Atomic absorption spectroscopy AAS Flame atomic absorption spectroscopy

Atomic absorption spectroscopy (AAS)



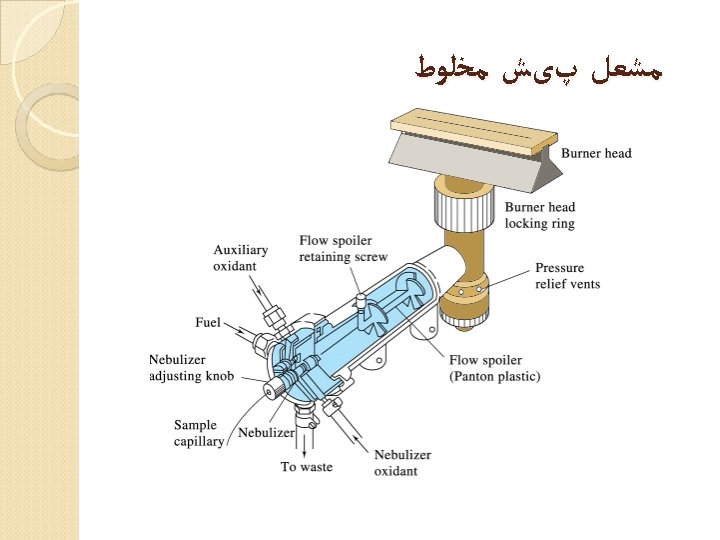
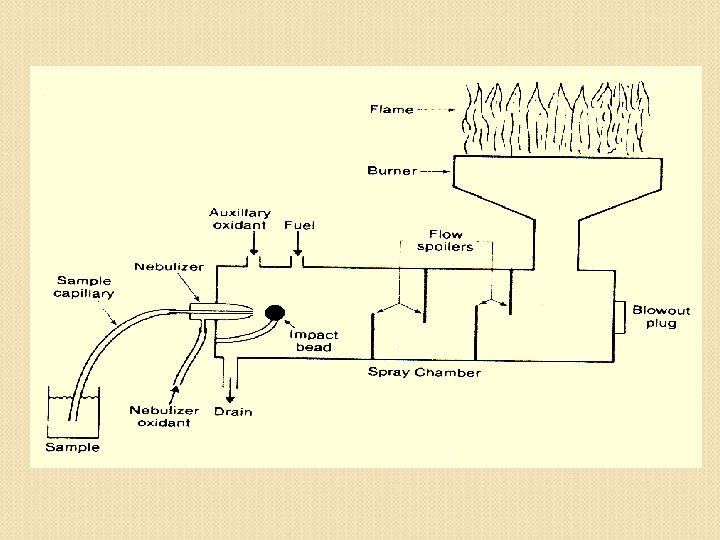

Flame atomic absorption spectroscopy
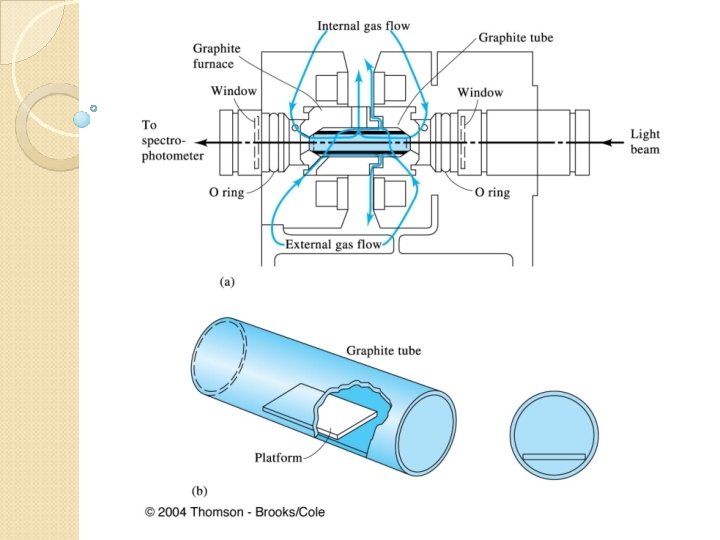
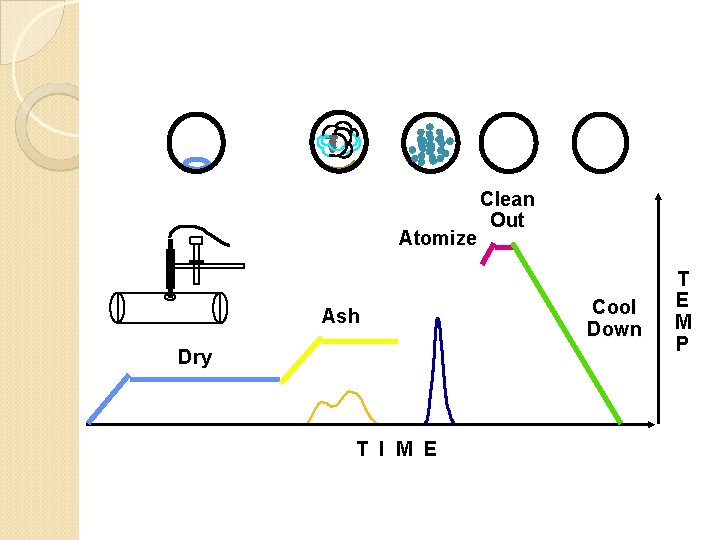
Atomize Ash Dry T I M E Clean Out Cool Down T E M P

