ATOC 4720 class 4 n Variable constituents n

ATOC 4720: class 4 n Variable constituents n Charged particles in the atmosphere
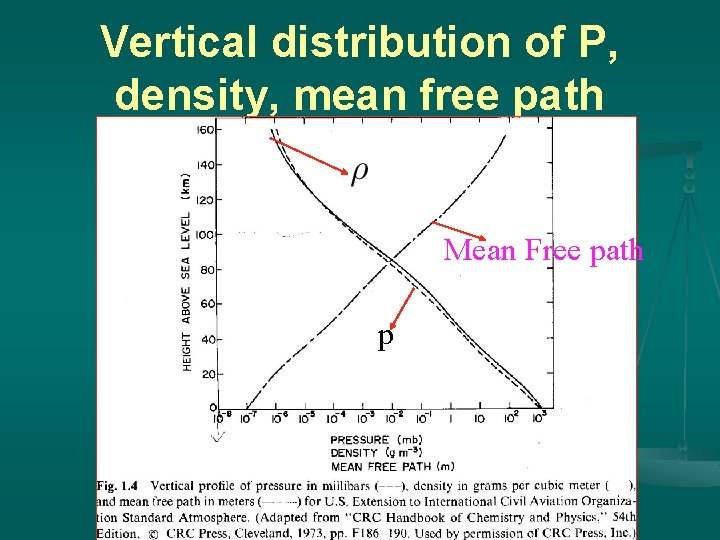
Vertical distribution of P, density, mean free path Mean Free path p
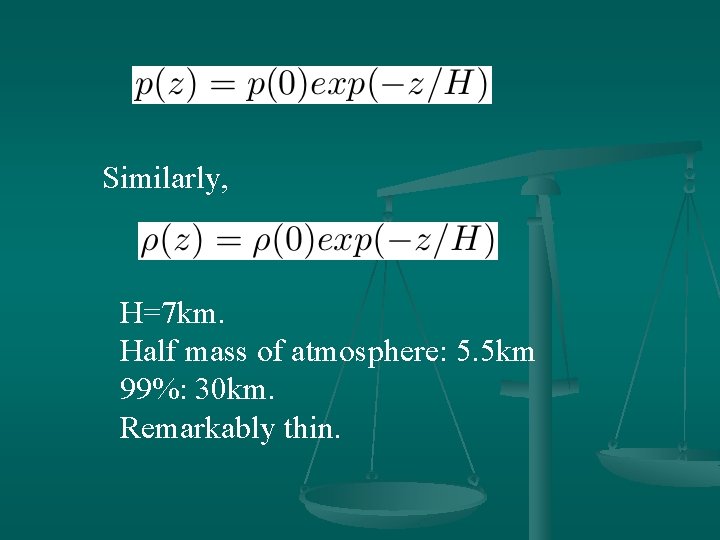
Similarly, H=7 km. Half mass of atmosphere: 5. 5 km 99%: 30 km. Remarkably thin.

Atmospheric composition as a function of height 2 processes: Molecular diffusion Mixing due to fluid motion The level of transition from turbulent Mixing to molecular diffusion is called The turbopause. The well mixed region Below the turbopause is called the Homosphere; the region above is called Heterosphere.

1. Variable constituents

Atmospheree composition SUM= 0 -0. 04 325 ppm 0 -12 ppm 5 ppm

Water vapor: sources and sinks

n n Main source: evaporation from the earth’s surface is the main source of atmospheric water vapor; Condensation which takes place in clouds is the main sink of atmospheric water vapor. Typical “lifetime” of a molecule of water vapor in the atmosphere is only “a week”. Concentration:

Ozone: source and sink

n n Source: Ozone is primarily generated by photochemical reactions in the layer between 20 -60 km; [polluted air: small] Sink: At the earth’s surface, reaction with plants and dissolving in water. At 10 -20 km, it is rather stable with a lifetime on the order of months. Long-term average, downward ozone flux

2. Charged particles in the atmosphere n n Importance: play a crucial role in geophysical phenomena: lightening, reflection of radio waves, fluctuations in the geomagnetic field, etc. Sources: (1) X-ray and UV radiation from the sun ionizes air molecules. All the sun’s ionizing radiation is absorbed above 60 km; (2) Highenergy cosmic rays; (3) Radioactive decay within the earth’s crust; (4) Electric charges: separated within clouds.

Most electrons: >60 km
- Slides: 13