Atomic Orbitals 403221 introduction 5 Atomic Orbitals 2





Atomic Orbitals ����� 403221 -introduction 5
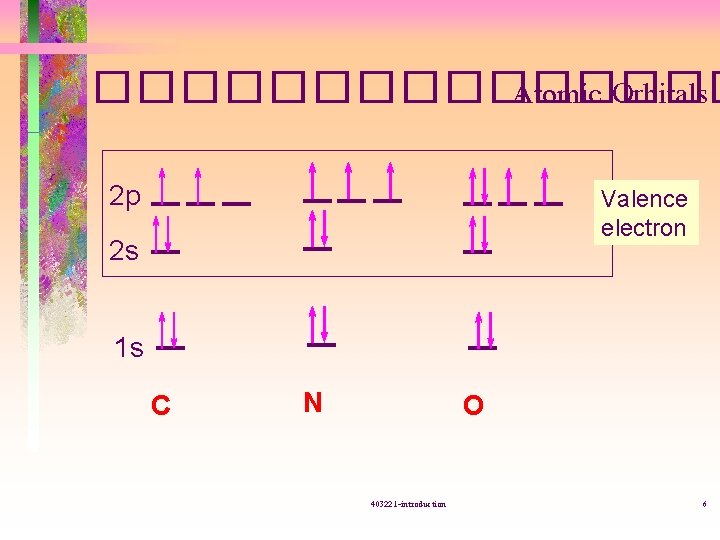
�������� Atomic Orbitals 2 p Valence electron 2 s 1 s C N O 403221 -introduction 6
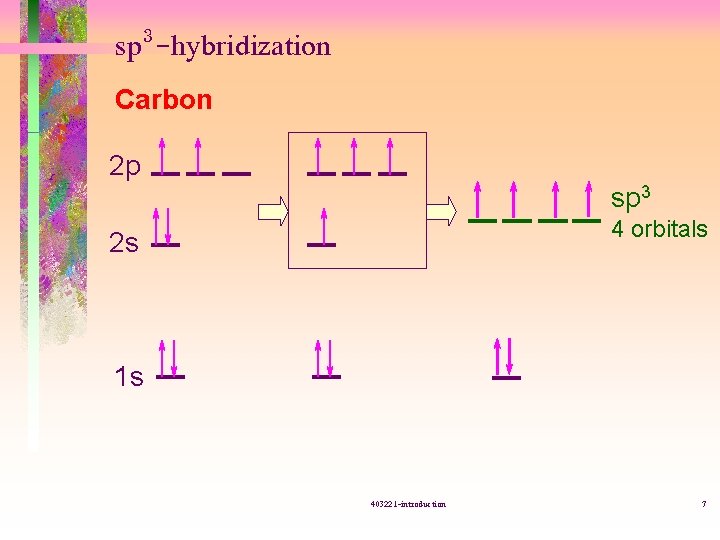
sp 3 -hybridization Carbon 2 p sp 3 4 orbitals 2 s 1 s 403221 -introduction 7
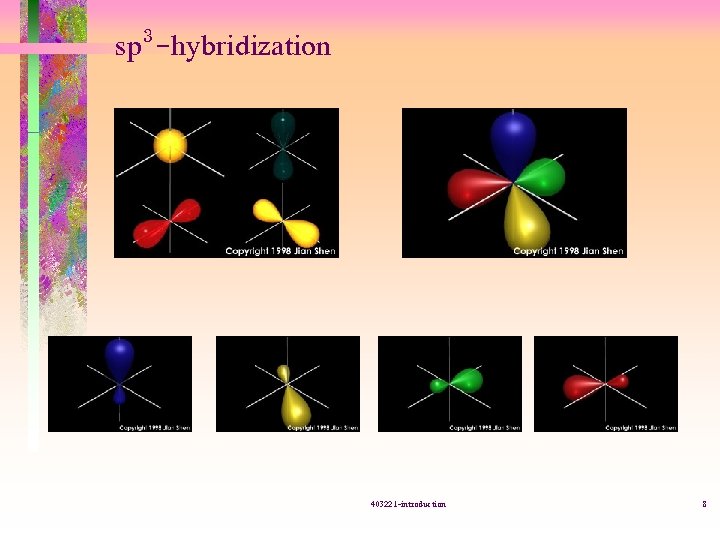
sp 3 -hybridization 403221 -introduction 8
403221 -introduction 9
