Atomic Absorption Spectroscopy Dr D Sweetlin Rajula Rubavathi

Atomic Absorption Spectroscopy Dr. D. Sweetlin Rajula Rubavathi, Department of Chemistry, Loyola College, Chennai.

Atomic Spectroscopy Introduction Based on the breakdown of a sample into atoms, followed by the measurement of the atom’s absorption or emission of light. i. deals with absorbance or emission (luminescence) of atoms or elemental ions rather then molecules - atomization: process of converting sample to gaseous atoms or elementary ions ii. Provides information on elemental composition of sample or compound - UV/Vis, IR, Raman gives molecular functional group information, but no elemental information. iii. Basic process the same as in UV/Vis, fluorescence etc. for molecules Absorbance Fluorescence

Sample Atomization i. Need to break sample into atoms to observe atomic spectra ii. Basic steps: a) nebulization – solution sample, get into fine droplets by spraying thru thin nozzle b) desolvation - heat droplets to evaporate off solvent just leaving analyte and other matrix compounds c) volatilization – convert solid analyte into gas phase d) dissociation – break-up molecules in gas phase into atoms. e) ionization – cause the atoms to become charged f) excitation – with light, heat, etc. for spectra measurement.

PRINCIPLE Atomic absorption spectroscopy (AAS) is an analytical technique that measures the concentrations of elements. It makes use of the absorption of light by these elements in order to measure their concentration. Atomic-absorption spectroscopy quantifies the absorption of ground state atoms in the gaseous state. - The atoms absorb ultraviolet or visible light and make transitions to higher electronic energy levels. The analyte concentration is determined from the amount of absorption.

The Atomic Absorption Spectrometer Atomic absorption spectrometers have 4 principal components 1 - A light source ( usually a hollow cathode lamp ) 2 – An atom cell ( atomizer ) 3 - A monochromator 4 - A detector , and read out device.
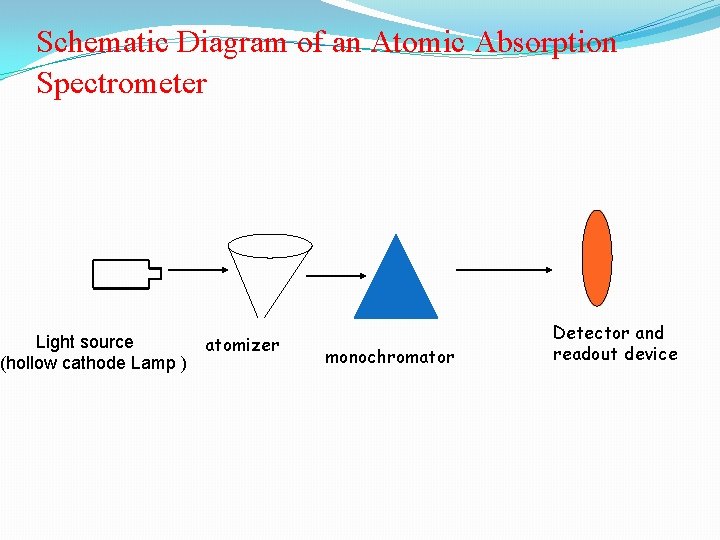
Schematic Diagram of an Atomic Absorption Spectrometer Light source (hollow cathode Lamp ) atomizer monochromator Detector and readout device

Light Source �The light source is usually a hollow cathode lamp of the element that is being measured. It contains a tungsten anode and a hollow cylindrical cathode made of the element to be determined. These are sealed in a glass tube filled with an inert gas (neon or argon ). Each element has its own unique lamp which must be used for that analysis.
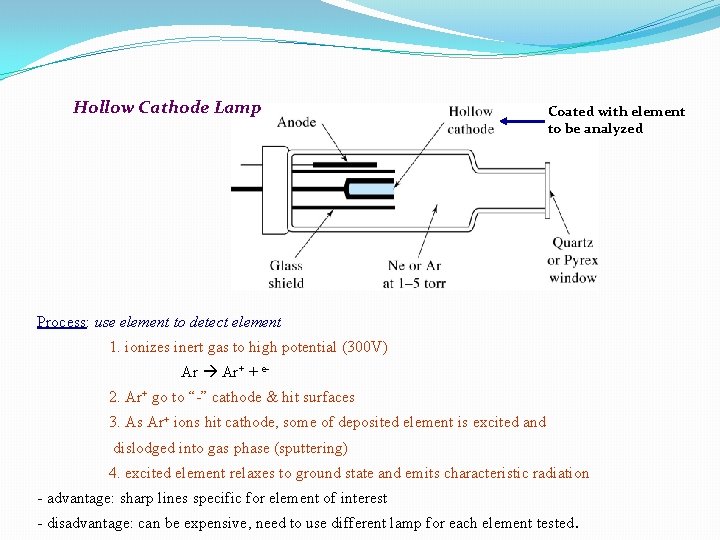
Hollow Cathode Lamp Coated with element to be analyzed Process: use element to detect element 1. ionizes inert gas to high potential (300 V) Ar Ar+ + e 2. Ar+ go to “-” cathode & hit surfaces 3. As Ar+ ions hit cathode, some of deposited element is excited and dislodged into gas phase (sputtering) 4. excited element relaxes to ground state and emits characteristic radiation - advantage: sharp lines specific for element of interest - disadvantage: can be expensive, need to use different lamp for each element tested.

