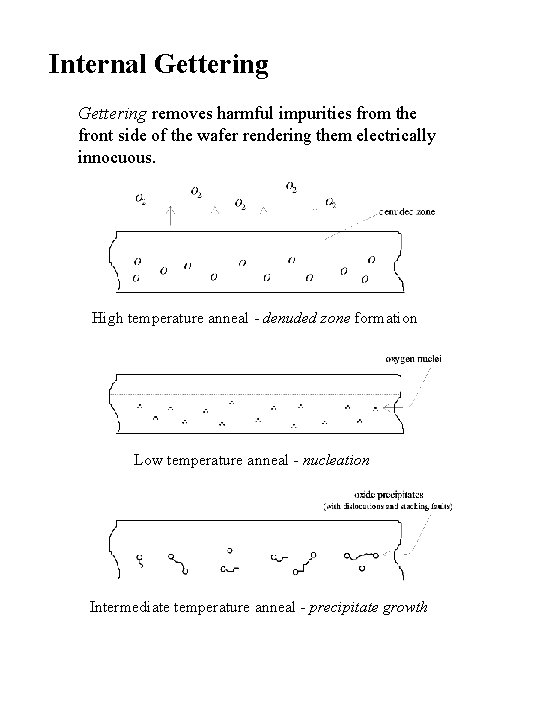
Internal Gettering removes harmful impurities from the front side of the wafer rendering them electrically innocuous. High temperature anneal - denuded zone formation Low temperature anneal - nucleation Intermediate temperature anneal - precipitate growth

Oxygen Solubility in Silicon
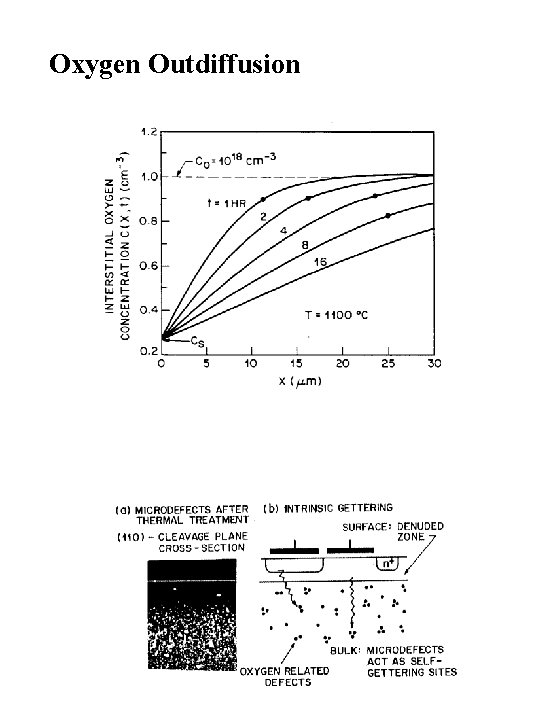
Oxygen Outdiffusion
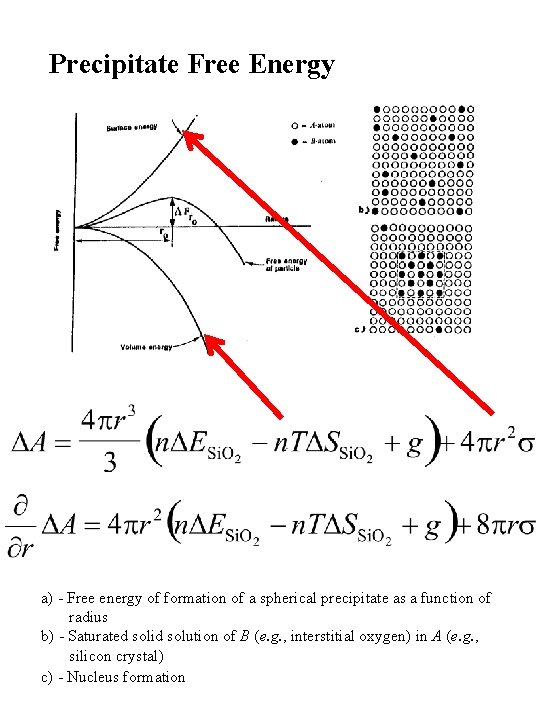
Precipitate Free Energy a) - Free energy of formation of a spherical precipitate as a function of radius b) - Saturated solid solution of B (e. g. , interstitial oxygen) in A (e. g. , silicon crystal) c) - Nucleus formation
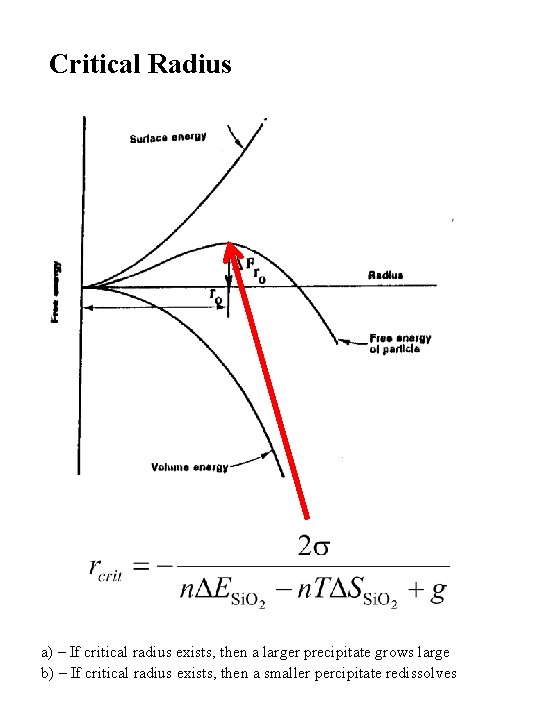
Critical Radius a) – If critical radius exists, then a larger precipitate grows large b) – If critical radius exists, then a smaller percipitate redissolves
![Substrate Characterization by XRD Bragg pattern - [hk 0], [h 0 l], or [0 Substrate Characterization by XRD Bragg pattern - [hk 0], [h 0 l], or [0](http://slidetodoc.com/presentation_image_h2/bce8d3e0e892fa88229cd2936227634f/image-48.jpg)
Substrate Characterization by XRD Bragg pattern - [hk 0], [h 0 l], or [0 kl]
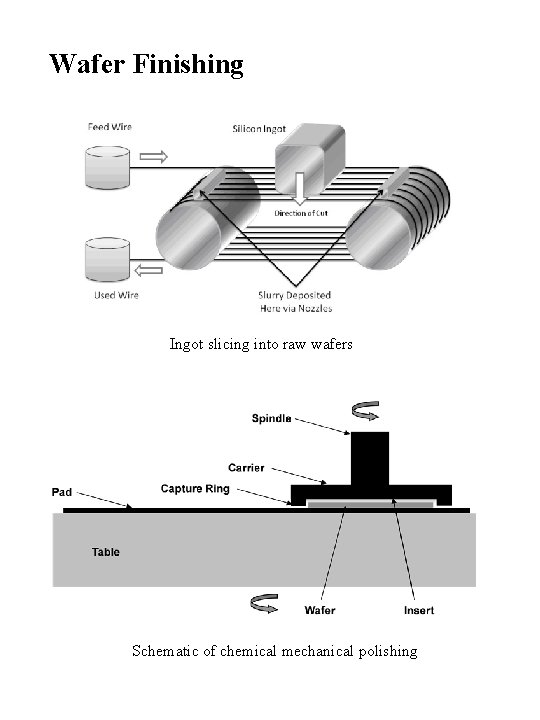
Wafer Finishing Ingot slicing into raw wafers Schematic of chemical mechanical polishing
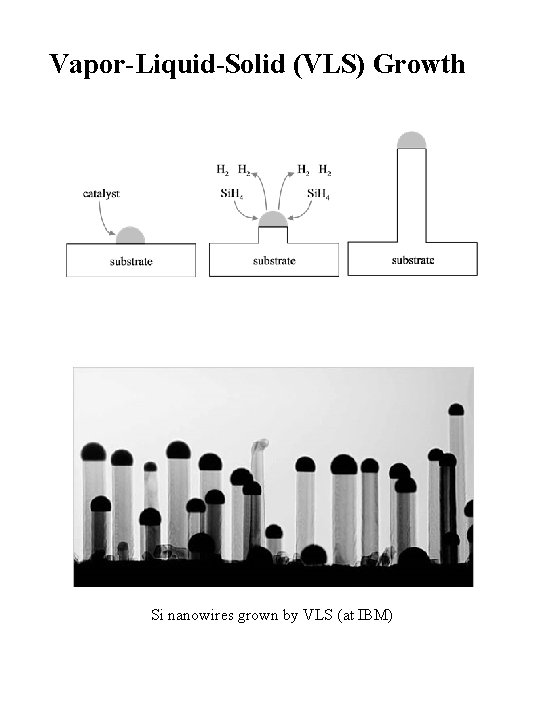
Vapor-Liquid-Solid (VLS) Growth Si nanowires grown by VLS (at IBM)
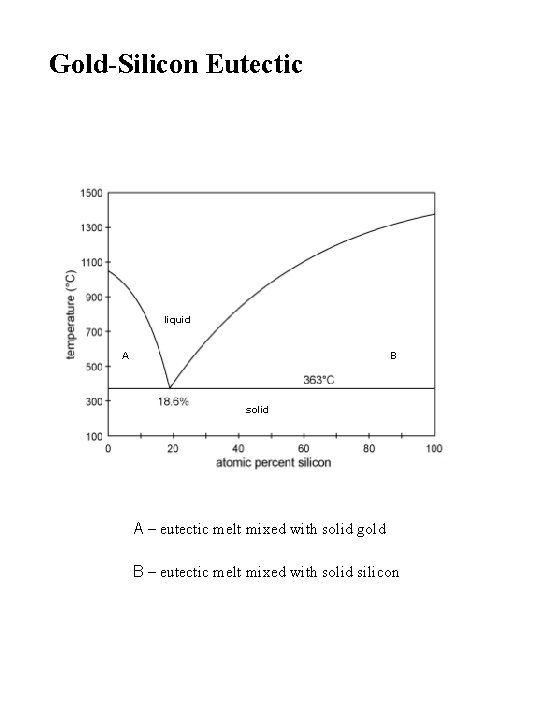
Gold-Silicon Eutectic liquid A B solid A – eutectic melt mixed with solid gold B – eutectic melt mixed with solid silicon
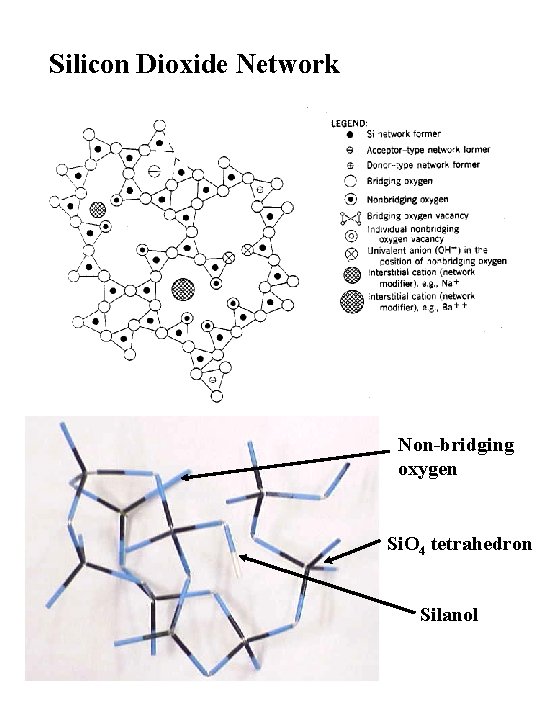
Silicon Dioxide Network Non-bridging oxygen Si. O 4 tetrahedron Silanol
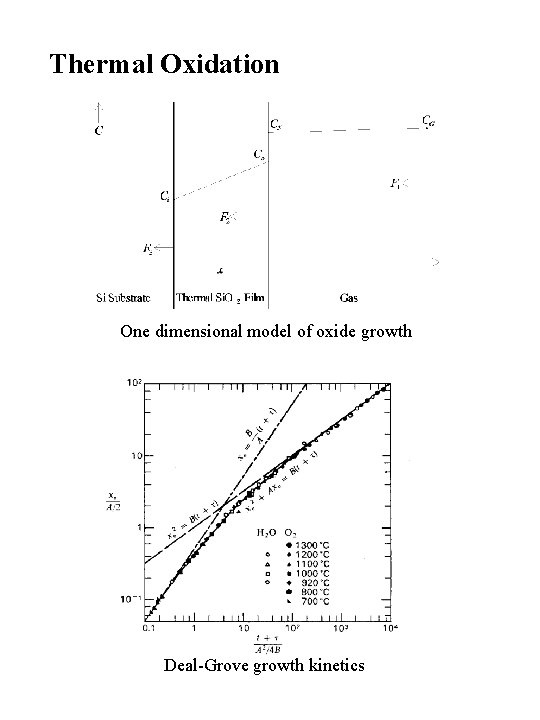
Thermal Oxidation One dimensional model of oxide growth Deal-Grove growth kinetics