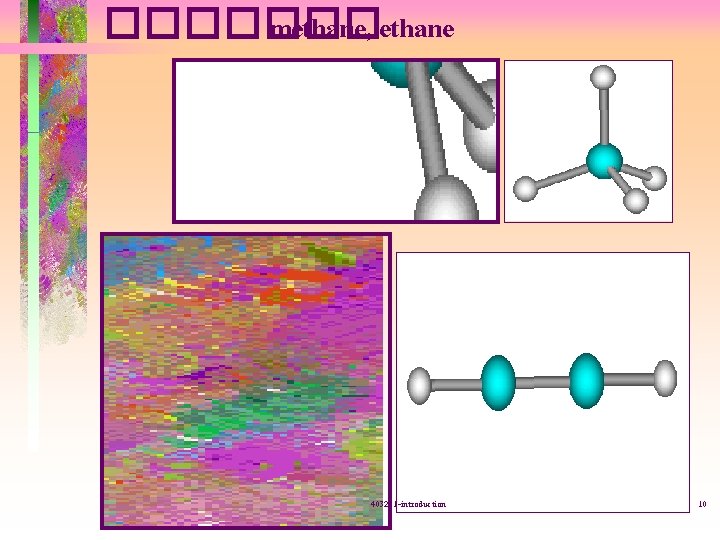
������� methane, ethane 403221 -introduction 10
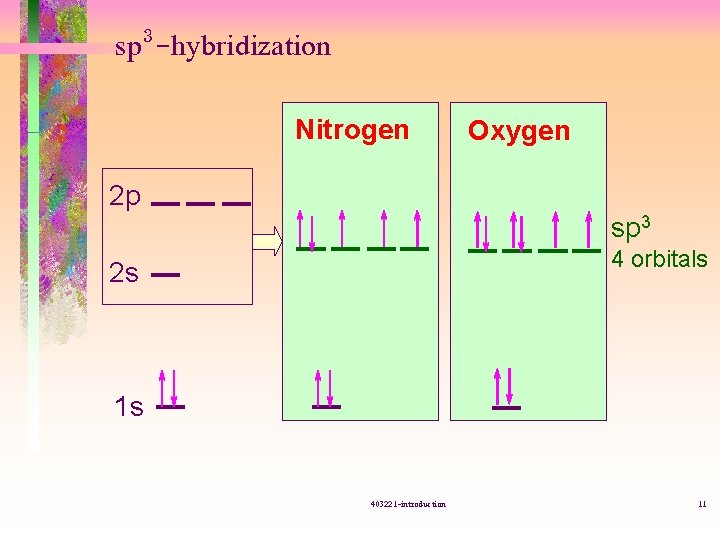
sp 3 -hybridization Nitrogen Oxygen 2 p sp 3 4 orbitals 2 s 1 s 403221 -introduction 11
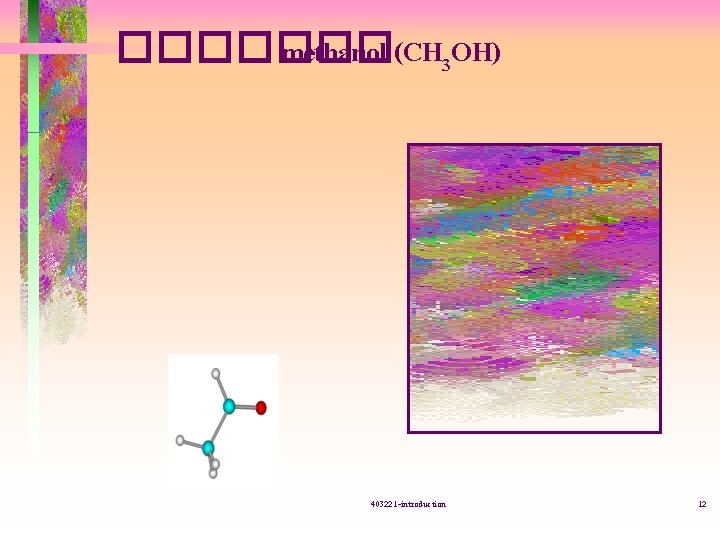
������� methanol (CH 3 OH) 403221 -introduction 12
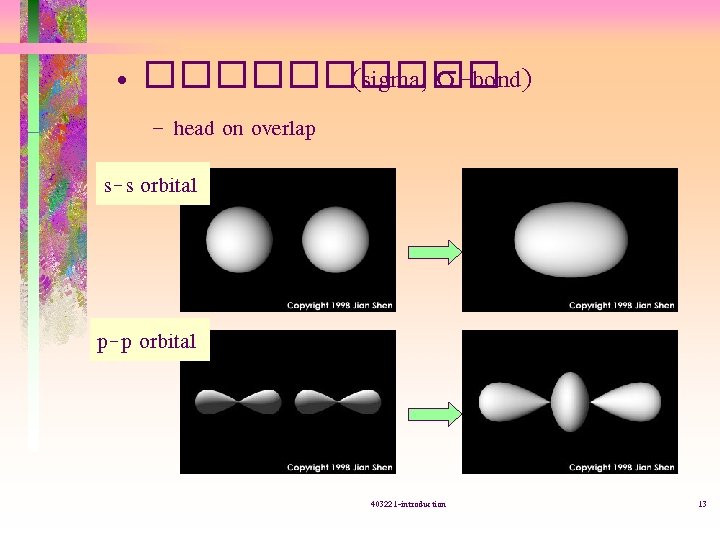
• ����� (sigma, s-bond) – head on overlap s-s orbital p-p orbital 403221 -introduction 13