Graphite furnace tube

Chemical modifier
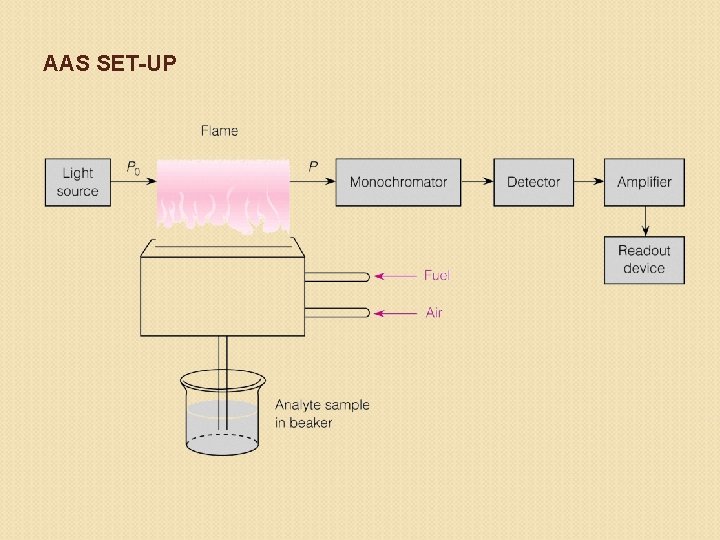
AAS SET-UP
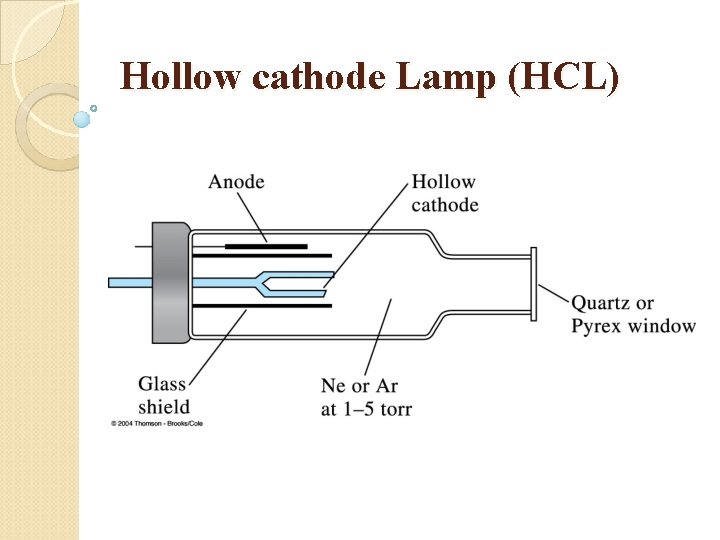
Hollow cathode Lamp (HCL)
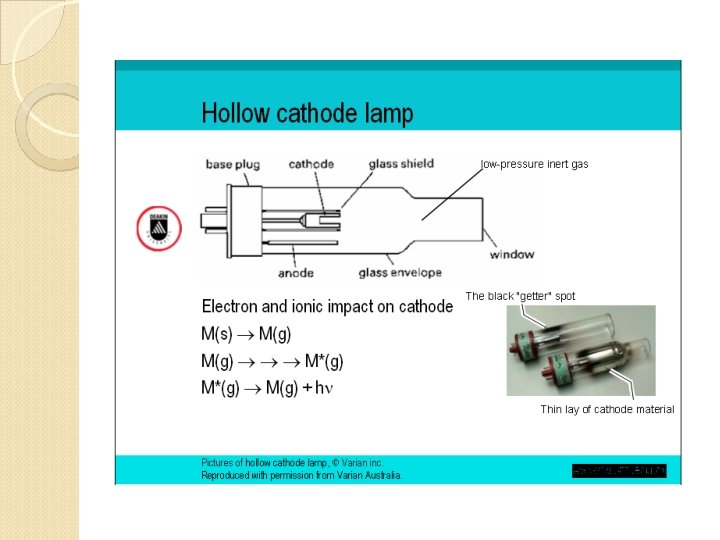

