Atomic Number n Atoms are composed of identical

Atomic Number n Atoms are composed of identical protons, neutrons, and electrons n How then are atoms of one element different from another element? n Elements are different because they contain different numbers of _____ n The “atomic number” of an element is the ________in the nucleus n# protons in an atom = # _____

Atomic Number Atomic number (Z) of an element is the number of protons in the nucleus of each atom of that element. Element # of protons Carbon 6 Phosphorus Gold Atomic # (Z) 15
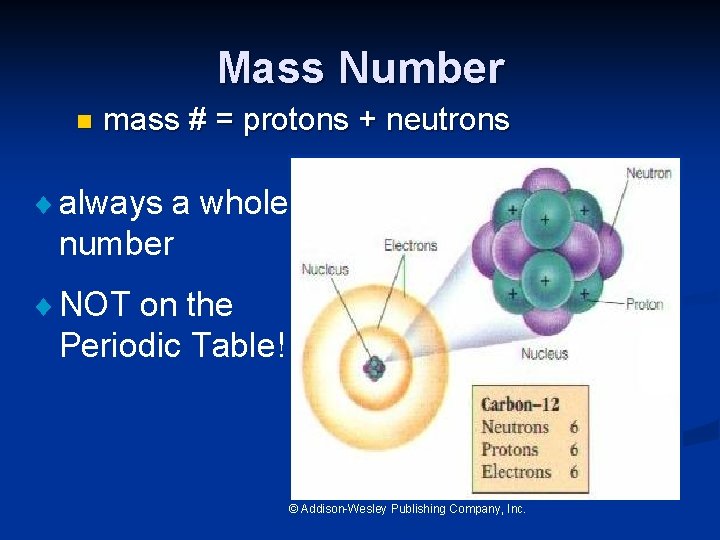
Mass Number n mass # = protons + neutrons ¨ always a whole number ¨ NOT on the Periodic Table! © Addison-Wesley Publishing Company, Inc.

Mass Number Mass number is the number of protons and neutrons in the nucleus of an isotope: Mass # = p+ + n 0 Nuclide p+ Oxygen - 18 Arsenic 75 Phosphorus - 31 n 0 e- Mass #

Symbols ¨ Nuclear symbol: Mass # Atomic # ¨ Hyphen notation: carbon-12

Symbols n Chlorine-37 n atomic #: n mass #: n # of protons: n # of electrons: n # of neutrons:

Symbols n Find each of these: a) number of protons b) number of neutrons c) number of electrons d) Atomic number e) Mass Number 80 35 Br

Symbols n If an element has an atomic number of 34 and a mass number of 78, what is the: a) number of protons b) number of neutrons c) number of electrons d) complete symbol

Symbols n If an element has 91 protons and 140 neutrons what is the a) Atomic number b) Mass number c) number of electrons d) complete symbol

Symbols n If an element has 78 electrons and 117 neutrons what is the a) Atomic number b) Mass number c) number of protons d) complete symbol

Isotopes n Dalton was wrong about all elements of the same type being identical n Atoms of the same element can have different numbers of ______. n Thus, different mass numbers. n These are called _____.

Isotopes n _______(1877 -1956) proposed the idea of isotopes in 1912 n Isotopes are atoms of the same element having different masses, due to varying numbers of neutrons. n Soddy won the Nobel Prize in Chemistry in 1921 for his work with isotopes and radioactive materials.
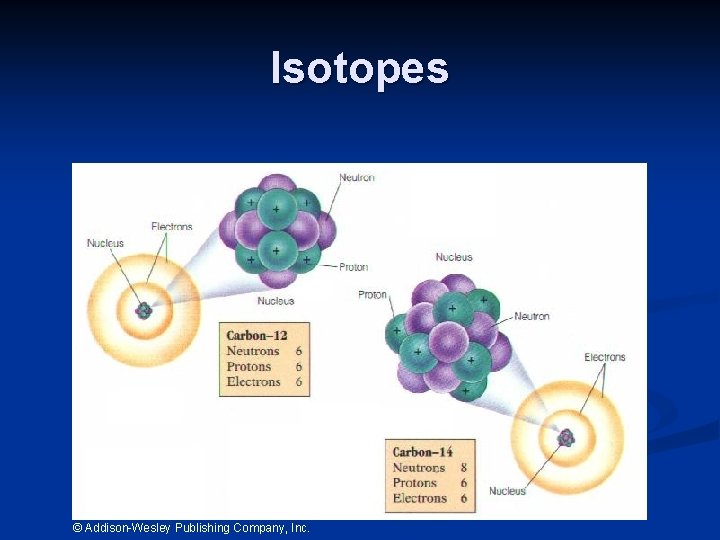
Isotopes © Addison-Wesley Publishing Company, Inc.

Isotopes are atoms of the same element having different masses, due to varying numbers of neutrons. Isotope Hydrogen– 1 (protium) Hydrogen-2 (deuterium) Hydrogen-3 (tritium) Protons Electrons Neutrons Nucleus
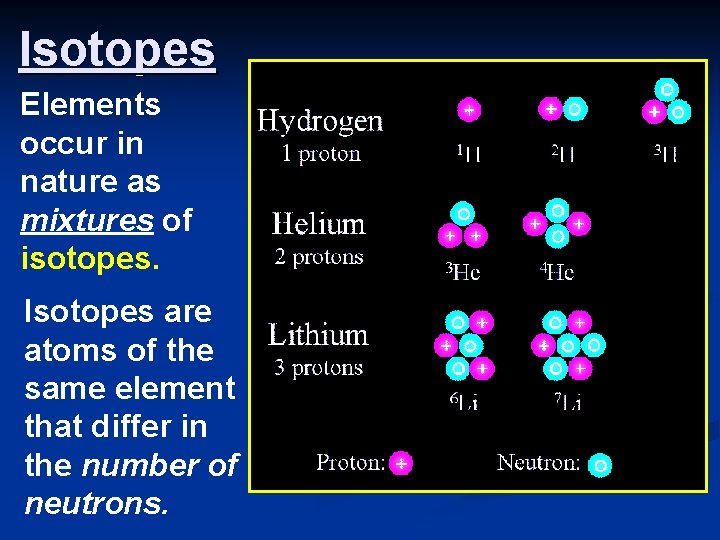
Isotopes Elements occur in nature as mixtures of isotopes. Isotopes are atoms of the same element that differ in the number of neutrons.

Atomic Masses Atomic mass is the average of all the naturally occurring isotopes of that element. Isotope Symbol Carbon-12 12 C Carbon-13 13 C Carbon-14 14 C Composition of the nucleus 6 protons 6 neutrons 6 protons 7 neutrons 6 protons 8 neutrons Carbon = 12. 011 % in nature 98. 89% 1. 11% <0. 01%

Average Atomic Mass weighted average of all isotopes n on the Periodic Table n Avg. Atomic Mass

Average Atomic Mass n EX: Calculate the avg. atomic mass of oxygen if its abundance in nature is 99. 76% 16 O, 0. 04% 17 O, and 0. 20% 18 O. Avg. Atomic Mass

Average Atomic Mass n EX: Find chlorine’s average atomic mass if approximately 8 of every 10 atoms are chlorine 35 and 2 are chlorine-37. Avg. Atomic Mass
- Slides: 19