Steady-state Fluxes Mass transport flux Diffusion flux Reaction “flux” 1) Diffusion flux is “in-diffusion”. Any products, e. g. , H 2, must “out-diffuse”. However, out-diffusion is fast and generally not limiting. 2) Mass transport is generally never limiting.

Henry's Law Distribution equilibrium (Henry's Law) Reaction = Mass Transport

Steady-state Concentrations Reaction = Diffusion Gas phase concentration related to reaction concentration

Deal-Grove Model Relationship between thickness and time: What if an oxide of thickenss, x 0, is already on the wafer? Must calculate equivalent growth time under desired conditions

Deal-Grove Rate Constants B/A => Linear Rate Constant B => Parabolic Rate Constant
![Oxidation Kinetics Rate constants for wet and dry oxidation on [100] and [111] surfaces Oxidation Kinetics Rate constants for wet and dry oxidation on [100] and [111] surfaces](http://slidetodoc.com/presentation_image_h2/bce8d3e0e892fa88229cd2936227634f/image-59.jpg)
Oxidation Kinetics Rate constants for wet and dry oxidation on [100] and [111] surfaces
![Linear Rate Constant Orientation dependence for [100] and [111] surfaces affects only the “pre-exponential” Linear Rate Constant Orientation dependence for [100] and [111] surfaces affects only the “pre-exponential”](http://slidetodoc.com/presentation_image_h2/bce8d3e0e892fa88229cd2936227634f/image-60.jpg)
Linear Rate Constant Orientation dependence for [100] and [111] surfaces affects only the “pre-exponential” factor and not the activation energy
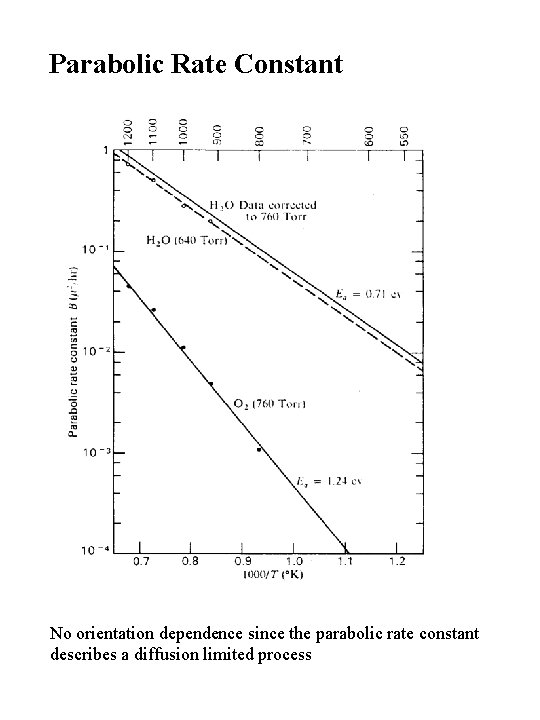
Parabolic Rate Constant No orientation dependence since the parabolic rate constant describes a diffusion limited process
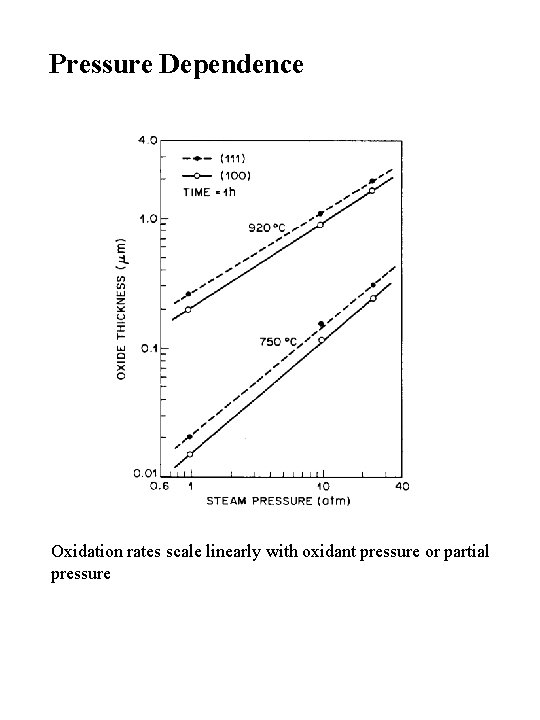
Pressure Dependence Oxidation rates scale linearly with oxidant pressure or partial pressure
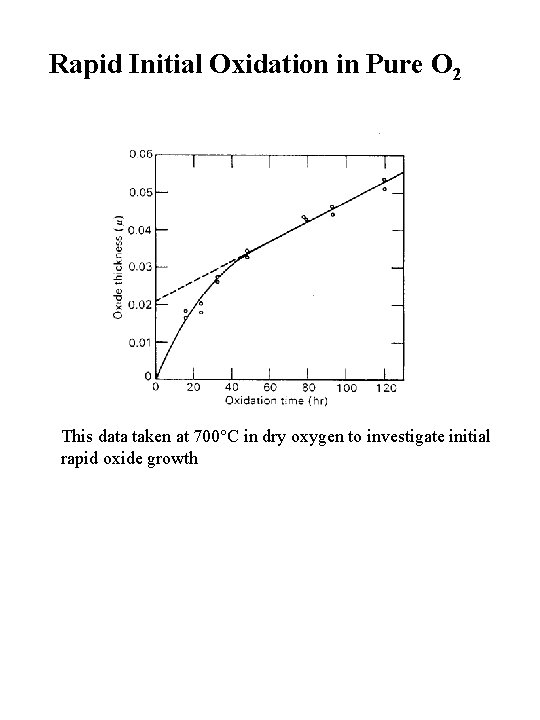
Rapid Initial Oxidation in Pure O 2 This data taken at 700 C in dry oxygen to investigate initial rapid oxide growth
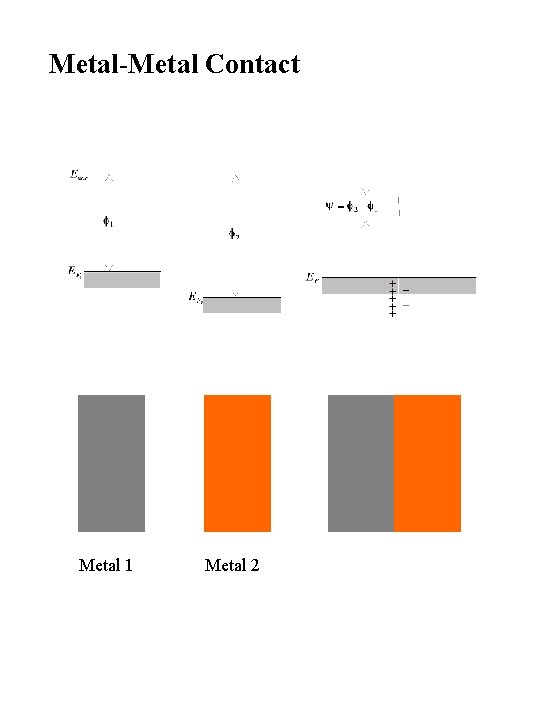
Metal-Metal Contact Metal 1 Metal 2
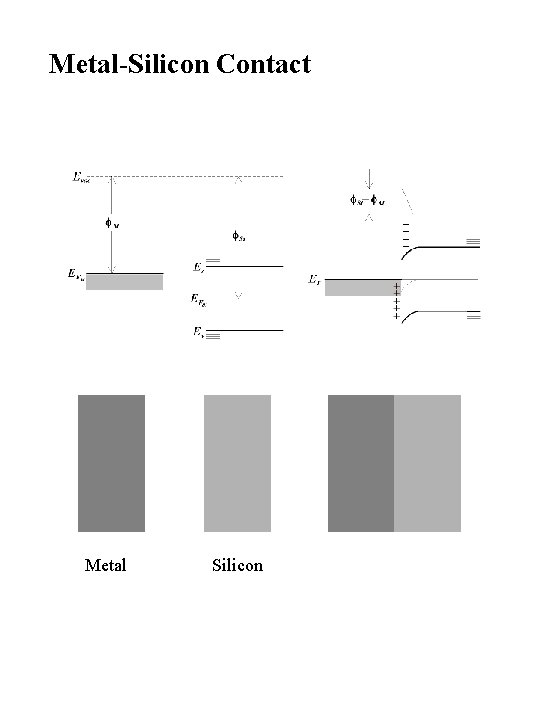
Metal-Silicon Contact Metal Silicon

