ATOMIC ELECTRON CONFIGURATIONS THE BOHR MODEL OF THE

ATOMIC ELECTRON CONFIGURATIONS

THE BOHR MODEL OF THE ATOM I pictured the electrons orbiting the nucleus much like planets orbiting the sun. Niels Bohr (O. G) However, electrons are found in specific circular paths around the nucleus, and can jump from one level to another.
ERNEST RUTHERFORD’S MODEL WAS A BUNCH OF BUNK • It did not explain the chemical properties of the elements – a better description of the electron behavior was needed That’s a bunch of crap

THE QUANTUM MECHANICAL MODEL • In 1926, Erwin Schrodinger created the new model to determine where electrons were located in an atom.

THE REAL ATOM • Organized into energy levels. Denoted with numbers 1 -7. • As the energy level goes up, the electrons have more energy • Electrons found in “clouds” where electrons can be found. • Denoted with letters; s, p, d, and f for the shape of the cloud.

1 ST RULE OF ELECTRON FILLING: • Orbitals are filled from the lowest energy up. Copy this mess --> • 1 st energy level: 2 • 2 nd energy level: 8 • 3 rd energy level: 18 • 4 th energy level: 32 • Etc…: 32
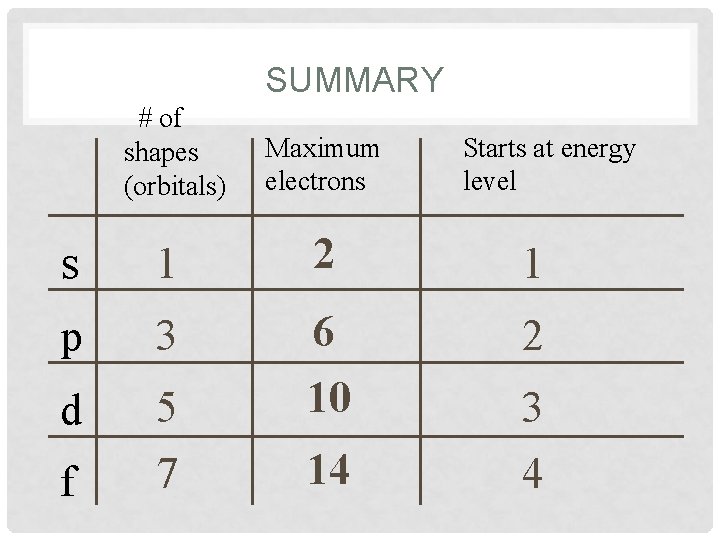
SUMMARY # of shapes (orbitals) Maximum electrons s 1 2 1 p 3 2 d 5 7 6 10 f 14 Starts at energy level 3 4

SECOND RULE OF ELECTRON FILLING: • No two things can occupy the same space at the same time. • The reason why the arrows are drawn one up and one down in the orbital box diagram
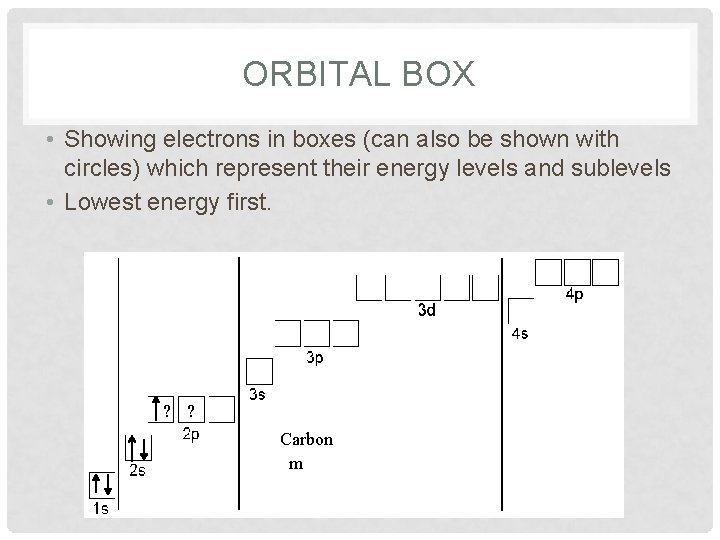
ORBITAL BOX • Showing electrons in boxes (can also be shown with circles) which represent their energy levels and sublevels • Lowest energy first. ? ? Carbon Boron Berylliu Hydrogen Lithium Helium m

