Atmospheric chemistry Day 3 Tropospheric chemistry Global tropospheric

Atmospheric chemistry Day 3 Tropospheric chemistry

Global tropospheric chemistry Questions to be addressed: 1. Many organic compounds emitted to the atmosphere are oxidised, eventually forming CO 2 and H 2 O. What determines the oxidising capacity of the atmosphere? 2. Methane is a greenhouse gas, whose atmospheric concentration has more than doubled since the industrial revolution. What governs it concentration? 3. Tropospheric oxidation is strongly influenced by NOx, whose lifetime is ~ 1 day. How is NOx transported to regions with no NOx emissions? 4. Ozone is a secondary pollutant. In the boundary layer it affects human health, growth of vegetation and materials. It is also a greenhouse gas. What governs its concentration?
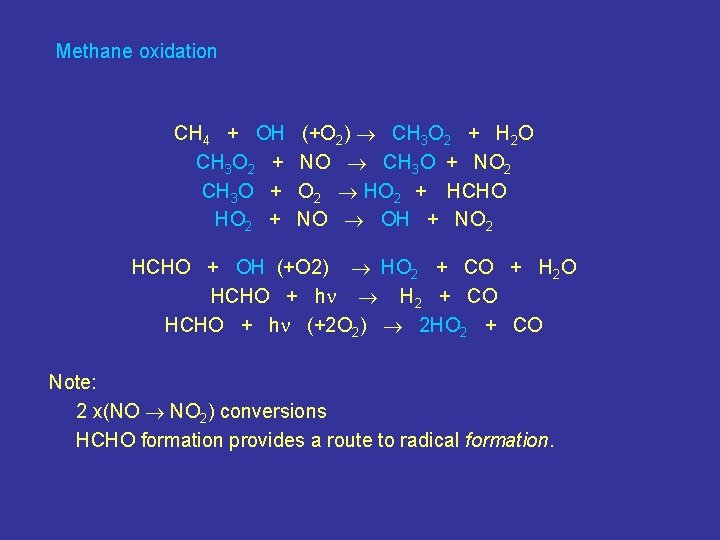
Methane oxidation CH 4 + OH CH 3 O 2 + CH 3 O + HO 2 + (+O 2) CH 3 O 2 + H 2 O NO CH 3 O + NO 2 HO 2 + HCHO NO OH + NO 2 HCHO + OH (+O 2) HO 2 + CO + H 2 O HCHO + h H 2 + CO HCHO + h (+2 O 2) 2 HO 2 + CO Note: 2 x(NO NO 2) conversions HCHO formation provides a route to radical formation.
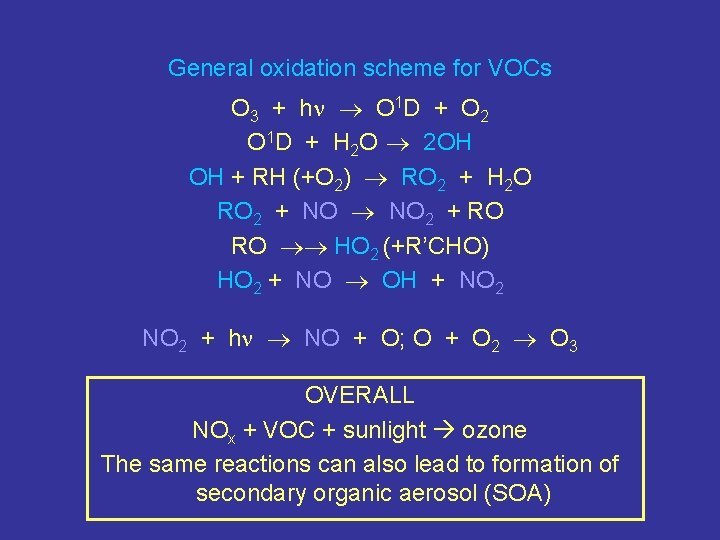
General oxidation scheme for VOCs O 3 + h O 1 D + O 2 O 1 D + H 2 O 2 OH OH + RH (+O 2) RO 2 + H 2 O RO 2 + NO 2 + RO RO HO 2 (+R’CHO) HO 2 + NO OH + NO 2 + h NO + O; O + O 2 O 3 OVERALL NOx + VOC + sunlight ozone The same reactions can also lead to formation of secondary organic aerosol (SOA)
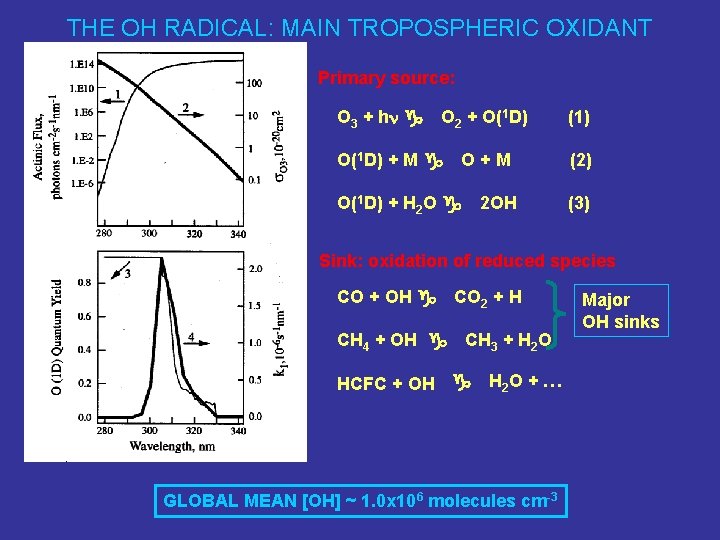
THE OH RADICAL: MAIN TROPOSPHERIC OXIDANT Primary source: O 3 + hn g O 2 + O(1 D) + M g O+M O(1 D) + H 2 O g 2 OH (1) (2) (3) Sink: oxidation of reduced species CO + OH g CH 4 + OH g CO 2 + H CH 3 + H 2 O HCFC + OH g H 2 O + … GLOBAL MEAN [OH] ~ 1. 0 x 106 molecules cm-3 Major OH sinks