Effect of a Metal Contact on Silicon
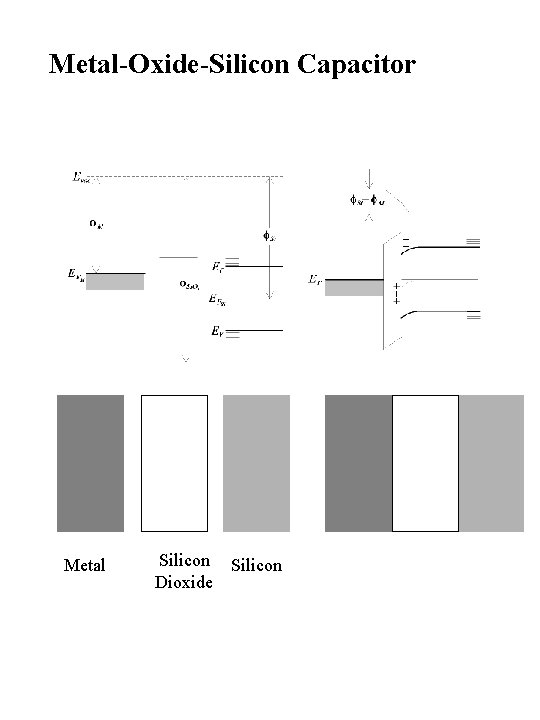
Metal-Oxide-Silicon Capacitor Metal Silicon Dioxide
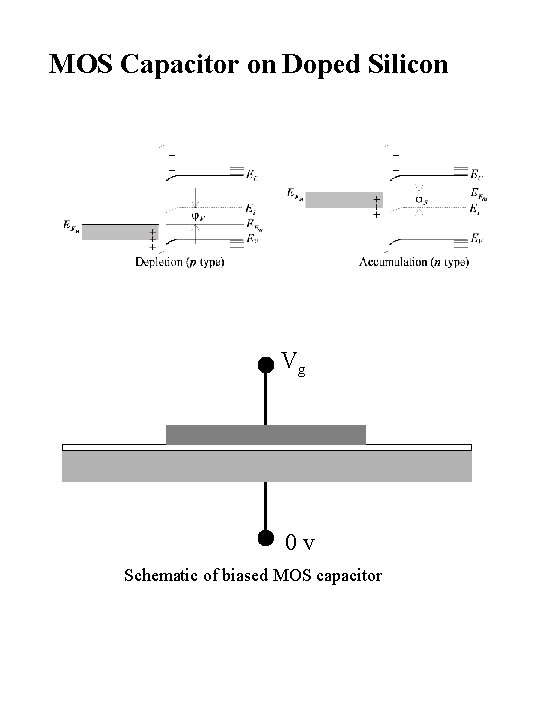
MOS Capacitor on Doped Silicon Vg 0 v Schematic of biased MOS capacitor

Biased MOS Capacitors
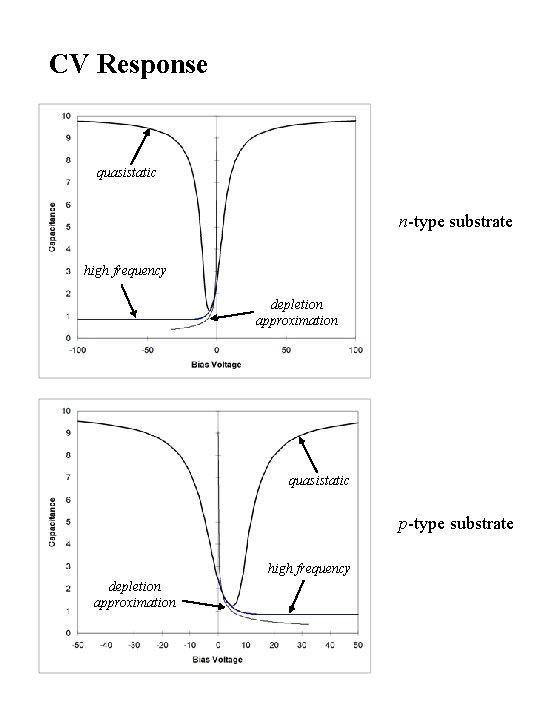
CV Response quasistatic n-type substrate high frequency depletion approximation quasistatic p-type substrate high frequency depletion approximation
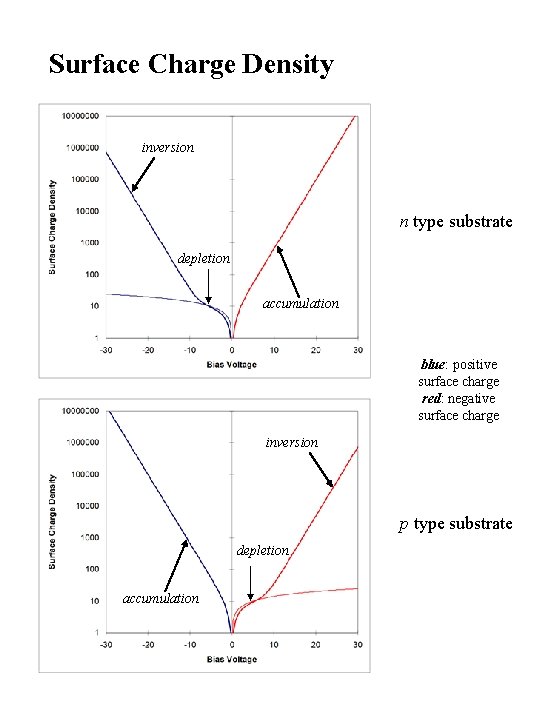
Surface Charge Density inversion n type substrate depletion accumulation blue: positive surface charge red: negative surface charge inversion p type substrate depletion accumulation

Capacitance, Charge, and Potential Poisson’s equation (1 -D) Charge density for a uniformly doped substrate Intrinsic Debye Length: a measure of how much an external electric field penetrates pure silicon

