ATOMIC MODELS from Daltons to Bohrs model 1900

ATOMIC MODELS from Dalton’s to Bohr's model
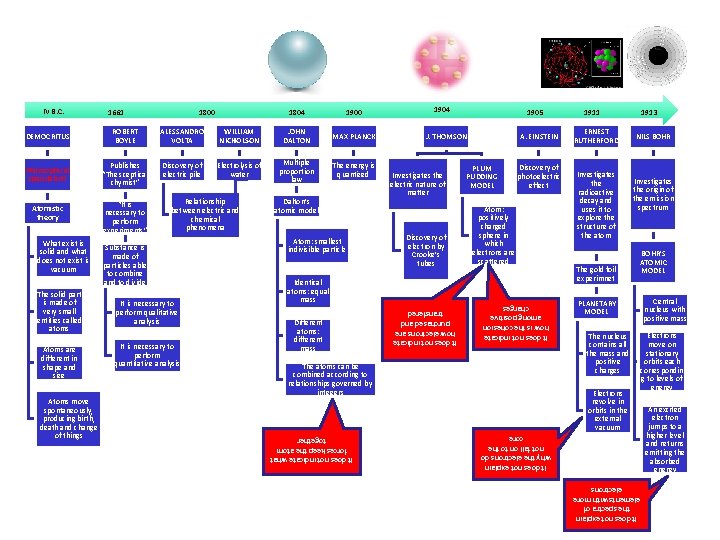
1900 DEMOCRITUS ROBERT BOYLE ALESSANDRO VOLTA WILLIAM NICHOLSON JOHN DALTON MAX PLANCK Philosophical speculation Publishes “The sceptical chymist” Discovery of electric pile Electrolysis of water Multiple proportion law The energy is quantized What exist is solid and what does not exist is vacuum The solid part is made of very small entities called atoms Atoms are different in shape and size Atoms move spontaneously, producing birth, death and change of things “It is necessary to perform experiments” Relationship between electric and chemical phenomena Substance is made of particles able to combine and to divide It is necessary to perform qualitative analysis It is necessary to perform quantitative analysis Dalton’s atomic model Atom: smallest indivisible particle 1905 J. THOMSON Investigates the electric nature of matter Discovery of electron by Crooke’s tubes A. EINSTEIN PLUM PUDDING MODEL Discovery of photoelectric effect Atom: positively charged sphere in which electrons are scattered Identical atoms: equal mass Different atoms: different mass The atoms can be combined according to relationships governed by integers It does not indicate what forces keep the atom together Atomistic theory 1904 1911 ERNEST RUTHERFORD Investigates the radioactive decay and uses it to explore the structure of the atom The gold foil experimnet 1913 NILS BOHR Investigates the origin of the emission spectrum BOHR’S ATOMIC MODEL PLANETARY MODEL The nucleus contains all the mass and positive charges Electrons revolve in orbits in the external vacuum It does not explain the spectra of elements with more electrons 1804 It does not explain why the electrons do not fall on to the core 1800 It does not indicate how electrons are purchased and transfered 1661 It does not indicate how is the cohesion among positive charges IV B. C. Central nucleus with positive mass Electrons move on stationary orbits each correspondin g to levels of energy An excited electron jumps to a higher level and returns emitting the absorbed energy