Atomizer Elements to be analyzed needs to be in atomic sate Atomization is separation of particles into individual molecules and breaking molecules into atoms. This is done by exposing the analyte to high temperatures in a flame or graphite furnace.

The role of the atom cell(atomizer) is to primarily dissolvate a liquid sample and then the solid particles are vaporized into their free gaseous ground state form. In this form atoms will be available to absorb radiation emitted from the light source and thus generate a measurable signal proportional to concentration. There are two types of atomization : Flame and Graphite furnace atomization.
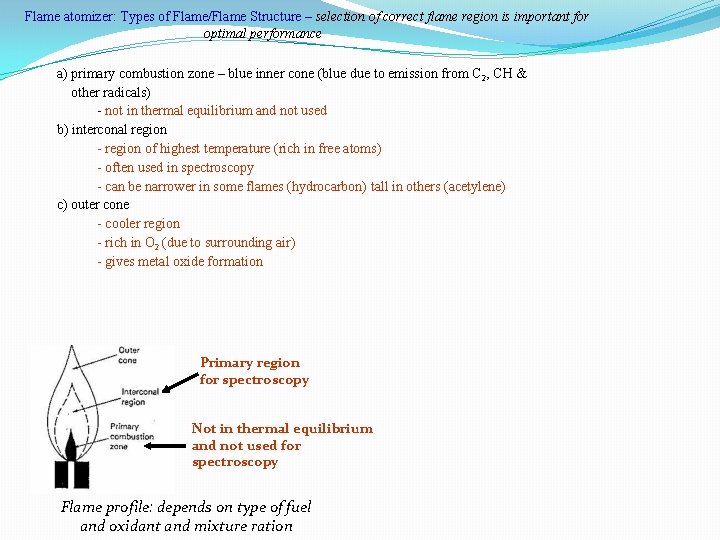
Flame atomizer: Types of Flame/Flame Structure – selection of correct flame region is important for optimal performance a) primary combustion zone – blue inner cone (blue due to emission from C 2, CH & other radicals) - not in thermal equilibrium and not used b) interconal region - region of highest temperature (rich in free atoms) - often used in spectroscopy - can be narrower in some flames (hydrocarbon) tall in others (acetylene) c) outer cone - cooler region - rich in O 2 (due to surrounding air) - gives metal oxide formation Primary region for spectroscopy Not in thermal equilibrium and not used for spectroscopy Flame profile: depends on type of fuel and oxidant and mixture ration
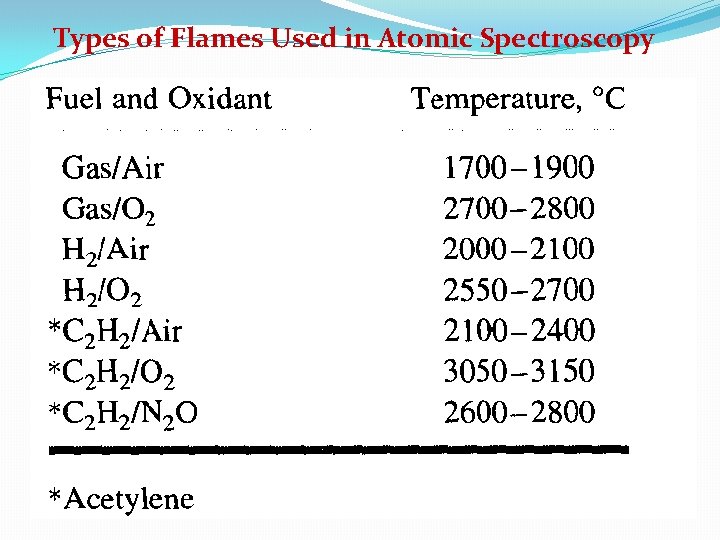
Types of Flames Used in Atomic Spectroscopy