The Depletion Approximation Carrier concentrations are negligible in the depletion region Maximum depletion width Extrinsic Debye Length: a measure of how much an external electric field penetrates doped silicon
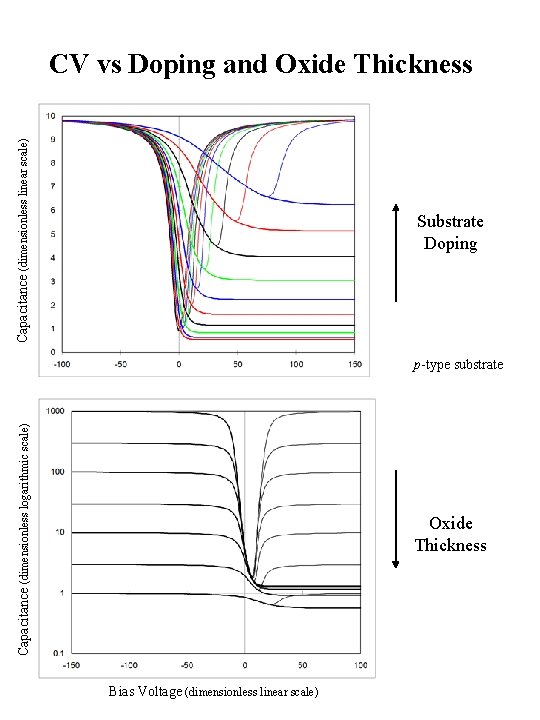
Capacitance (dimensionless linear scale) CV vs Doping and Oxide Thickness Substrate Doping Capacitance (dimensionless logarithmic scale) p-type substrate Oxide Thickness Bias Voltage (dimensionless linear scale)
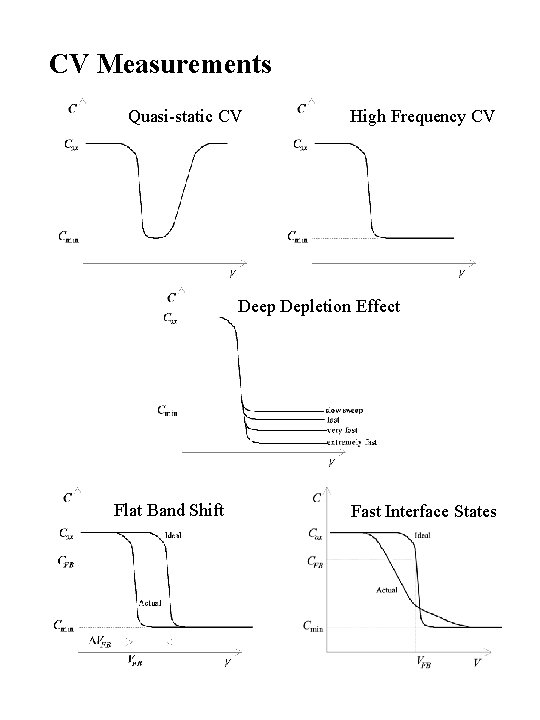
CV Measurements Quasi-static CV High Frequency CV Deep Depletion Effect Flat Band Shift Fast Interface States
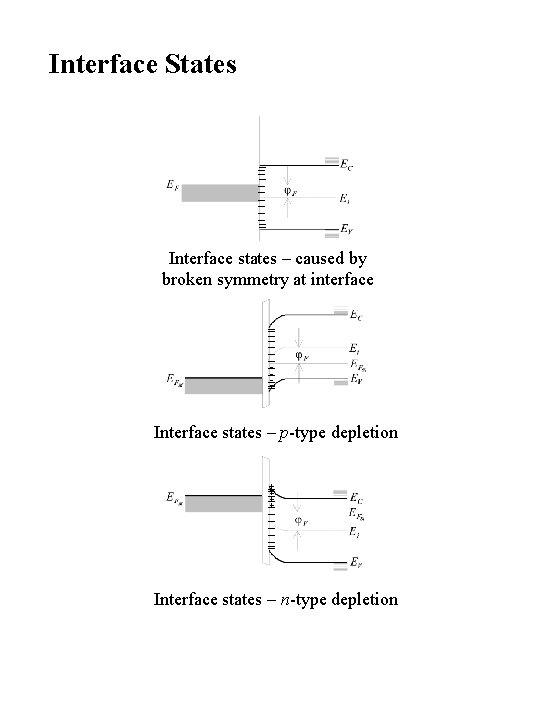
Interface States Interface states – caused by broken symmetry at interface Interface states – p-type depletion + + + Interface states – n-type depletion
![Interface State Density Interface state density is always higher on [111] than [100] Interface State Density Interface state density is always higher on [111] than [100]](http://slidetodoc.com/presentation_image_h2/bce8d3e0e892fa88229cd2936227634f/image-77.jpg)
Interface State Density Interface state density is always higher on [111] than [100]
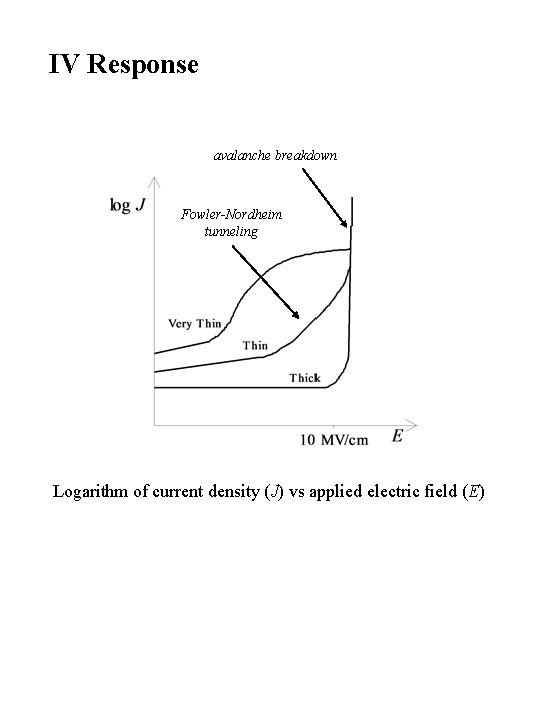
IV Response avalanche breakdown Fowler-Nordheim tunneling Logarithm of current density (J) vs applied electric field (E)
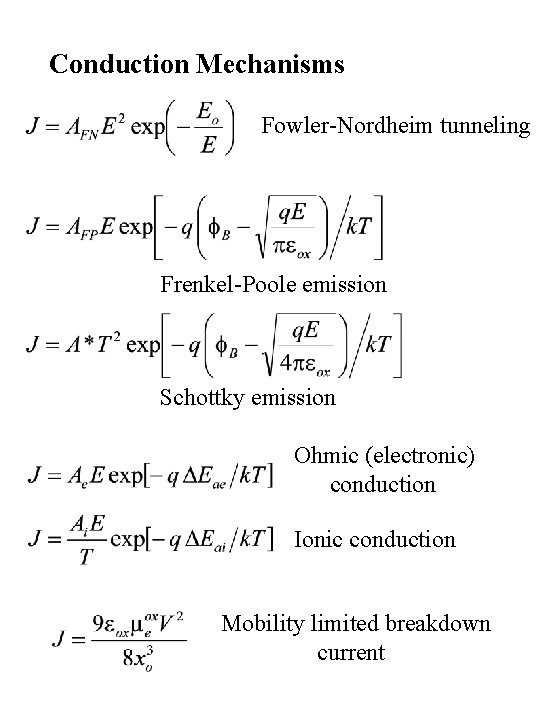
Conduction Mechanisms Fowler-Nordheim tunneling Frenkel-Poole emission Schottky emission Ohmic (electronic) conduction Ionic conduction Mobility limited breakdown current
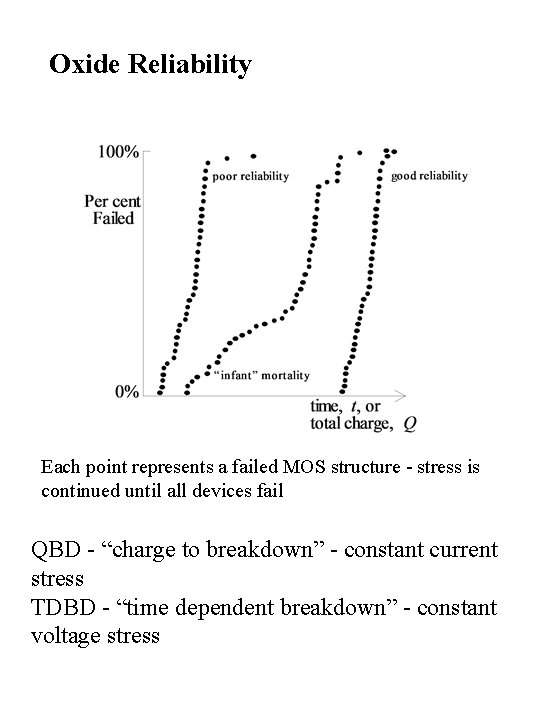
Oxide Reliability Each point represents a failed MOS structure - stress is continued until all devices fail QBD - “charge to breakdown” - constant current stress TDBD - “time dependent breakdown” - constant voltage stress