A hollow cathode lamp for Aluminum (Al)
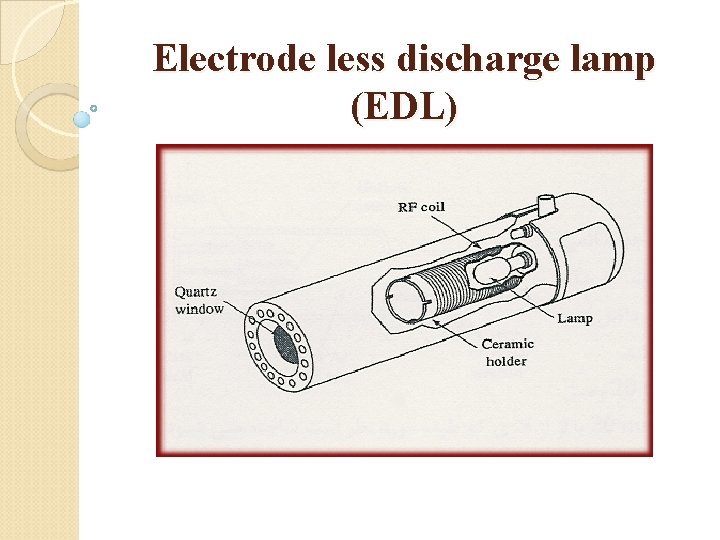
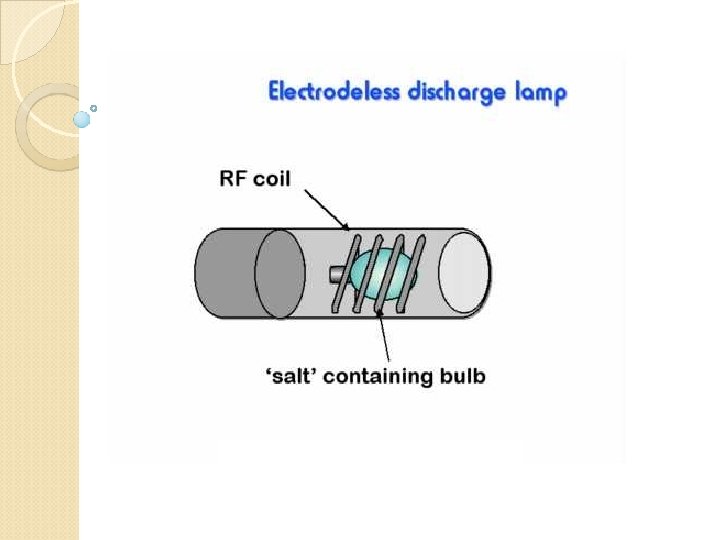
Electrode less discharge lamp (EDL)