Electrothermal (Graphite furnace) Atomizer : consist of a hollow graphite cylinder through which the radiation ( from hollow-cathode tube) passes • electrodes at the end of the cylinder are connected to power supply which can deliver up to 3. 6 k. W to the cylinder wall • liquid sample are injected with a microsyringe through the small opening in the top of cylinder • solid samples can be introduced through the end of the tube with a special sampling spoon
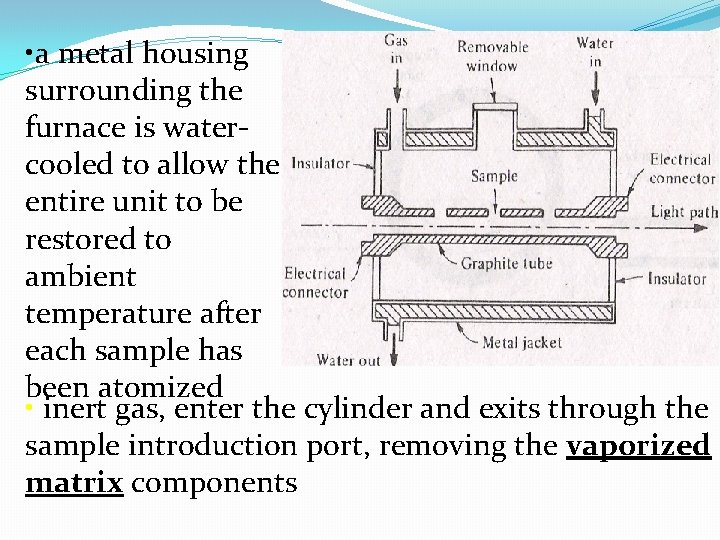
• a metal housing surrounding the furnace is watercooled to allow the entire unit to be restored to ambient temperature after each sample has been atomized • inert gas, enter the cylinder and exits through the sample introduction port, removing the vaporized matrix components

Advantages and Disadvantages of Flame atomizer �Advantages �equipment relatively cheap �easy to use (training easy compared to furnace) �good precision �high sample throughput �relatively facile method development �cheap to run �Disadvantages �lack of sensitivity (compared to furnace) �problems with refractory elements �require large sample size �sample must be in solution 15

Advantages and Disadvantages of Electrothermal Atomisation �Advantages �very sensitive for many elements �small sample size �Disadvantages �poor precision �long cycle times means a low sample throughput �expensive to purchase and run (argon, tubes) �requires background correction �method development lengthy and complicated �requires a high degree of operator skill (compared to flame AAS) 16

�Burner-Premix and total consumption burner �Premix-sample is aspirated into a large chamber by a stream of oxidant –fine mist of sample, oxidant, fuel – forced to burner opening �Suitable for IA, IB, IIB �Total Consumption burner-sample solution, fuel, oxidant enter through separate passage and meet at an opening at the base of the flame.

Monochromators This is a very important part in an AA spectrometer. It is used to separate out all of the thousands of lines. Without a good monochromator, detection limits are severely compromised. A monochromator is used to select the specific wavelength of light which is absorbed by the sample, and to exclude other wavelengths. The selection of the specific light allows the determination of the selected element in the presence of others.

Chopper �In order to eliminate the unwanted emissions from the flame, the light source is modulated by a chopper which is located between the hollow cathode lamp and the flame. The amplifier which modifies the signal from the photomultiplier is tuned in to the same frequency.

Detector and Read out Device The light selected by the monochromator is directed onto a detector that is typically a photomultiplier tube , whose function is to convert the light signal into an electrical signal proportional to the light intensity. The processing of electrical signal is fulfilled by a signal amplifier. The signal could be displayed for readout , or further fed into a data station for printout by the requested format.

Calibration Curve A calibration curve is used to determine the unknown concentration of an element in a solution. The instrument is calibrated using several solutions of known concentrations. The absorbance of each known solution is measured and then a calibration curve of concentration vs absorbance is plotted. The sample solution is fed into the instrument, and the absorbance of the element in this solution is measured. The unknown concentration of the element is then calculated from the calibration curve
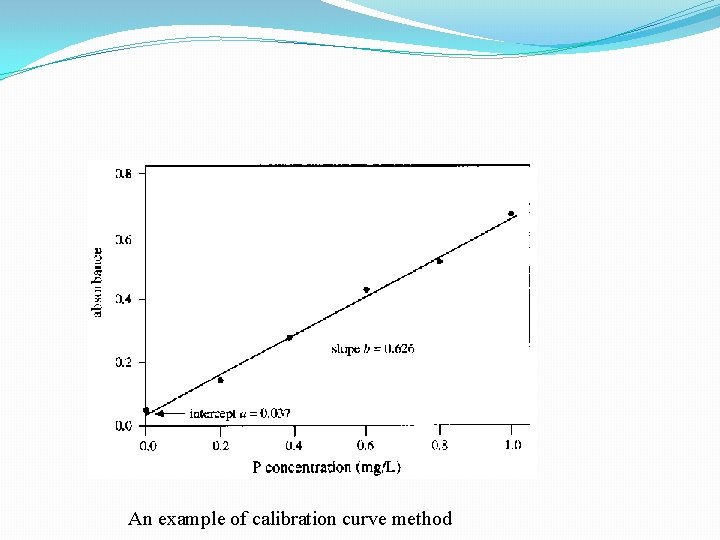
An example of calibration curve method
- Slides: 22