Linear Transport Processes J = LX J = Flux, X = Force, L = Transport Coefficient Ohm’s Law of electrical conduction: j = E/ J = electric current density, j (units: A/cm 2) X = electric field, E = V (units: volt/cm) V = electrical potential L = conductivity, = 1/ (units: mho/cm) = resistivity ( cm) Fourier’s Law of heat transport: q = T J = heat flux, q (units: W/cm 2) X = thermal force, T (units: K/cm) T = temperature L = thermal conductivity, (units: W/ K cm) Fick’s Law of diffusion: F = D C J = material flux, F (units: /sec cm 2) X = diffusion force, C (units: /cm 4) C = concentration L = diffusivity, D (units: cm 2/sec) Newton’s Law of viscous fluid flow: Fu = u J = velocity flux, Fu (units: /sec 2 cm) X = viscous force, u (units: /sec) u = fluid velocity L = viscosity, (units: /sec cm)
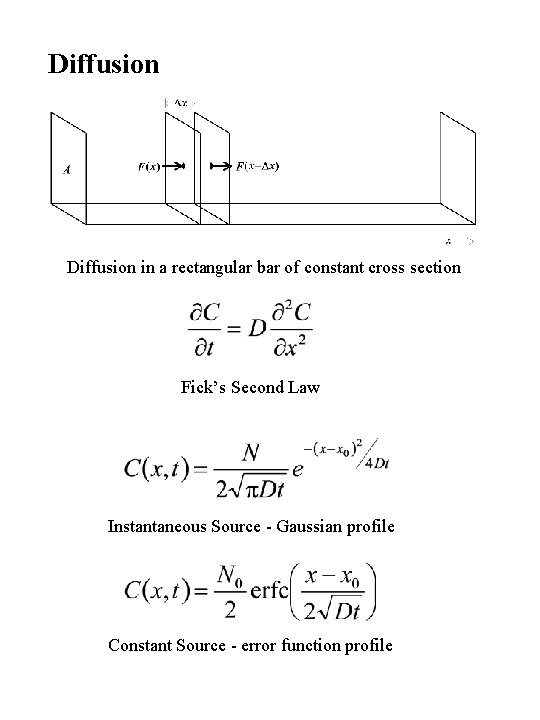
Diffusion in a rectangular bar of constant cross section Fick’s Second Law Instantaneous Source - Gaussian profile Constant Source - error function profile
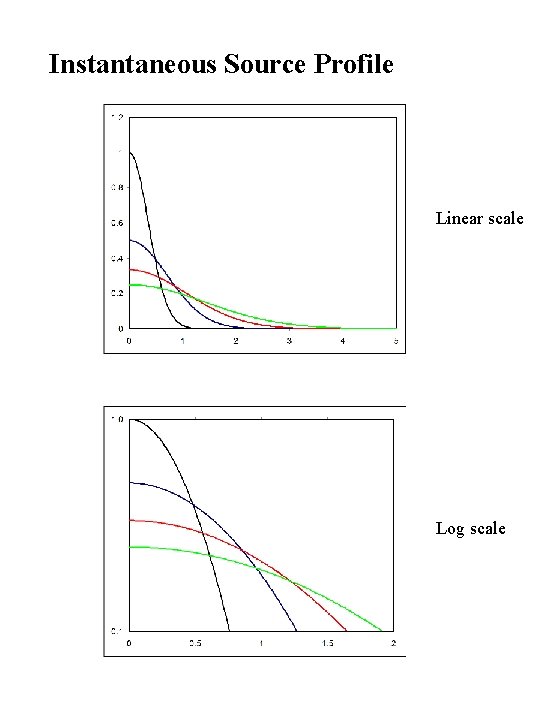
Instantaneous Source Profile Linear scale Log scale
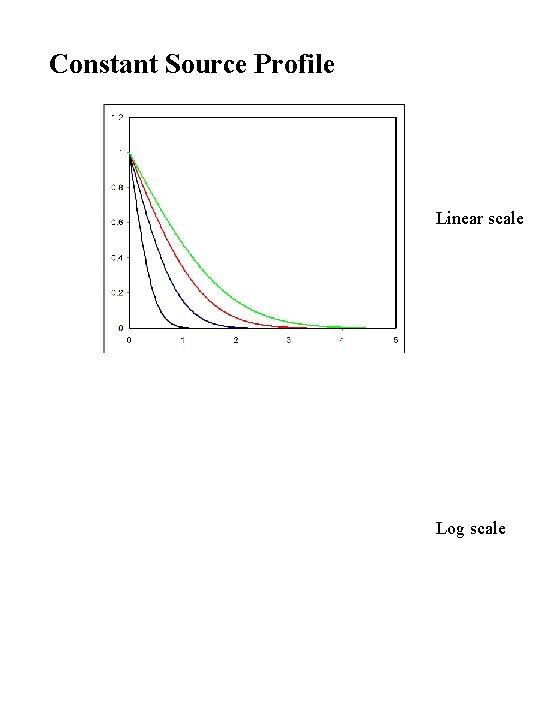
Constant Source Profile Linear scale Log scale
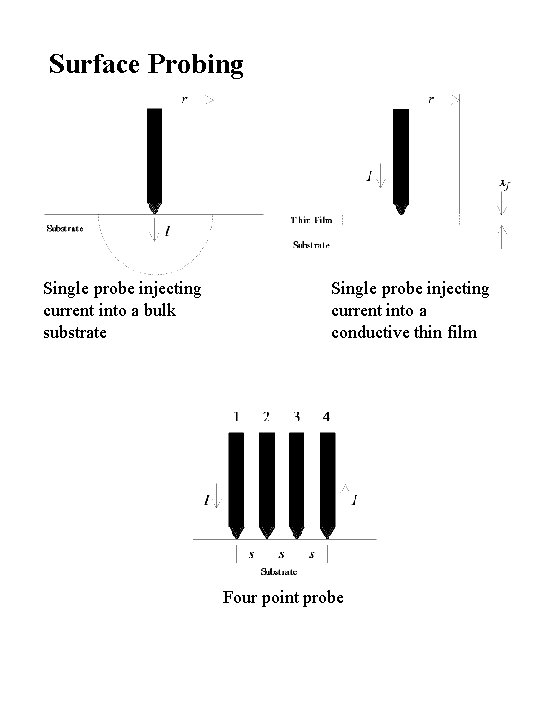