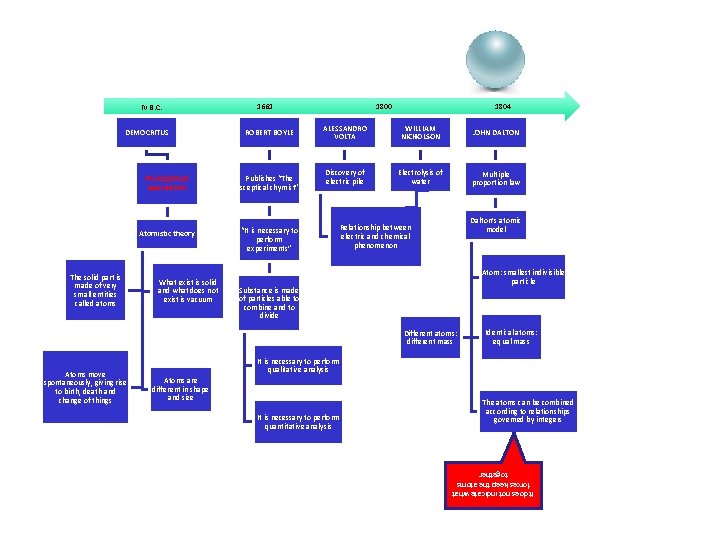
IV B. C. DEMOCRITUS The solid part is made of very small entities called atoms 1661 ROBERT BOYLE Philosophical speculation Publishes “The sceptical chymist” Atomistic theory “It is necessary to perform experiments” What exist is solid and what does not exist is vacuum 1800 1804 ALESSANDRO VOLTA WILLIAM NICHOLSON JOHN DALTON Discovery of electric pile Electrolysis of water Multiple proportion law Dalton’s atomic model Relationship between electric and chemical phenomenon Atom: smallest indivisible particle Substance is made of particles able to combine and to divide Different atoms: different mass It is necessary to perform qualitative analysis Atoms are different in shape and size It is necessary to perform quantitative analysis The atoms can be combined according to relationships governed by integers It does not indicate what forces keep the atoms together Atoms move spontaneously, giving rise to birth, death and change of things Identical atoms: equal mass
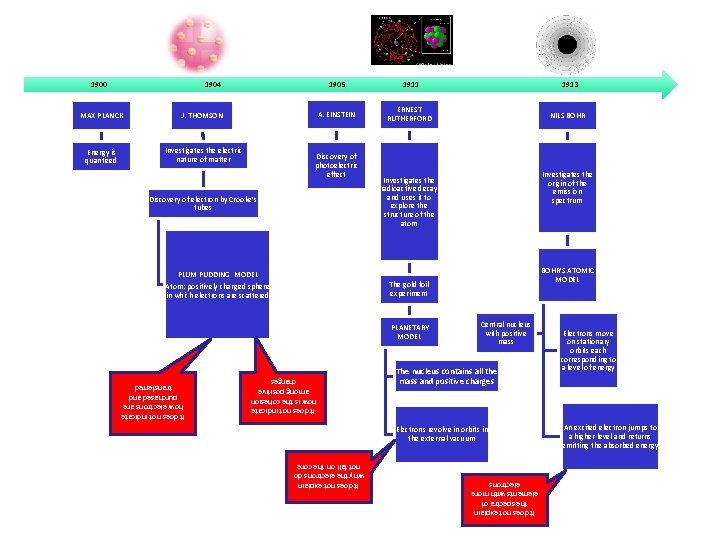
1904 MAX PLANCK J. THOMSON Energy is quantized Investigates the electric nature of matter 1905 1911 A. EINSTEIN ERNEST RUTHERFORD Discovery of photoelectric effect Discovery of electron by Crooke’s tubes PLUM PUDDING MODEL Atom: positively charged sphere in which electrons are scattered 1913 NILS BOHR Investigates the origin of the emission spectrum Investigates the radioactive decay and uses it to explore the structure of the atom BOHR’S ATOMIC MODEL The gold foil experiment PLANETARY MODEL Central nucleus with positive mass The nucleus contains all the mass and positive charges It does not indicate how is the cohesion among positive charges Electrons revolve in orbits in the external vacuum It does not explain the spectra of elements with more electrons 1900 Electrons move on stationary orbits each corresponding to a level of energy An excited electron jumps to a higher level and returns emitting the absorbed energy It does not explain why the electrons do not fall on the core It does not indicate how electrons are purchased and transferred