3 RD RULE OF ELECTRON FILLING: • The most stable arrangement of electrons is that with the maximum number of unpaired electrons. 3 electrons in the 2 p orbital would look like Not

LETS PRACTICE THIS JUNK • Use spdf notation and orbital box. • B • O • Ga • K • Mg
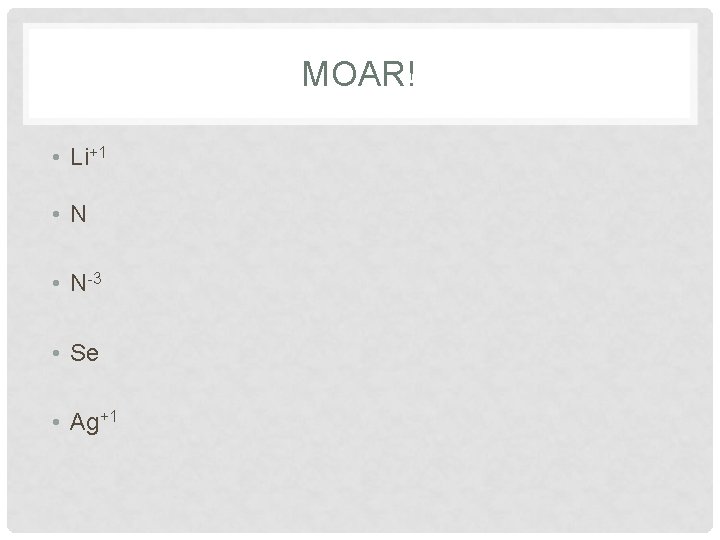
MOAR! • Li+1 • N-3 • Se • Ag+1
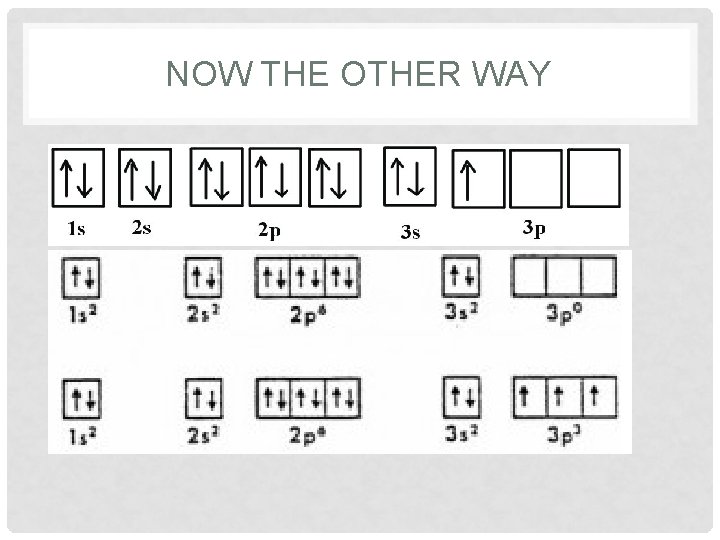
NOW THE OTHER WAY

NOBLE GAS CONFIGURATION • We can use a shortcut to show configurations by using the noble gasses. • Instead of filling the whole list, we start at a noble gas and work from there. • To do Calcium, we would start from Argon which is 3 p 6. So calcium would be [Ar]4 s 2 • 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 • [1 s 2 2 p 6 3 s 2 3 p 6] 4 s 2 • Look at the practice problems we already did together and turn them into noble gas config.

SUMMARIZE WHAT YOU LEARNED • What did you learn about electrons this lecture? • What are the two models of the atom and how are they different? • Summarize on your own.
- Slides: 15