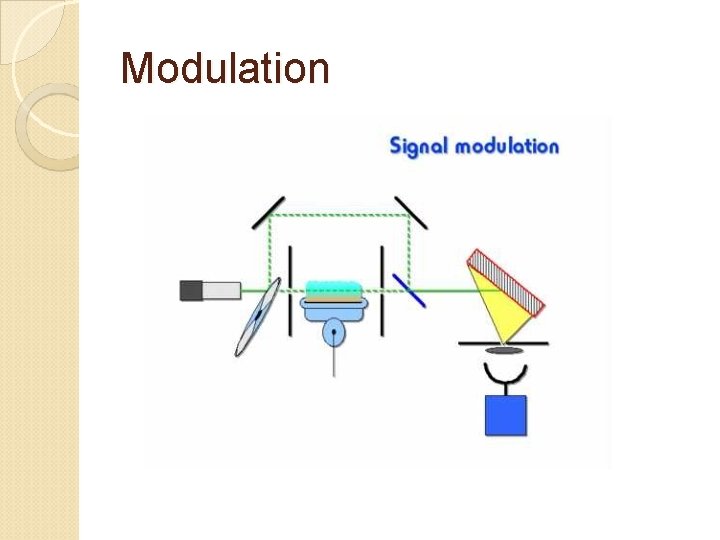
Modulation

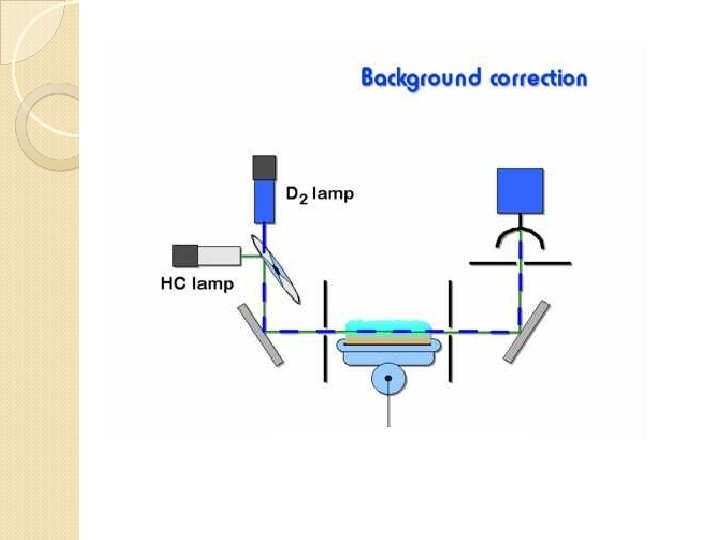
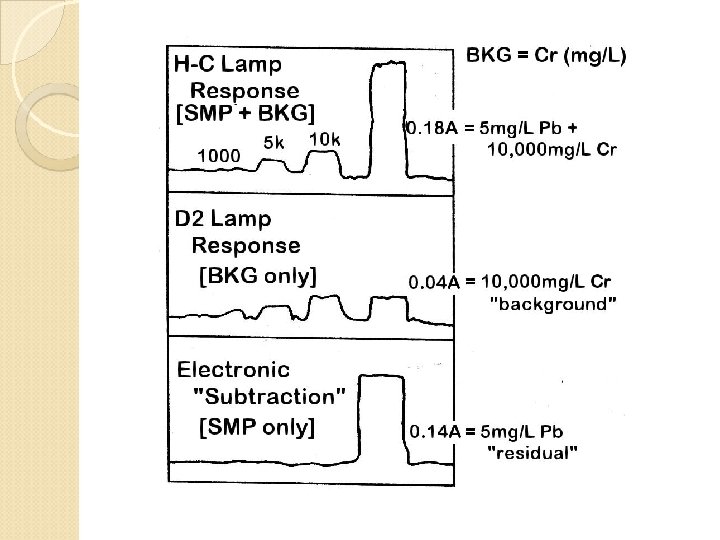
Interferences 1) Spectral interference 2) Chemical interference