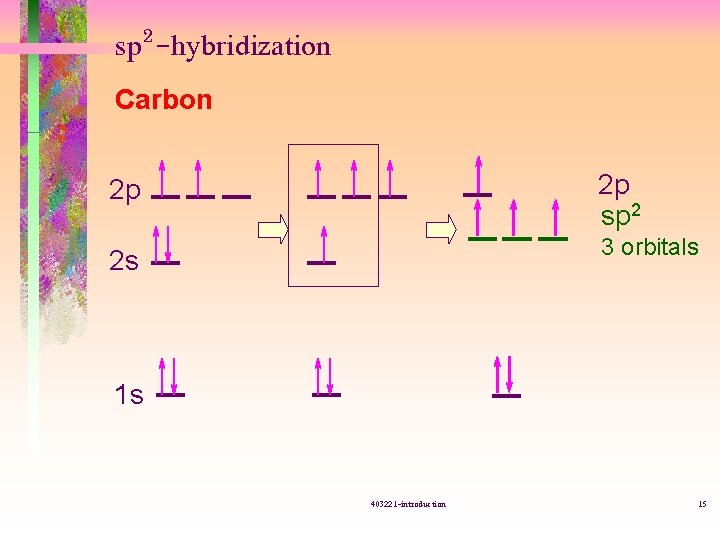
sp 2 -hybridization Carbon 2 p sp 2 2 p 3 orbitals 2 s 1 s 403221 -introduction 15
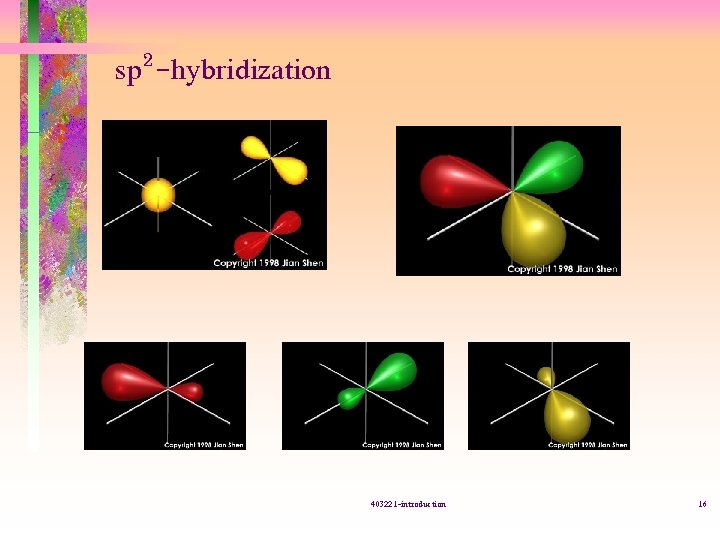
sp 2 -hybridization 403221 -introduction 16
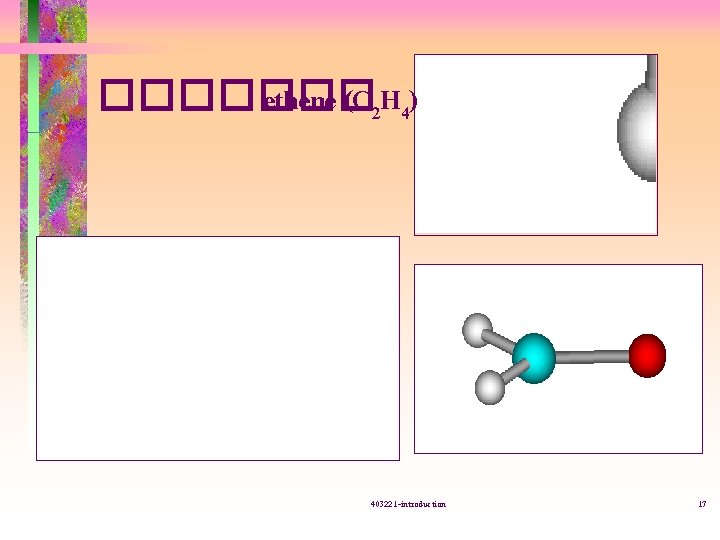
������� ethene (C 2 H 4) 403221 -introduction 17
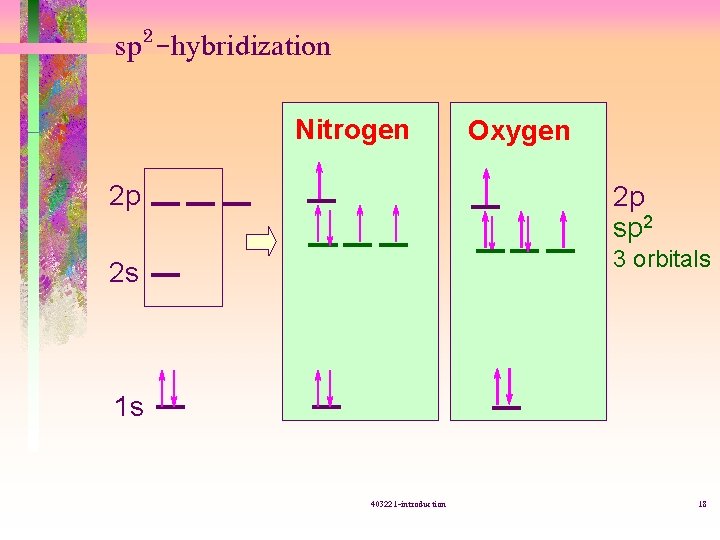
sp 2 -hybridization Nitrogen 2 p Oxygen 2 p sp 2 3 orbitals 2 s 1 s 403221 -introduction 18

