ATOMIC BONDING FORMING COMPOUNDS AND MOLECULES PART 2

ATOMIC BONDING: FORMING COMPOUNDS AND MOLECULES PART 2

Why do atoms bond? Just like people, all atoms have a goal of being “complete” (stable) They do this by completely filling OR emptying their outer shell of electrons

Outer Electron Shells Atom with a full shell Atom with neither 0 nor 8 electrons Atom with an empty shell The electrons in the outer shell of an atom are called valence electrons. Atoms are most stable when they obtain 8 valence electrons (a full shell) OR 0 valence electrons (an empty shell)

A little more detail… https: //www. youtube. com/watch? v=OTgp. N 6 2 ou 24

Full vs. Empty Which atoms. Shells prefer to have a full shell, and which prefer empty? Atoms from elements in groups 1, 2, and 3 prefer to have an empty shell Atoms from groups 4, 5, 6, 7, and 8 prefer to have a full shell Prefer to be empty (0 valence electrons) E R O N IG SE E H T Prefer to be full (8 valence electrons)

Valence Electron Practice #1 Can you count how many valence electrons are in the atom to the right? Would this atom prefer to have 0 electrons in its outer shell, or 8?

Valence Electron Practice #2 Can you count how many valence electrons are in the atom to the right? Would this atom prefer to have 0 electrons in its outer shell, or 8?

Valence Electron Practice #3 Can you count how many valence electrons are in the atom to the right? Would this atom prefer to have 0 electrons in its outer shell, or 8?

Valence Electron Practice #4 Can you count how many valence electrons are in the atom to the right? Would this atom prefer to have 0 electrons in its outer shell, or 8?

Bonds hold atoms together They come in two forms: Ionic and Covalent

Dogs Teaching Chemistry! https: //www. youtube. com/watch? v=_M 9 khs 87 x. Q 8

Ionic Bonds • One atom takes an electron from a second atom • Usually occurs between a metal and a non-metal

Ionic Bonds • Creates two ions • The atom that lost an electron becomes a positive ion • The atom that gained an electron becomes a negative ion
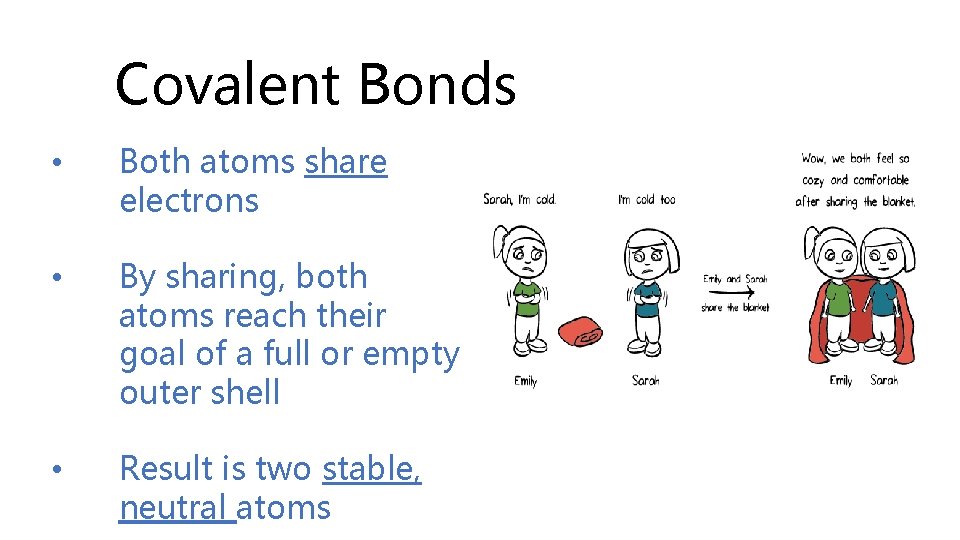
Covalent Bonds • Both atoms share electrons • By sharing, both atoms reach their goal of a full or empty outer shell • Result is two stable, neutral atoms

Non-Bonding Atoms • Some atoms do not make bonds with others • These atoms already have a full shell of 8 valence electrons • All of the elements in the LAST group on the periodic table have fulls shells, so they do not react or form bonds – these are called Noble Gases
- Slides: 15