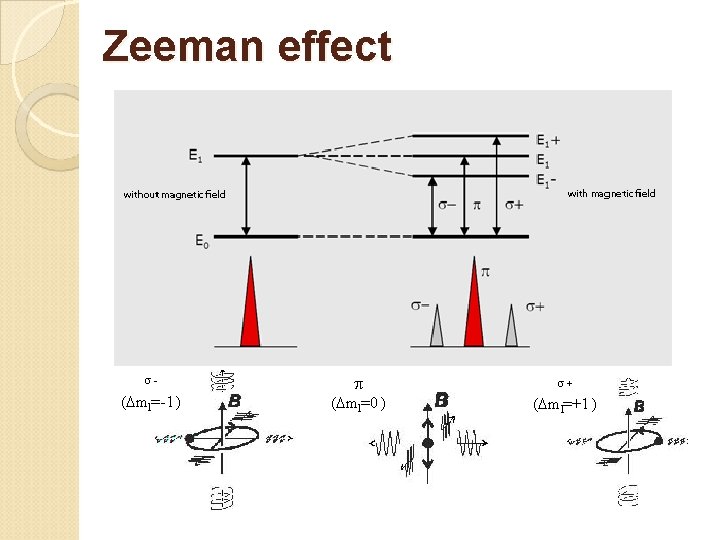
Zeeman effect s- ( ml=-1 ) ( ml=0 ) s+ ( ml=+1 )


Cold vapor generation
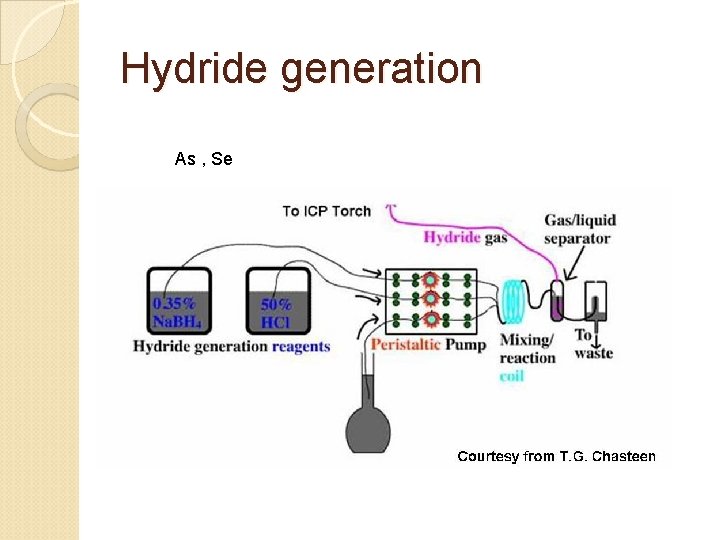
Hydride generation As , Se
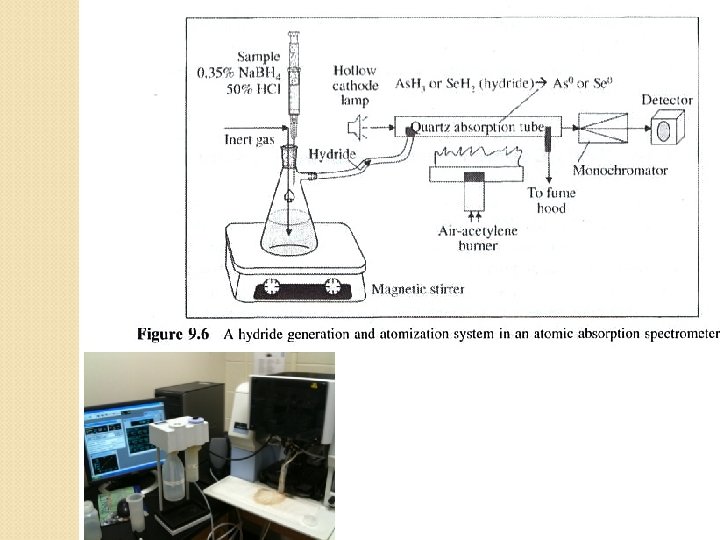