Surface Probing Single probe injecting current into a bulk substrate Single probe injecting current into a conductive thin film Four point probe
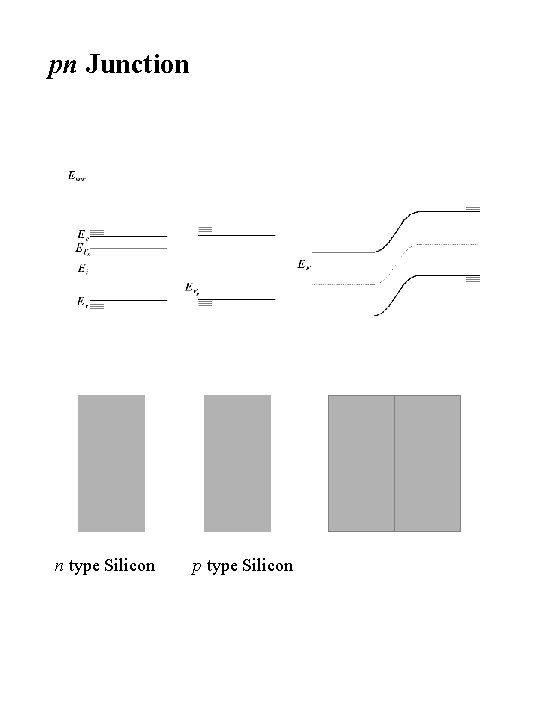
pn Junction n type Silicon p type Silicon
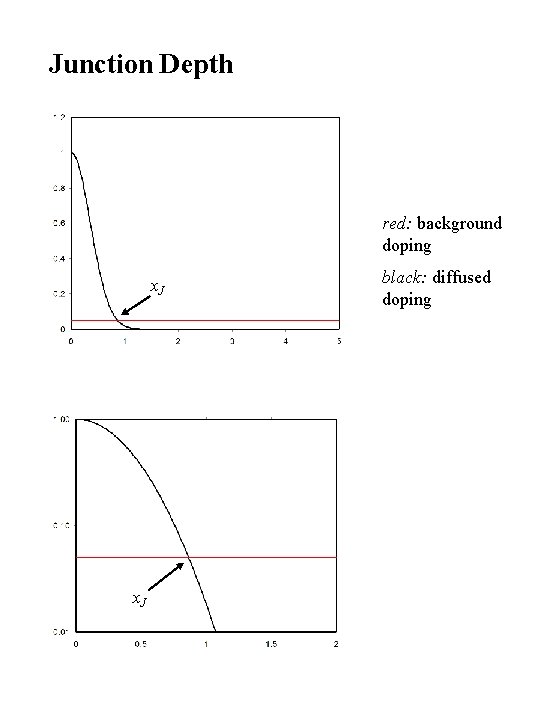
Junction Depth red: background doping x. J black: diffused doping
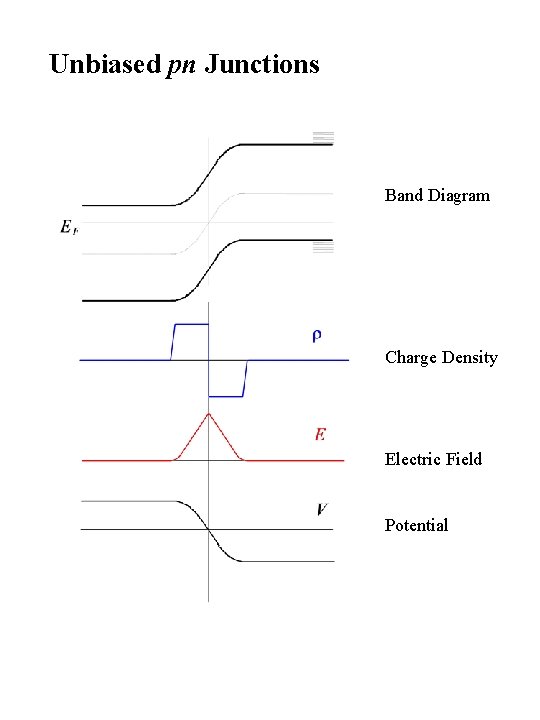
Unbiased pn Junctions Band Diagram Charge Density Electric Field Potential
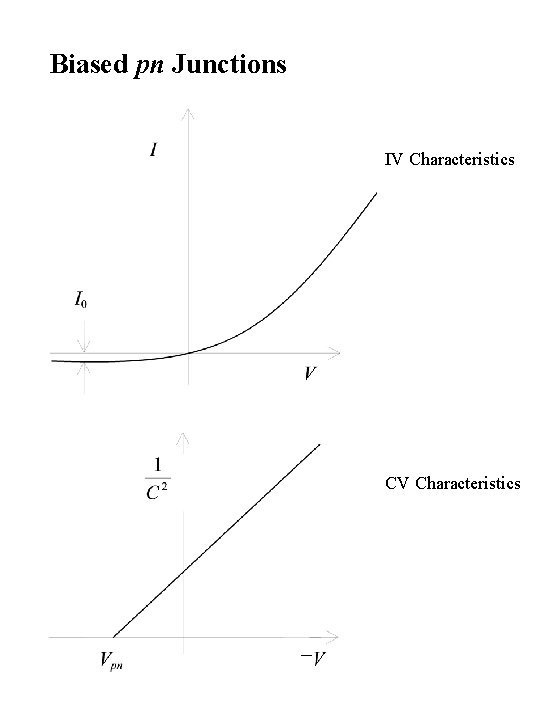
Biased pn Junctions IV Characteristics CV Characteristics
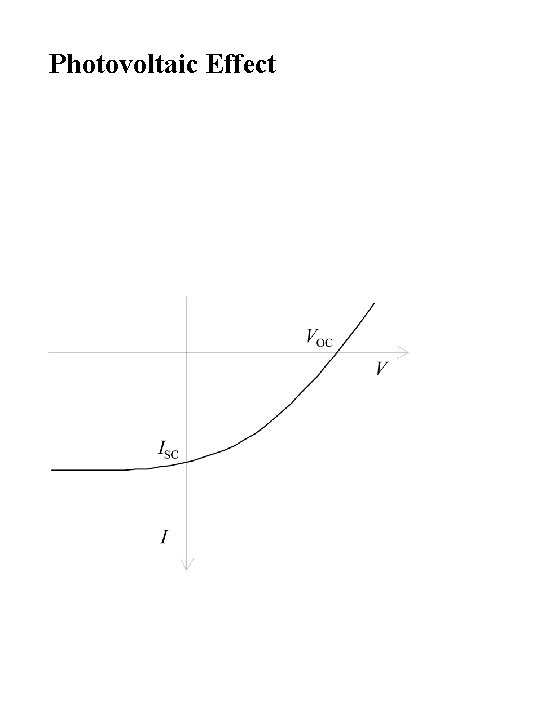
Photovoltaic Effect
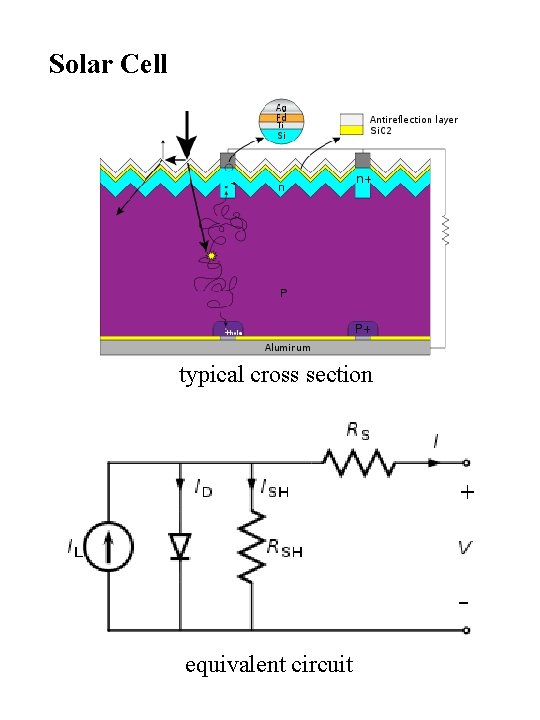
Solar Cell typical cross section equivalent circuit
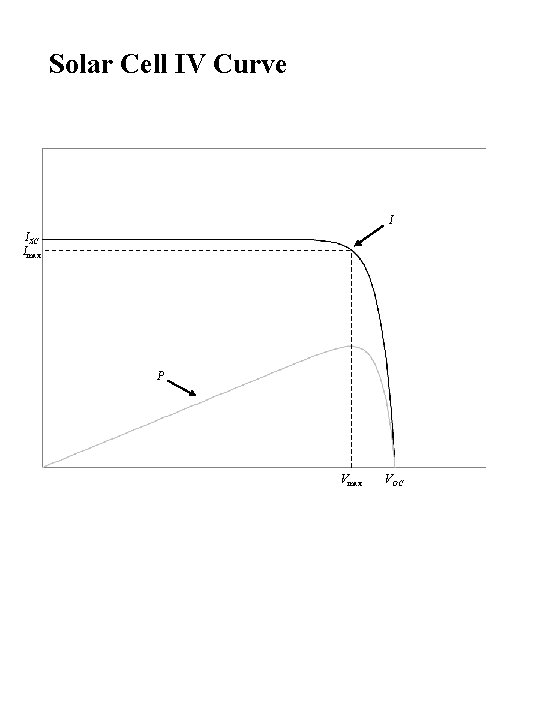
Solar Cell IV Curve I ISC Imax P Vmax VOC
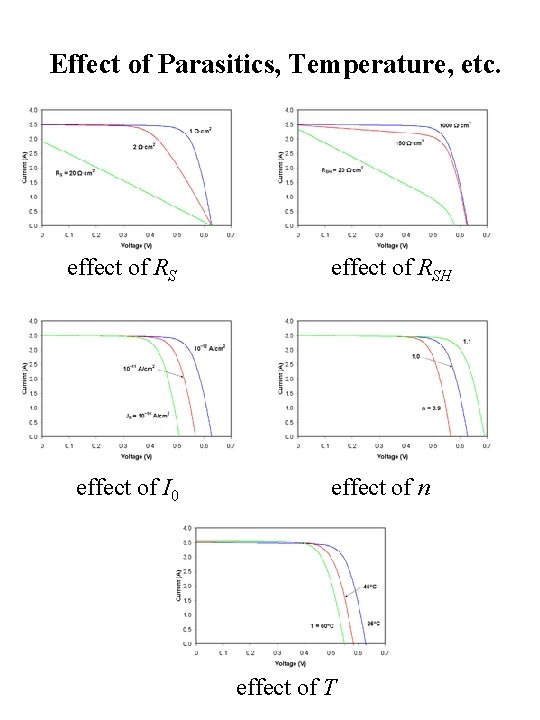
Effect of Parasitics, Temperature, etc. effect of RS effect of I 0 effect of RSH effect of n effect of T
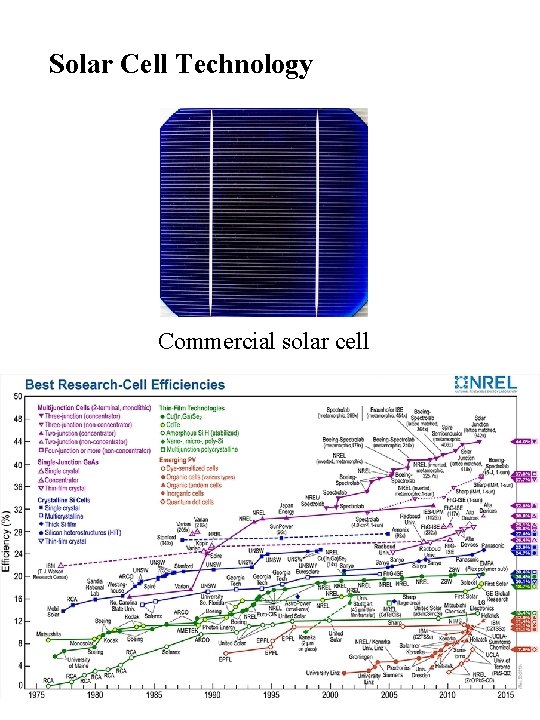
Solar Cell Technology Commercial solar cell
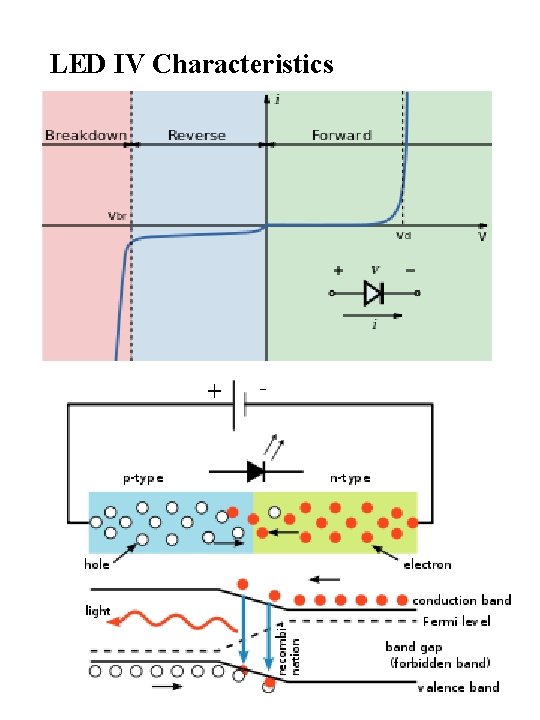
LED IV Characteristics
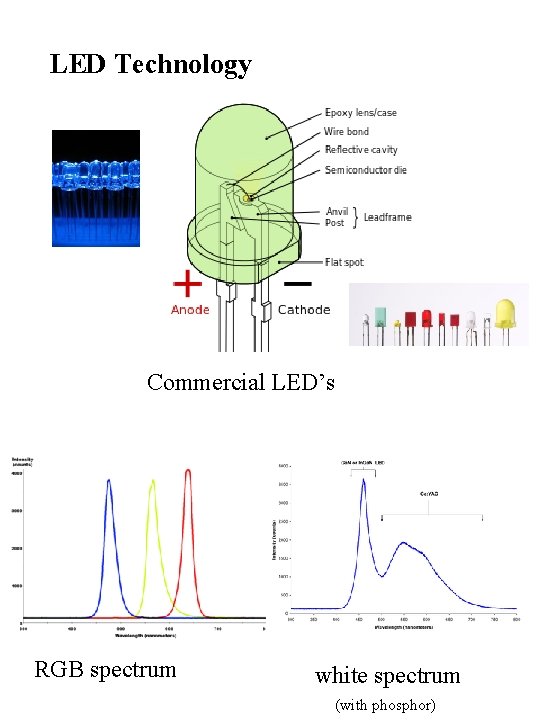
LED Technology Commercial LED’s RGB spectrum white spectrum (with phosphor)