������� formaldehyde ��� acetaldehyde formaldehyde CH 2 O acetaldehyde CH 3 CHO 403221 -introduction 19
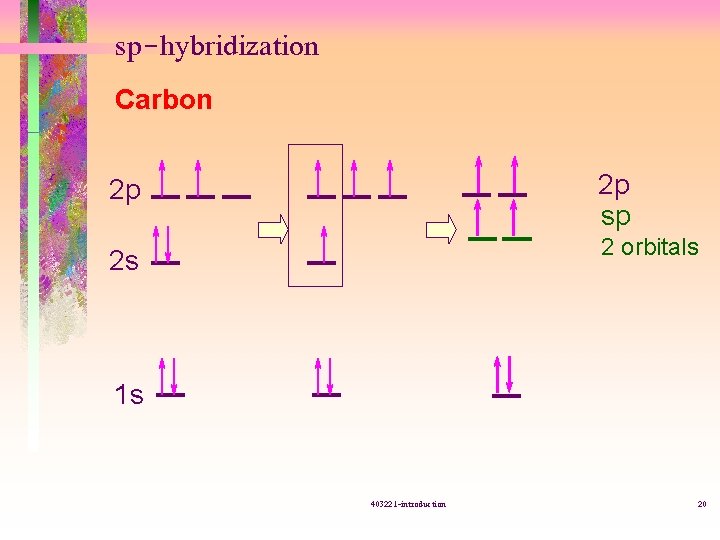
sp-hybridization Carbon 2 p sp 2 p 2 orbitals 2 s 1 s 403221 -introduction 20

