atomic excitation and ionisation a mad scientists guide







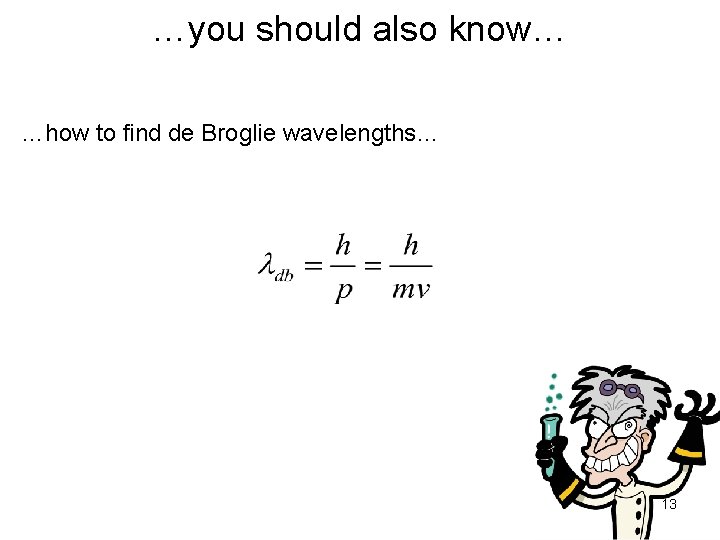






- Slides: 19

atomic excitation and ionisation …a mad scientist’s guide… 1

…so what do we need to know. . . ? • about quantisation of electron orbitals • about energy levels • how excitation & ionisation work • what a line spectrum is 2

…so what do we need to know. . . ? • how to calculate the frequencies of the photons emitted by transitions • how to convert between electron-volts and Joules 3

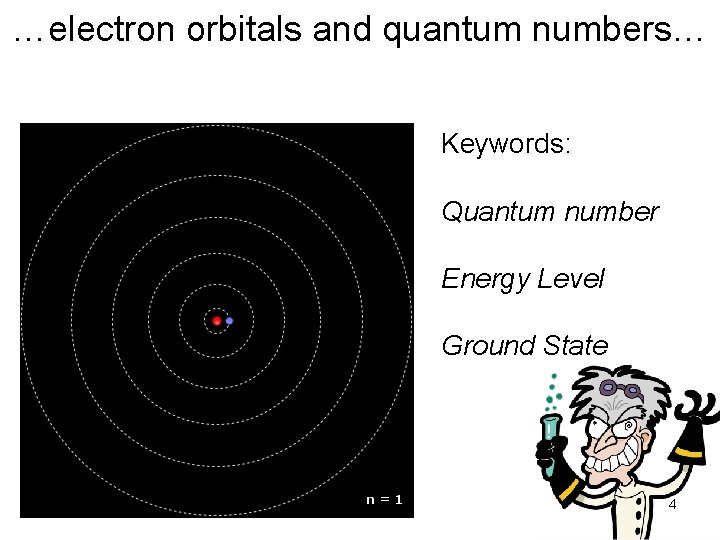
…electron orbitals and quantum numbers… Keywords: Quantum number Energy Level Ground State 4

…electron orbitals and quantum numbers… Keywords: Excited State 5

…collisions (or absorption of a photon) can lead to excitation… …and then subsequent relaxation leads to the emission of a photon. 6