Diffusion Mechanisms Vacancy Diffusion - Substitutional impurities, e. g. , shallow level dopants (B, P, As, Sb, etc. ), Diffusivity is relatively small for vacancy diffusion. Interstitial Diffusion - Interstitial impurities, e. g. , small atoms and metals (O, Fe, Cu, etc. ), Diffusivity is much larger, hence interstitial diffusion is fast compared to vacancy diffusion. Interstitialcy Mechanism - Enhances the diffusivity of substitutional impurities due to exchange with silicon self-interstitials. This leads to enhanced diffusion in the vicinity of the substrate surface during thermal oxidation (socalled “oxidation enhanced diffusion”).

Defect-Carrier Equilibria Vacancies interact with mobile carriers and become charged. In this case, the concentrations are governed by classical mass action equilibria.

Arrhenius Constants for Dopant Atoms

Arrhenius Constants for Other Species
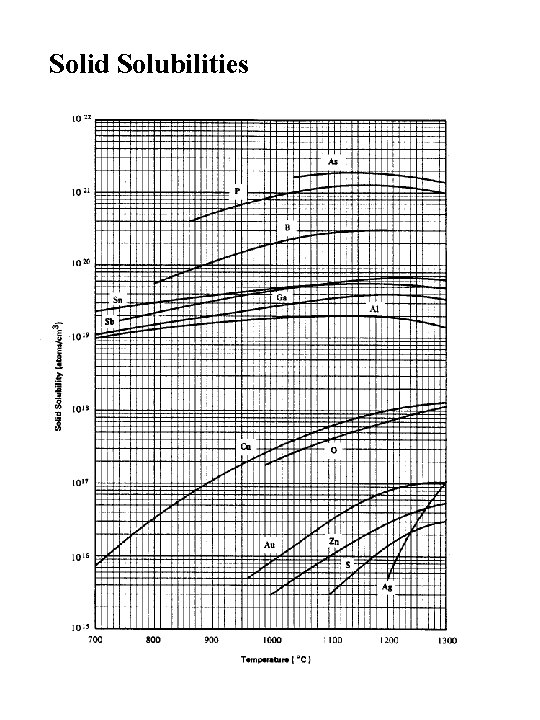
Solid Solubilities
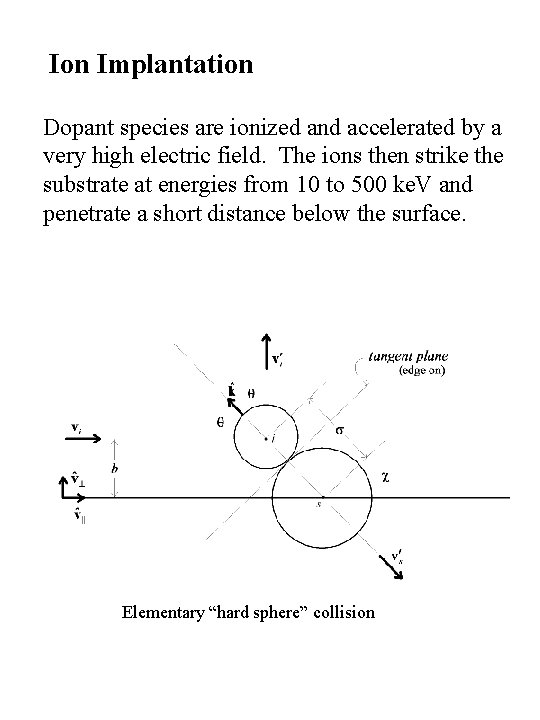
Ion Implantation Dopant species are ionized and accelerated by a very high electric field. The ions then strike the substrate at energies from 10 to 500 ke. V and penetrate a short distance below the surface. Elementary “hard sphere” collision
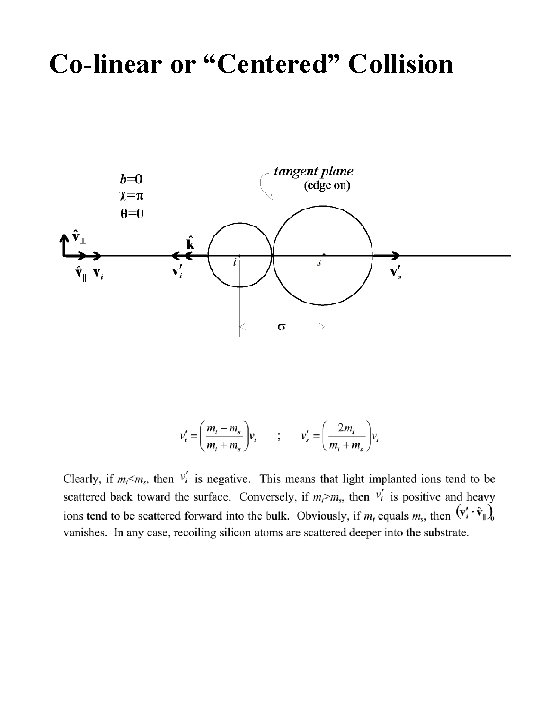
Co-linear or “Centered” Collision
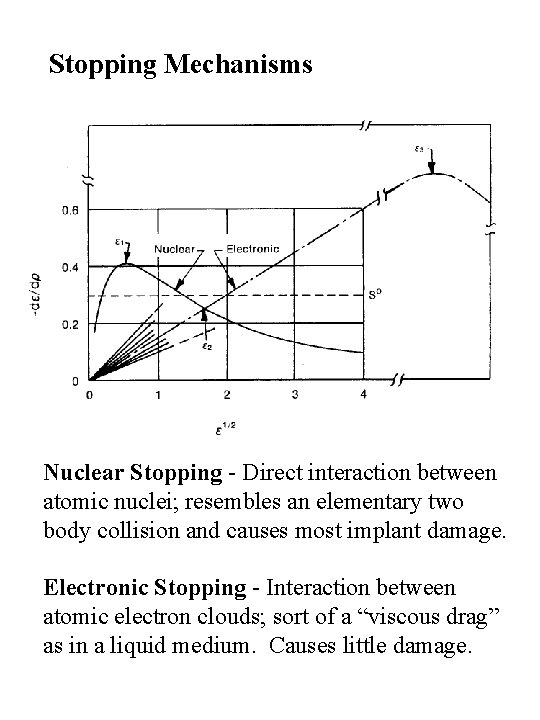
Stopping Mechanisms Nuclear Stopping - Direct interaction between atomic nuclei; resembles an elementary two body collision and causes most implant damage. Electronic Stopping - Interaction between atomic electron clouds; sort of a “viscous drag” as in a liquid medium. Causes little damage.
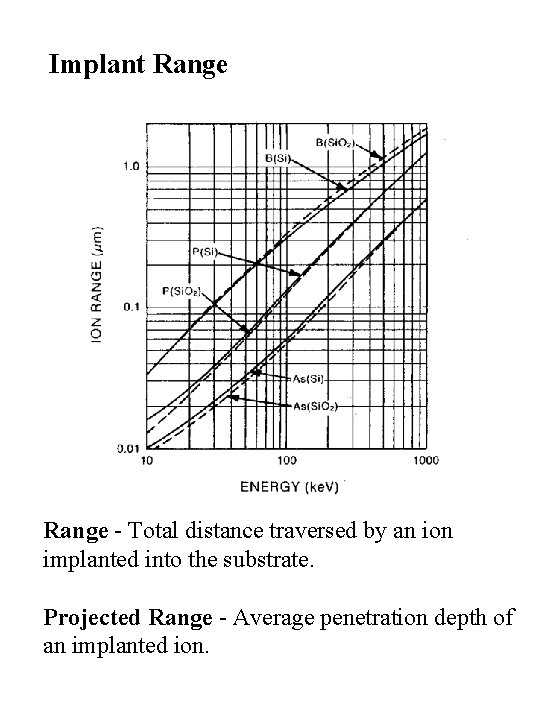
Implant Range - Total distance traversed by an ion implanted into the substrate. Projected Range - Average penetration depth of an implanted ion.
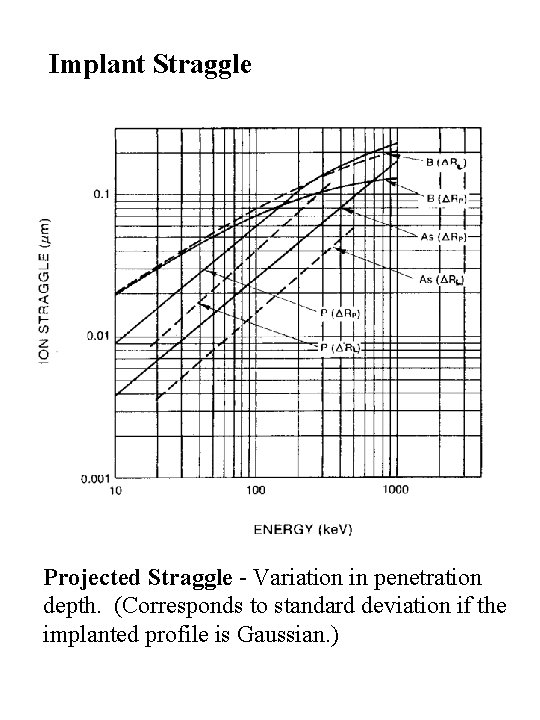
Implant Straggle Projected Straggle - Variation in penetration depth. (Corresponds to standard deviation if the implanted profile is Gaussian. )
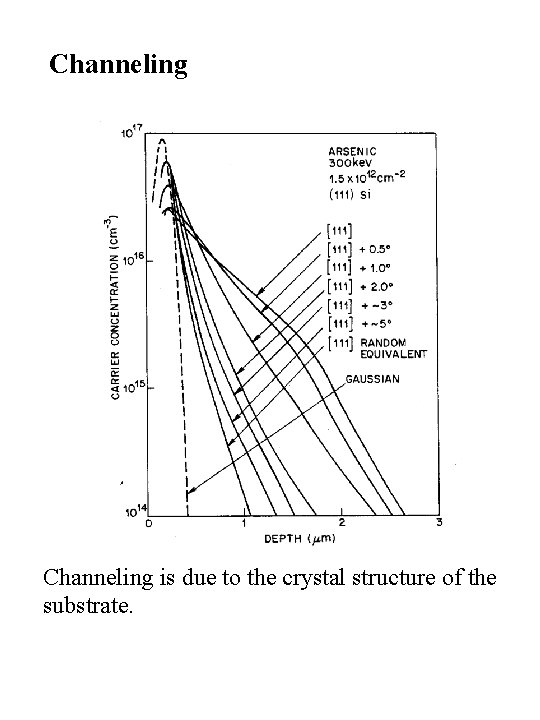
Channeling is due to the crystal structure of the substrate.
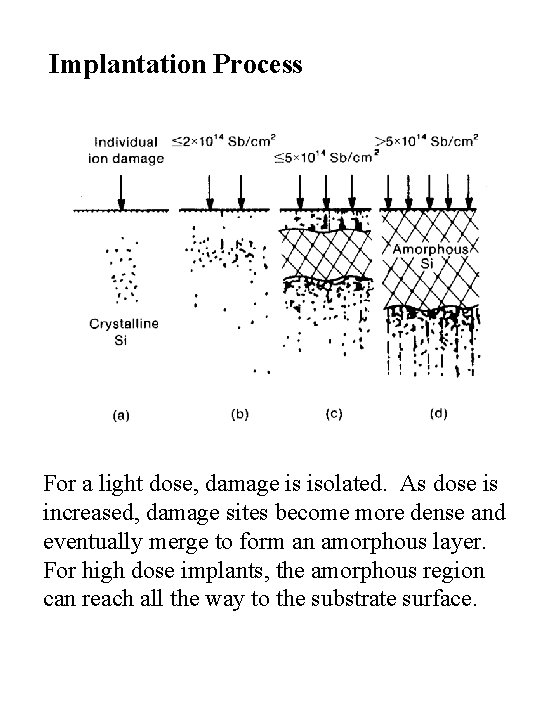
Implantation Process For a light dose, damage is isolated. As dose is increased, damage sites become more dense and eventually merge to form an amorphous layer. For high dose implants, the amorphous region can reach all the way to the substrate surface.