The starting point • Ancient Greece (IV century BC) • DEMOCRITUS • Early assumptions about the discontinuous nature of matter (never verified )

DEMOCRITUS' HYPOTESIS • atoms have only two qualities: size and geometric shape • each aggregate of atoms can be arranged in a different order, resulting in different compounds.

The first experimental model • England 1803 • DALTON • From the law of multiple proportions Dalton deduced the discontinuous nature ( particulate ) of matter

DALTON'S ATOMIC MODEL (1803 ) • • • any form of matter is composed of atoms; atoms are all unalterable and not divisible into smaller parts • in an elementary substance atoms are all equal and have equal mass; atoms of different elements differ for mass and other characteristics; • chemical transformations take place by a union or separation of atoms between them, are neither created nor destroyed (Lavoisier), nor divided into smaller parts, or even transformed into other types of atoms. • atoms of different elements combine in simple whole-number ratios to form chemical compounds

…. the limitations of the model. . . • Dalton’s atom didn’t allow to interpret the electrical phenomena of matter • experimental evidence (electrification, electrodeposition of metals. . . ) showed that the presence of electric charges in the material • since Dalton’s model contemplated an atom which was indivisible and electrically neutral, it was abandoned.

• Where are the electrical charges in the atom ? • What sign are they? • How many are they?

The second experimental model • England, 19 th-20 th centuries • THOMSON • A study of cathode ray tubes (discharge of electricity in rarefied gases) shows the existence, inside the atom, of ELECTRONS, negative electrical particles

THOMSON’S ATOMIC MODEL (1904) • The atom is a positively charged sphere • inside there are small particles with negative charge (electrons) that neutralize the positive charge • overall the atom is neutral

• After a few years , it was also demonstrated the existence of positive particles (protons ). • but how are they arranged within these two types of particles of the atom ? • Is Thomson’s model still valid?

The third experimental model • England 20 th century • RUTHERFORD • By studying the nature of positively charged in the atom came the discovery of PROTON and its position in the atomic CORE (planetary model -1911 )
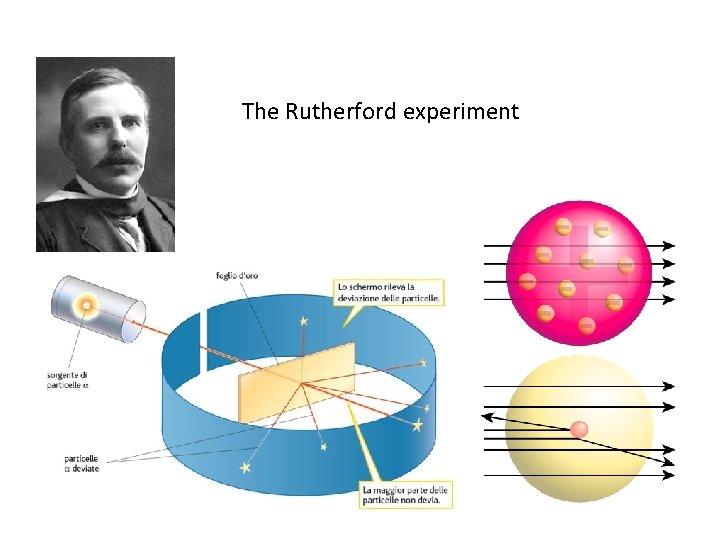
The Rutherford experiment