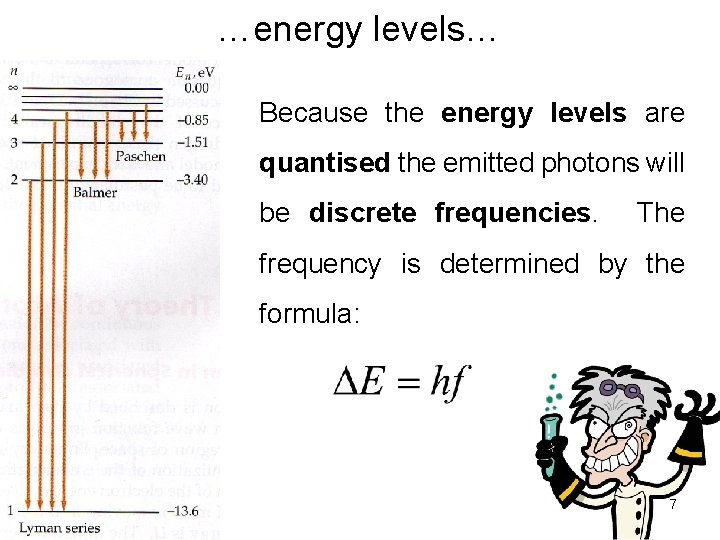
…energy levels… Because the energy levels are quantised the emitted photons will be discrete frequencies. The frequency is determined by the formula: 7

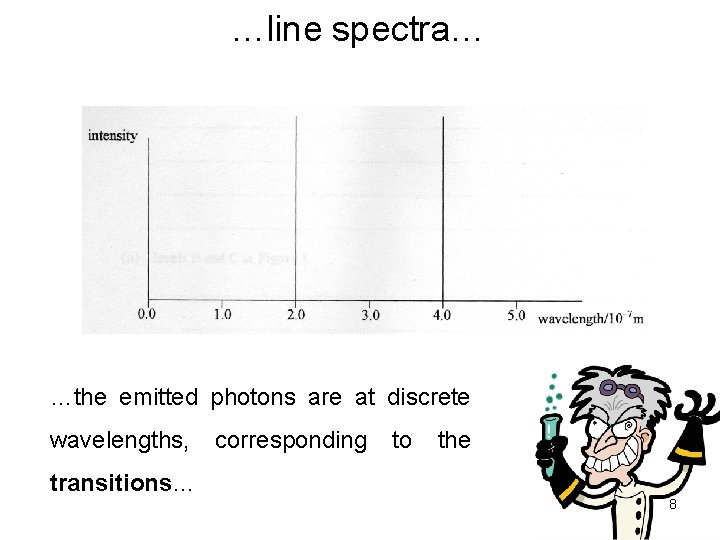
…line spectra… …the emitted photons are at discrete wavelengths, transitions… corresponding to the 8

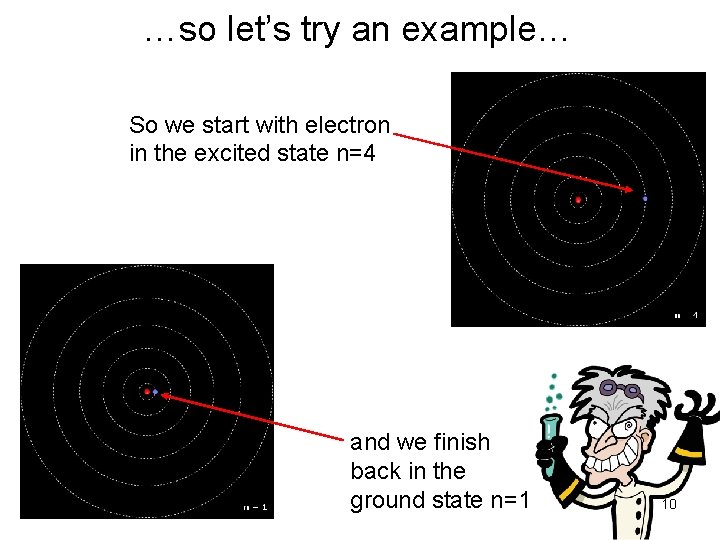
…so let’s try an example… Consider… …an excited hydrogen atom with quantum number n=4 relaxes to the ground state (n=1). What will be the wavelength of the emitted photon…? 9

…so let’s try an example… So we start with electron in the excited state n=4 and we finish back in the ground state n=1 10