sp-hybridization 403221 -introduction 21

������� ethyne (C 2 H 2) 403221 -introduction 22
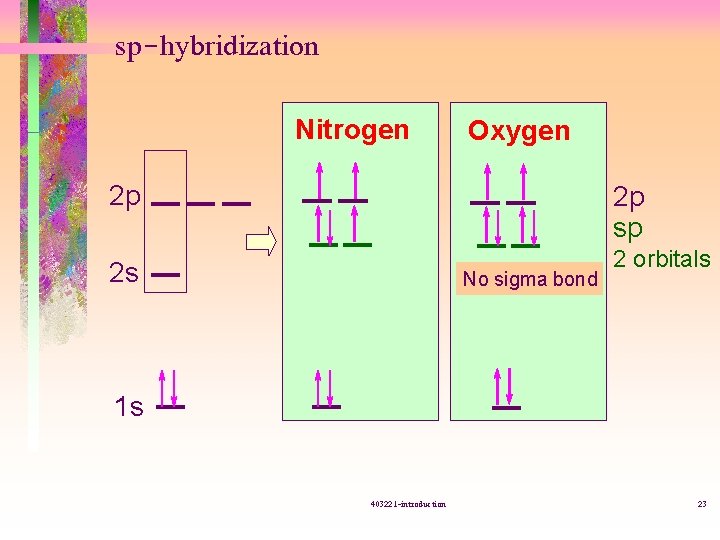
sp-hybridization Nitrogen Oxygen 2 p 2 p sp 2 s No sigma bond 2 orbitals 1 s 403221 -introduction 23
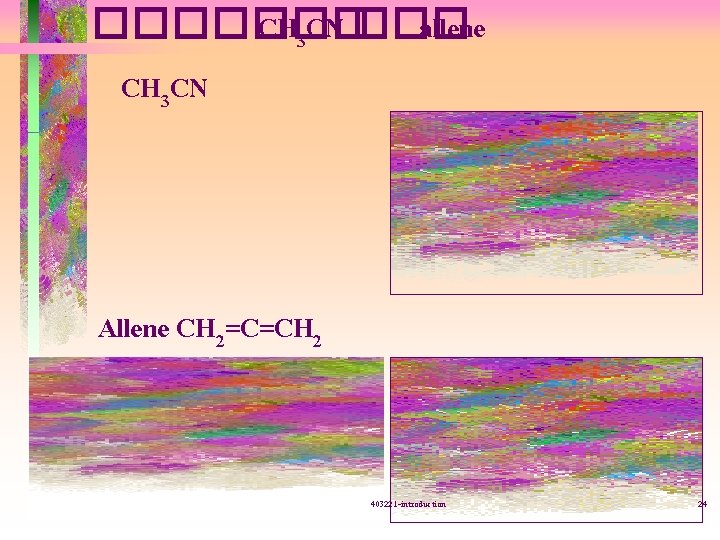
������� CH 3 CN ��� allene CH 3 CN Allene CH 2=C=CH 2 403221 -introduction 24

