ATOMIC ABSORPTION SPECTROMETER SELN CANSU ZTRK EYMA ATAKUL

ATOMIC ABSORPTION SPECTROMETER SELİN CANSU ÖZTÜRK ŞEYMA ATAKUL SEZİN GÜNER

ATOMIC ABSORPTION SPECTROMETER �Introduction �Invention �Working Principle of AAS �Instrumentation �Interferences & Correlation Methods �Applications

INVENTION �Introduced in 1955 by Alan Walsh in Australia �Firstly used for mining, medical treatment&agriculture �Alan Walsh(1916 -1998) http: //www. science. org. au/academy/memoi rs/walsh 2. html

PROPERTIES OF AAS �The most widely used method in analysis of elements �Based on the absorption of radiation �So sensitive (ppb) �Quantitative analysis

WORKING PRINCIPLE OF AAS �Electrons promote to higher orbitals for a short amount of time by absorbing a energy �M + hv → M* �Relises on Beer-Lambert Law A= a. b. c
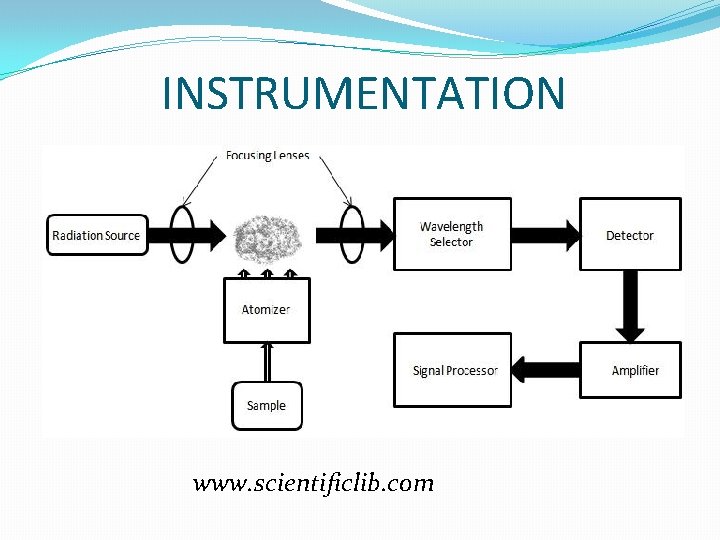
INSTRUMENTATION www. scientificlib. com
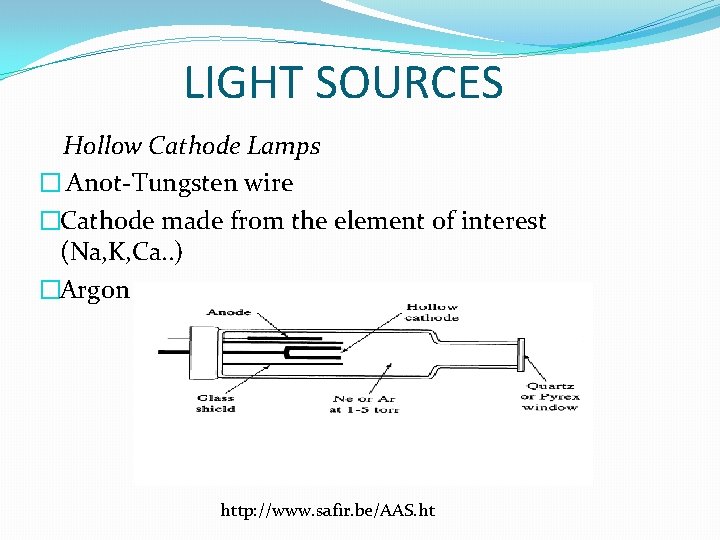
LIGHT SOURCES Hollow Cathode Lamps � Anot-Tungsten wire �Cathode made from the element of interest (Na, K, Ca. . ) �Argon or neon gas http: //www. safir. be/AAS. ht

LIGHT SOURCES Electrodeless Discharge Lamp �Typically argon gas at low pressure � Narrower line width �Not prefered www. freepatentsonline. com

ATOMIZATION �Compounds making up the sample are broken into free atoms. �High temperature is necessary �Basic two types -Flame atomizer -Electrothermal atomizer

TYPES OF ATOMIZERS FLAME ATOMIZER �Simplest atomization �Converts analyte into free atoms of vapor phase �Flammable &caustic gases �Not has an inert medium (−) �Short analysis time (−)

TYPES OF ATOMIZERS ELECTROTHERMAL ATOMIZER �A cylindirical graphite tube �Inert gas medium (Argon gas) �Longer anlaysis time than flame �Superior sensitivity, high accuracy

MONOCHROMATOR �Also it is called wavelengh selector �Select the specific wavelenght �Polychromatic light →monochromatic light �Simple one is enough for AAS

DETECTOR �Electromagnetic waves → electric current �The most used one Photomultiplier tube �Have fast response times www. answers. com

CALIBRATION TECHNIQUES • Two main techniques Ø Calibration curve method ØStandart addition method