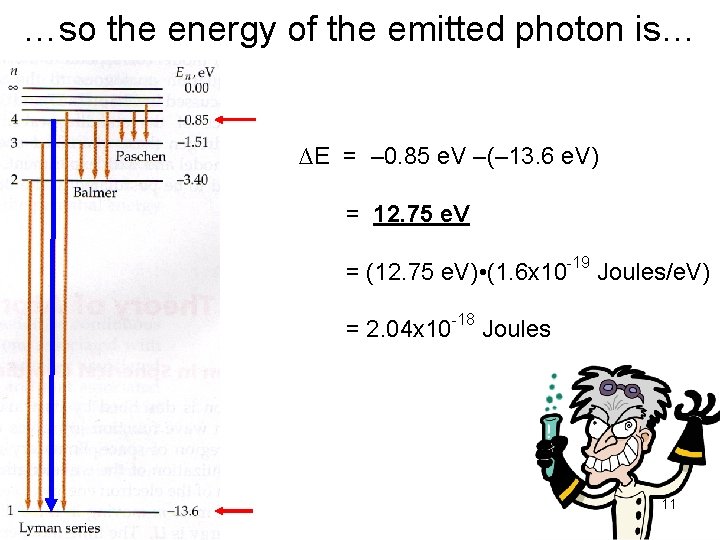
…so the energy of the emitted photon is… ∆E = – 0. 85 e. V –(– 13. 6 e. V) = 12. 75 e. V = (12. 75 e. V) • (1. 6 x 10 -19 Joules/e. V) = 2. 04 x 10 -18 Joules 11

…and so… …we use the formula …and find… (…or 97. 5 nm or 975Å or…) 12
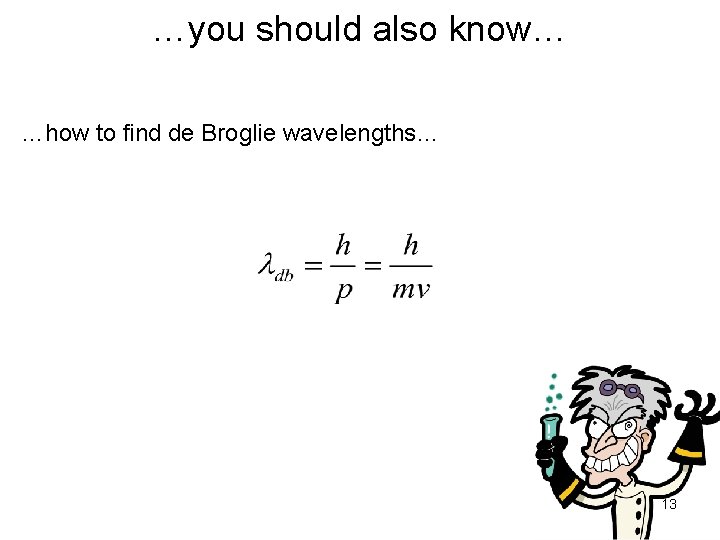
…you should also know… …how to find de Broglie wavelengths… 13

…so a quick recap… • ground state – when the electron is in its lowest orbital, with quantum number n=1 • excitation – when an electron is ‘kicked’ into a higher orbital by a collision • relaxation – when the electron drops down to a lower energy level, or back to ground state 14

…so a quick recap… • ionisation – when an atom completely loses an electron due to a collision • energy level – the energy associated with the quantum state of the atom • line spectrum – a graph showing the wavelength or frequency of the emitted photons 15

…so a quick recap… • electron-volt (e. V) – a more practical unit of energy for working on these scales. - to convert Joules to e. V we just multiply by the electronic charge - to convert e. V to Joules we just divide by the electronic charge 16

…different approaches to the questions… • determine transition from wavelength of emitted radiation • determine transition and values of energy levels from energy of emitted radiation • find ionisation energy • find e. g. longest wavelength 17

…different approaches to the questions… • determine all frequencies of radiation that could be emitted given collision energy • label transitions on diagram • calculate de Broglie Wavelength of incident particles • …and lots of wordy ones… 18

…so now it’s your turn… …you’ll need some formulae… …and some constants …so that’s it! enjoy…!!! 19