Metals Analysis

Wet Digestion Methods The common methods used for dissolving samples for metals analysis are : Digestion in open flask Digestion in a pressurized sealed container Microwave assisted decomposition
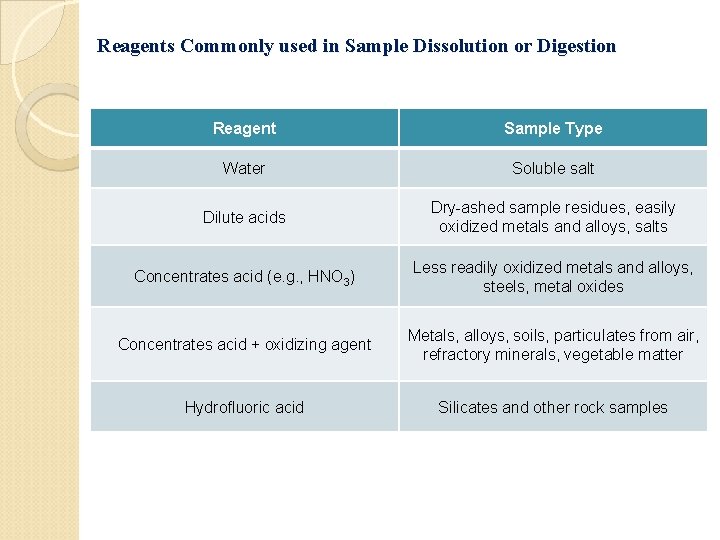
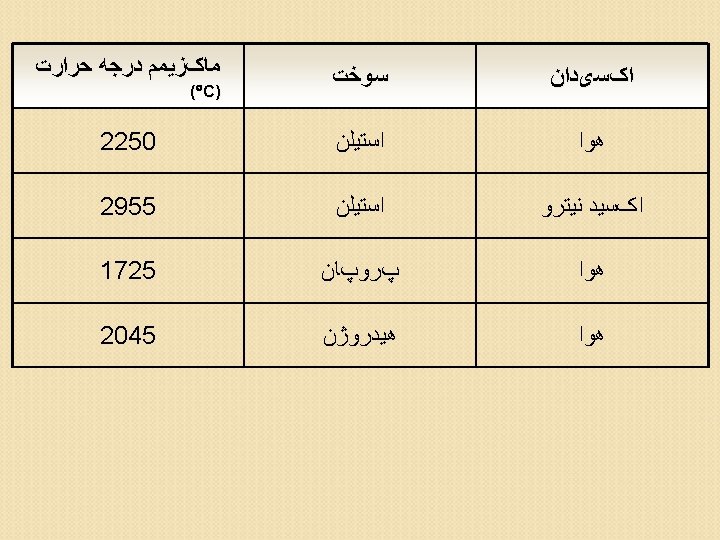
Reagents Commonly used in Sample Dissolution or Digestion Reagent Sample Type Water Soluble salt Dilute acids Dry-ashed sample residues, easily oxidized metals and alloys, salts Concentrates acid (e. g. , HNO 3) Less readily oxidized metals and alloys, steels, metal oxides Concentrates acid + oxidizing agent Metals, alloys, soils, particulates from air, refractory minerals, vegetable matter Hydrofluoric acid Silicates and other rock samples

Acid Digestion-Wet Ashing The simplest method for wet digestion is carried out in an open container. Samples are dried, weighed, and placed in a beaker. The digestion reagent is added. The beaker is covered with a watch glass and placed on a hot plate.
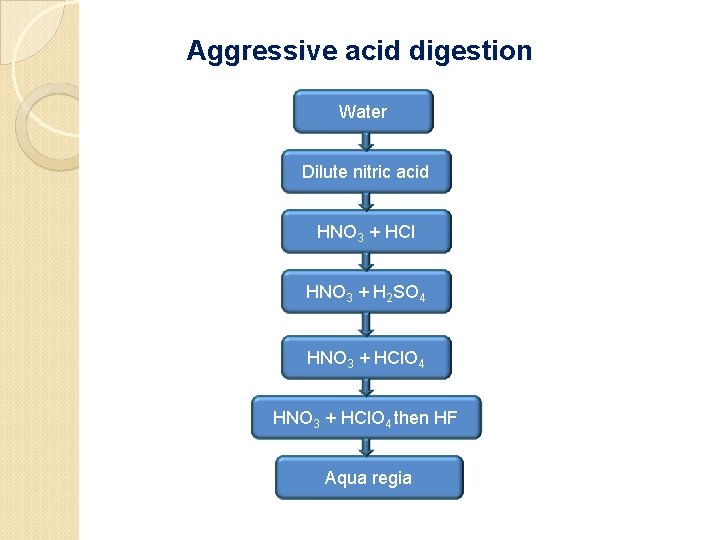
Aggressive acid digestion Water Dilute nitric acid HNO 3 + HCl HNO 3 + H 2 SO 4 HNO 3 + HCl. O 4 then HF Aqua regia