but also. . . Rutherford's model had limitations. . .
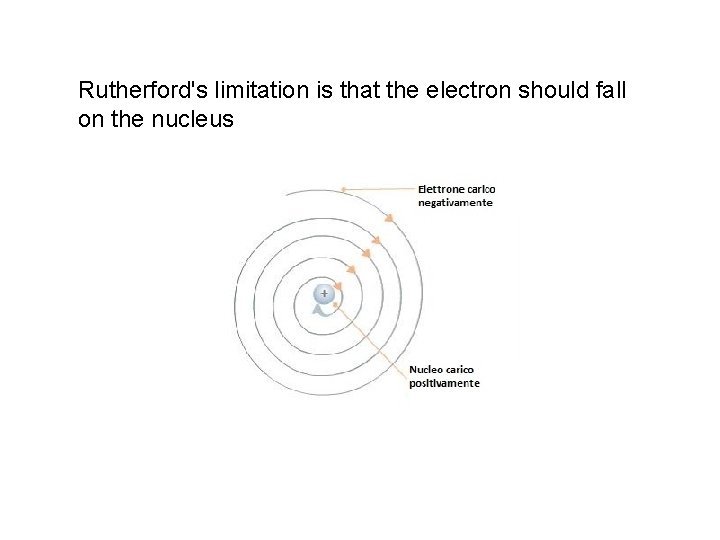
Rutherford's limitation is that the electron should fall on the nucleus

Bohr’s atomic model (1913) Bohr observed that the emission spectra of the chemical elements are different from each other, and analyzed the spectrum of hydrogen
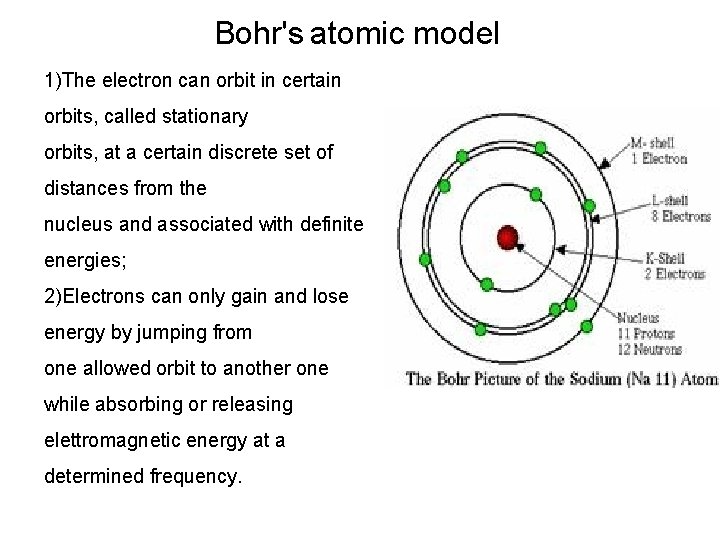
Bohr's atomic model 1)The electron can orbit in certain orbits, called stationary orbits, at a certain discrete set of distances from the nucleus and associated with definite energies; 2)Electrons can only gain and lose energy by jumping from one allowed orbit to another one while absorbing or releasing elettromagnetic energy at a determined frequency.
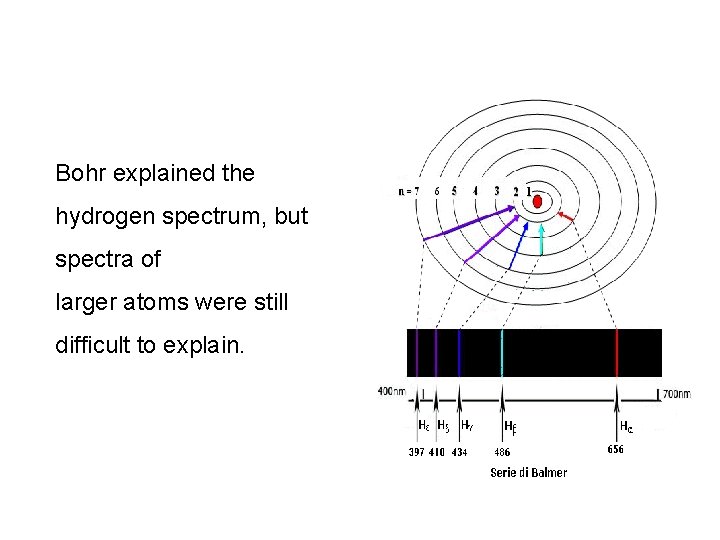
Bohr explained the hydrogen spectrum, but spectra of larger atoms were still difficult to explain.
- Slides: 20