CALIBRATION TECHNIQUES CALIBRATION CURVE METHOD �Draw a graph �Have two or more variables -One is set at known values -One is measured response �Most convenient for a large number of similar samples analysis.
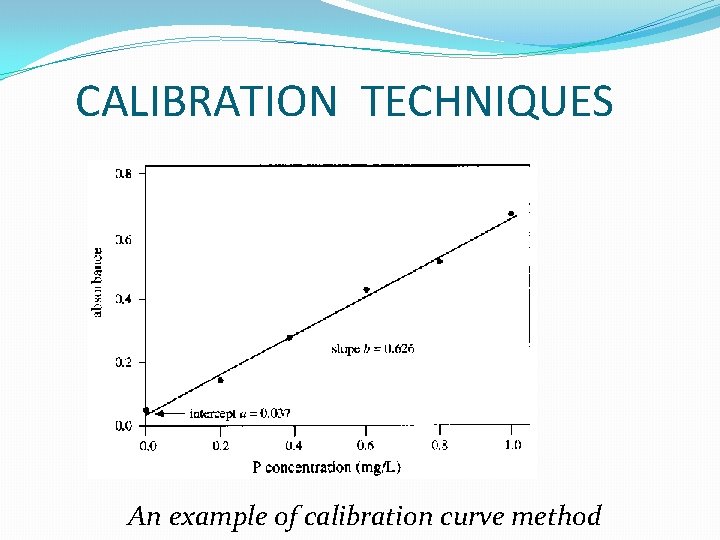
CALIBRATION TECHNIQUES An example of calibration curve method

CALIBRATION TECHNIQUES STANDART ADDITION METHOD �To measure the analyte concentration in a complex matrix. �Most convenient for small number of samples analysis �Prevent effect of chemical & spectral interferences

INTERFERENCES �Causes higher or lower absorbance value � Two major groups Ø Chemical Interferences Ø Spectral Interferences

CHEMICAL INTERFERENCES �The most common one in flame atomizer. �Consequence of chemical reactions. �Reduce amount of oxygen in flame to overcome

SPECTRAL INTERFERENCES �Absorption or emission of the radiation at the same wavelength �Radiation which is absorbed→pozitive errors �Radiation which is emmitted→negative errors

SPECTRAL CORRELATION METHODS TWO-LINE CORRELATION METHOD �Select two line Ø characteristic wavelength of analyte Ø very close to analyte line but not absorbed by analyte �Measure the difference between two lines

SPECTRAL CORRELATION METHODS CONTINUUM SOURCE CORRELATION METHOD �Select two lamps Ø Deuterium lamp & hollow cathode lamp �When hollow cathode lamp is used total absorbace is measured �When deuterium lamp is used only background absorption is measured �Measure the difference between two lines.
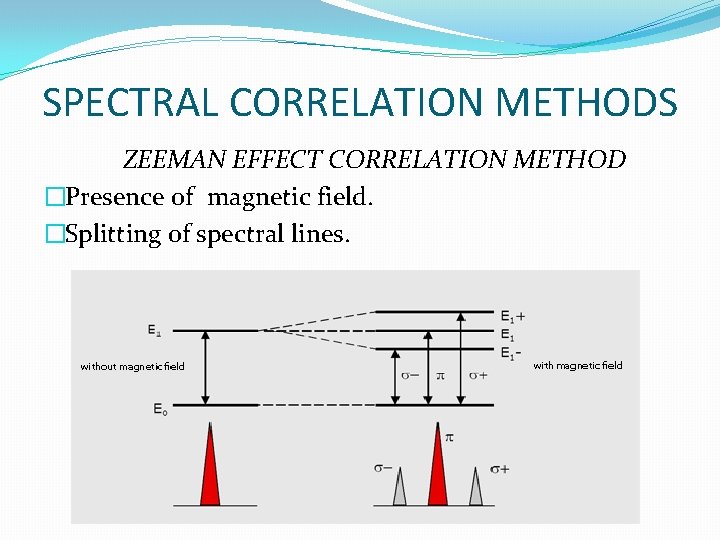
SPECTRAL CORRELATION METHODS ZEEMAN EFFECT CORRELATION METHOD �Presence of magnetic field. �Splitting of spectral lines.

CONCLUSION �One of the most important technique in quantitative analysis �It is based on the absorption of radiation �Measurements could be done at ppb levels �It’s widely used method �The preparation of the sample is usually simple and rapid

CONCLUSION �There are many adventages ü High sensitivity [10 -10 g (flame), 10 -14 g (non-flame)] ü Good accuracy (Relative error 0. 1 ~ 0. 5 % ) ü High selectivity

APPLICATIONS OF AAS �Water analysis (e. g. Ca, Mg, Fe, Si, Al, Ba content) �Food analysis �Analysis of animal feedstuffs (e. g. Mn, Fe, Cu, Cr, Se, Zn) �Analysis of soils �Clinical analysis (blood samples: whole blood, plasma, serum; Ca, Mg, Li, Na, K, Fe)

REFERENCES �http: //www. scribd. com/doc/10513921/Atomic. Absorption-Determination-of-Zinc-and-Copper-in-a. Multivitamin �http: //www. hemlin. pp. se/USstarten. html �http: //www. learn-english-today. com/businessenglish/presentations-phrases. htm

THANKS FOR YOUR ATTENTION. .
- Slides: 28