For samples that show significant losses of analyte due to the retention of metals in silica residues, the sample is first digested thoroughly with HNO 3 in a PTFE beaker. Then concentrated HCl. O 4 and a small amount of HF are added. A mixture of an oxidizing acid with HF provides acidity, oxidizing power, and complexation to dissolve all metals and alloys and most refractory minerals, soils, rocks, and sediments.

Microwave Digestion Advantages of digestion in a closed container : • The containers are fabricated of high-temp polymers, which are less likely to contain metal contaminants than are glass or ceramic beakers or crucibles. • The sealed container eliminates the chance of airborne dust contamination. • The sealed container reduce evaporation, so that less acid digestion solution is required. • The sealed container also eliminates losses of more volatile metal species, which can be a problem in open container. • The electronic controls on modern microwave digesters allow very reproducible digestion conditions.

Dry Ashing For samples that contain much organic matter, which are analyzed for nonvolatile metals, dry ashing is a relatively simple method of removing the organic matter. A general procedure is to place the weighed sample into a platinum or glass crucible and heat it in a furnace to a white ash. The temperature should be kept at 400 to 450°C if any of the more volatile metals are being determined. Salts or sulfuric acid may be added, if needed, and a final ashing step can be done with HF if required. The residue is then dissolved in concentrated nitric acid and water, and diluted to volume. The final concentration of acid should be between 1 and 5%.

Extraction, Separation, and concentration It is not always necessary or required to digest the entire sample in order to free the metal for analysis. The analyte of interest may be present as a soluble salt from a pollution source or being present in the structure of the mineral crystals. üOrganic extraction of metals (LLE) üExtraction with Supercritical Fluids üUltrasonic sample preparation üSolid-Phase Extraction for preconcentration
Structures of some chelating agents

Colorimetric Methods Fairly rapid and simple analyses can be performed on solutions using a variety of colorimetric reagents. These are reagents that are more or less specific for certain metals and will produce a solution, usually colored, whose absorbance at a particular wavelength is related to the concentration of the analyte. Preparation of samples for colorimetric analysis : • Buffering or p. H adjustment of sample solution • Oxidize or reduce the analyte to bring it to the correct oxidation state • Add color-forming reagent • Using specific condition of temp. or time to ensure complete reaction
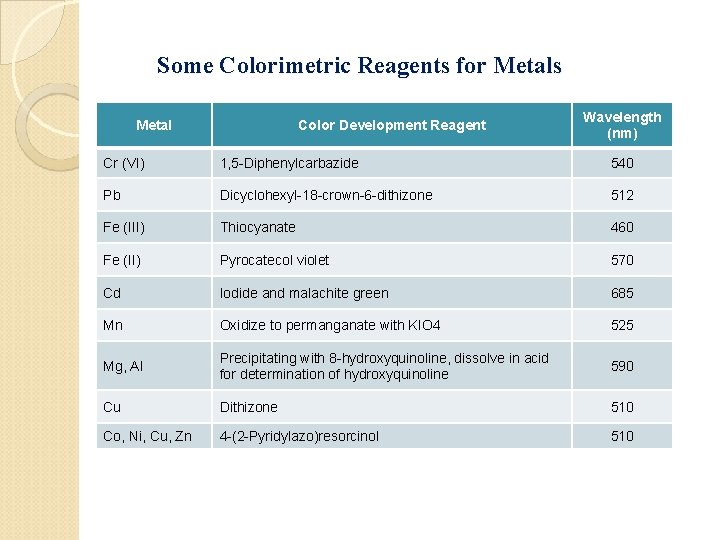
Some Colorimetric Reagents for Metals Metal Color Development Reagent Wavelength (nm) Cr (VI) 1, 5 -Diphenylcarbazide 540 Pb Dicyclohexyl-18 -crown-6 -dithizone 512 Fe (III) Thiocyanate 460 Fe (II) Pyrocatecol violet 570 Cd Iodide and malachite green 685 Mn Oxidize to permanganate with KIO 4 525 Mg, Al Precipitating with 8 -hydroxyquinoline, dissolve in acid for determination of hydroxyquinoline 590 Cu Dithizone 510 Co, Ni, Cu, Zn 4 -(2 -Pyridylazo)resorcinol 510