เปรยบเทยบโครงสรางของ ethyne ethene 403221 -introduction ethyne ethene ethane 25
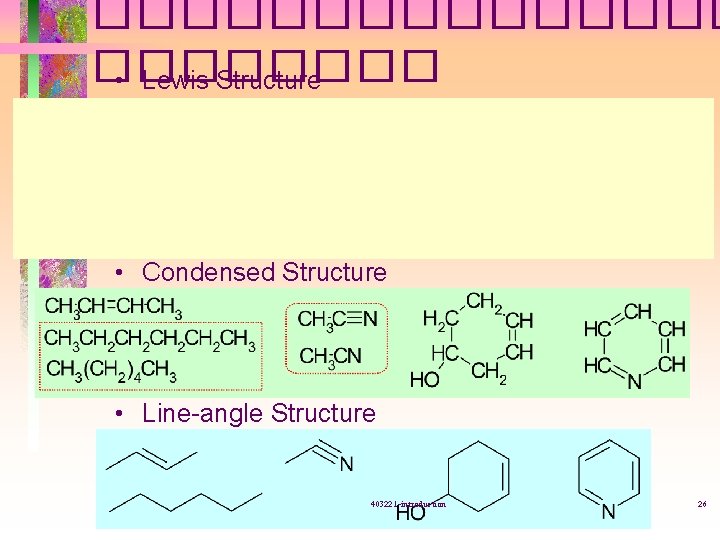
�������� • Lewis Structure • Condensed Structure • Line-angle Structure 403221 -introduction 26
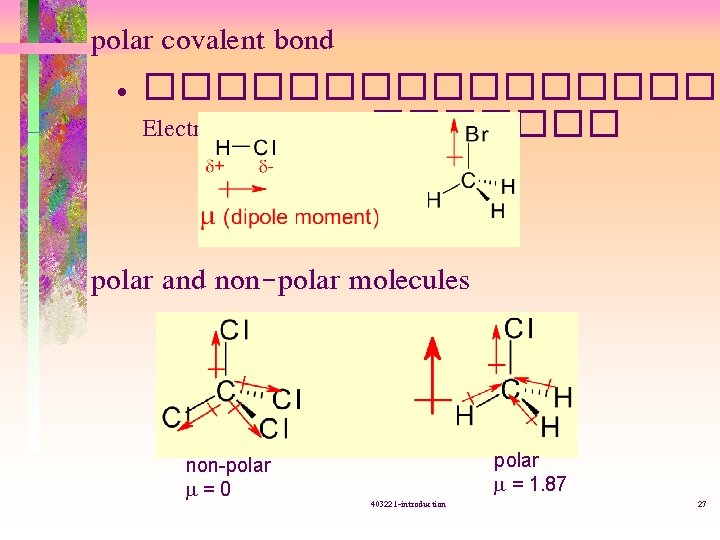
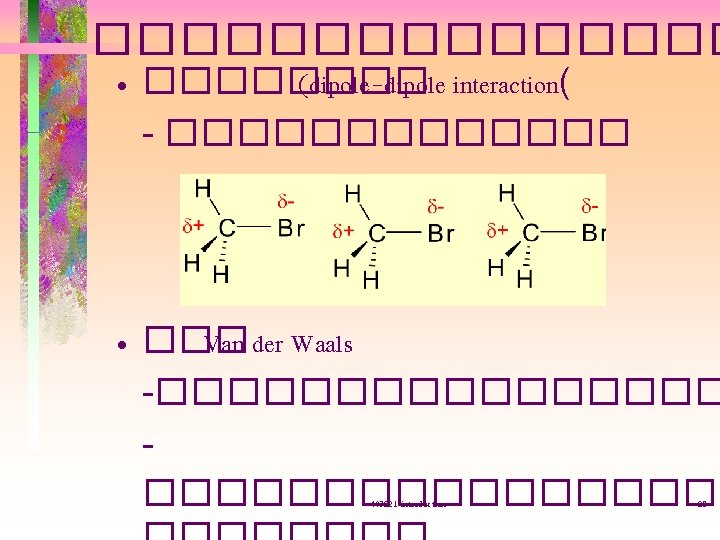
polar covalent bond • ��������� Electronegavity (EN) ������� polar and non-polar molecules non-polar m=0 403221 -introduction polar m = 1. 87 27