Point-Contact Transistor
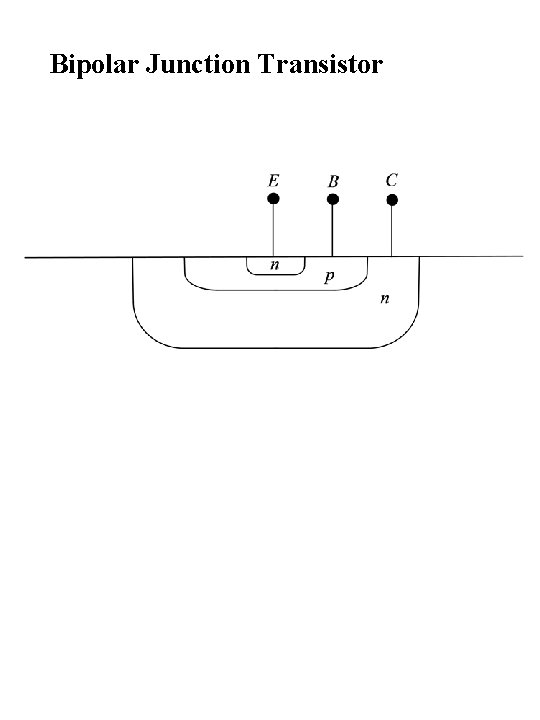
Bipolar Junction Transistor
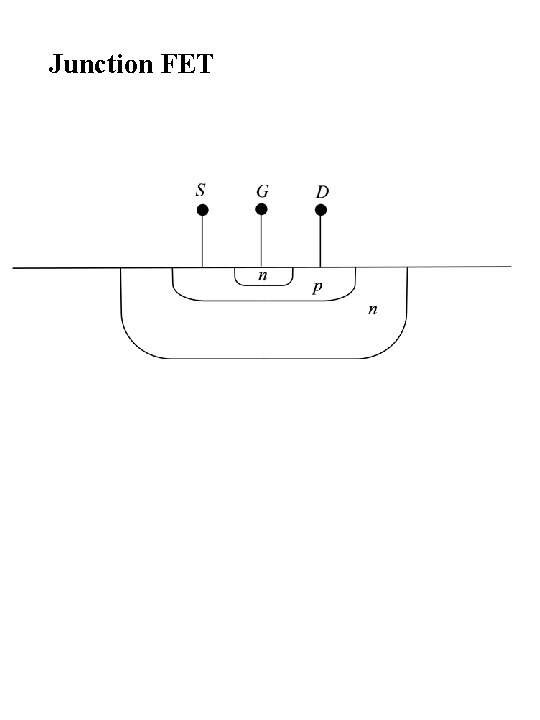
Junction FET
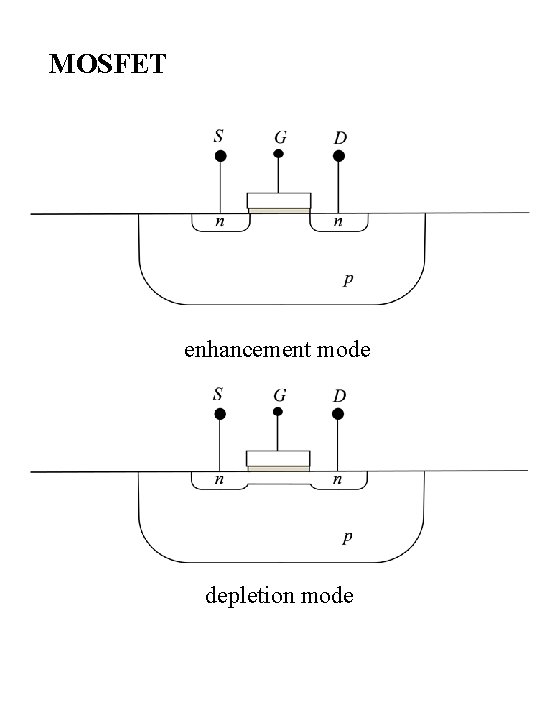
MOSFET enhancement mode depletion mode
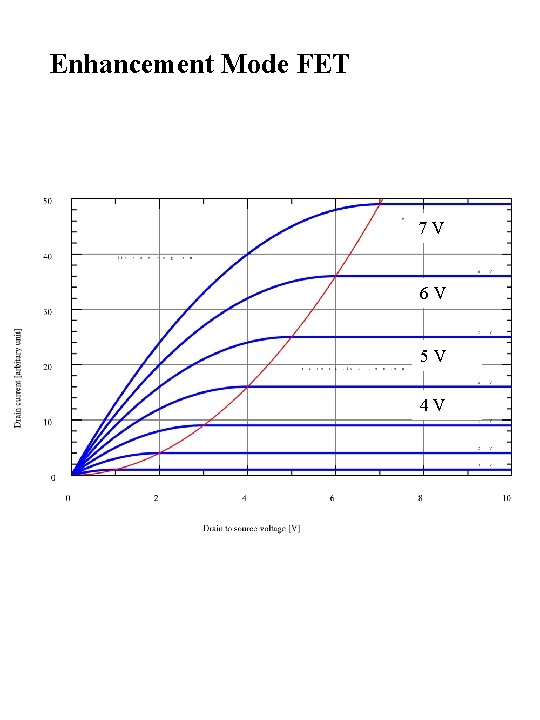
Enhancement Mode FET 7 V 6 V 5 V 4 V
- Slides: 113