Calibration Curves The common calibration method is to prepare standards of known concentrations, covering the concentration rang expected in the sample. The matrix standard should be as close to the samples as possible. If the sample is to be extracted into certain organic solvent, the standards should be prepared in the same solvent. The calibration curve is a plot of detector response as a function of concentration
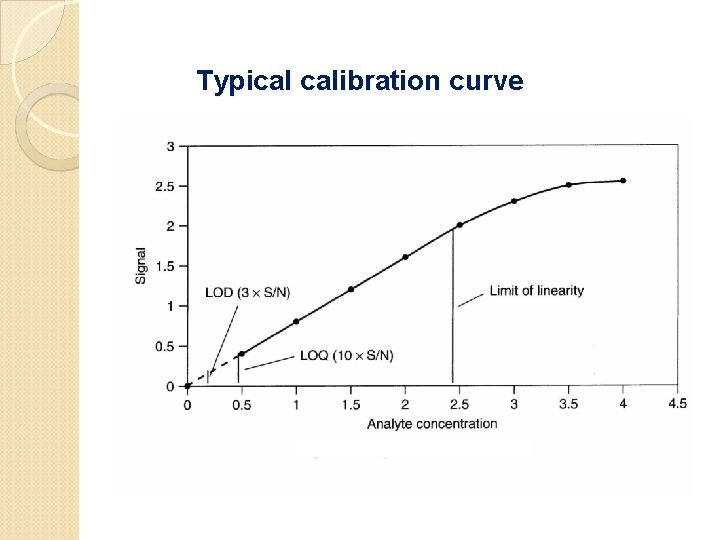
Typical calibration curve

The calibration can be done in two ways • 1) Measuring the amount of Pb in soil : Sample preparation by acid extraction followed by analysis using atomic absorption. The standards can be made by spiking clean soil with known quantities of Pb. Then the standards are taken through the entire process of extraction and analysis. Finally, the instrument response is plotted as a function of concentration. 2) The other option assumes quantitative extraction, and the standards are used to calibrate only the AA. The first approach is more accurate; the latter is simpler. A calibration method that takes the matrix effects into account is the method of standard addition.

Standard addition methods
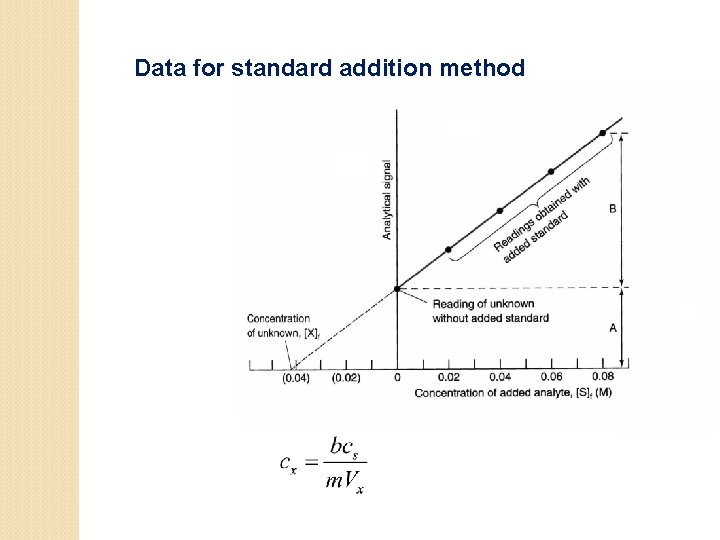
Data for standard addition method
- Slides: 49