403221 -introduction 31

403221 -introduction 32

403221 -introduction 33

403221 -introduction 34

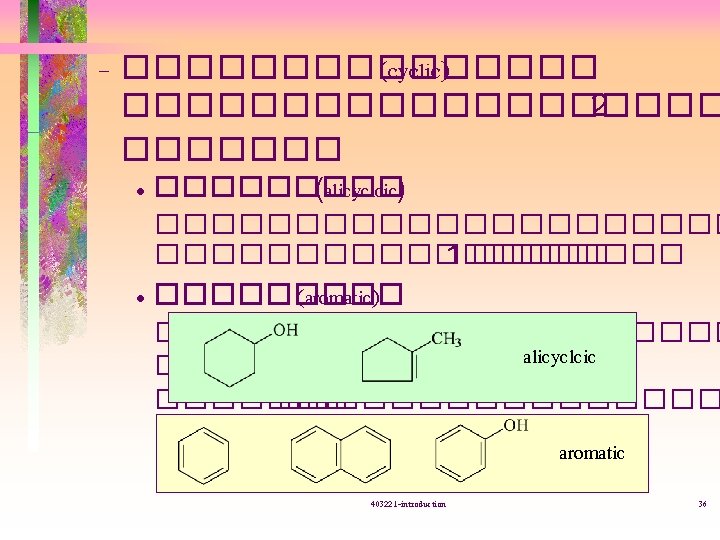

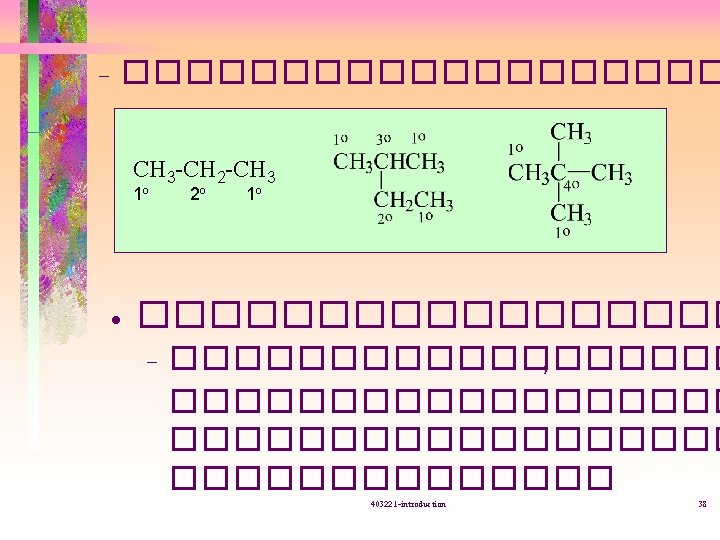

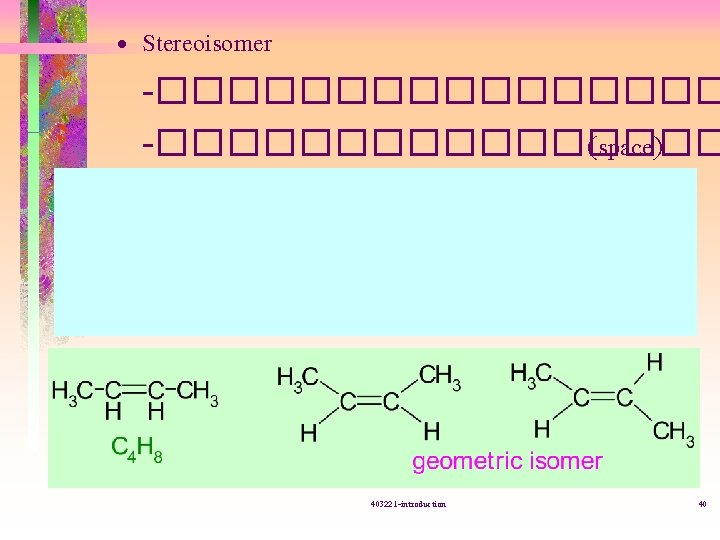
����� (isomer) ��������� • Structural isomer = constitutional isomer -�������� (connectivity( -�������� 403221 -introduction 39
- Slides: 40