Other oxidising species NO 3 NO 2 + O 3 NO 3 + O 2 NO 2 + NO 3 + M N 2 O 5 NO 3 is rapidly lost in the day by photolysis and reaction with NO ( NO 2), so that its daytime concentration is low. It is an important night time oxidant. It adds to alkenes to form nitroalkyl radicals which form peroxy radicals in the usual way. O 3 Ozone reacts with alkenes to form a carbonyl + an energised Criegee biradical. The latter can be stabilised or decompose. One important reaction product is OH: O 3 reactions with alkenes can act as a source of OH, even at night.

Removal of organic compounds from the atmosphere by reaction with OH • Lifetime = 1/{k[OH]} = 1/{1 x 106 k} s Rate coefficients at 298 K/10 -12 cm 3 molecule-1 s-1 CH 4: 7 x 10 -3; CO: 0. 24; isoprene: 110; ethane: 0. 25 • Atmospheric distribution depends on lifetime. NH is major source of anthropogenic pollutants. CH 4 is distributed globally with only a slight NH/SH difference. Isoprene is found only close to its (very extensive) sources. • The oxidising capacity of the atmosphere refers to its capacity to remove VOCs and depends on [OH] (and the concentrations of other oxidants – O 3, NO 3

Global budget for methane (Tg CH 4 yr-1) • Sources: • Sinks: Natural 160 – Trop. oxidation Anthropogenic 375 by OH Total 535 – Transfer to Natural Sources: stratosphere wetlands, termites, oceans… – Uptake by soils Anthropogenic Sources: Total natural gas, coal mines, enteric fermentation, rice paddies, 445 40 30 515 Notes: 1. The rate of oxidation is k 5[CH 4][OH], where the concentrations are averaged over the trop. 2. Concentrations of CH 4 have increased from 800 to 1700 ppb since preindustrial times 3. Methane is a greenhouse gas.
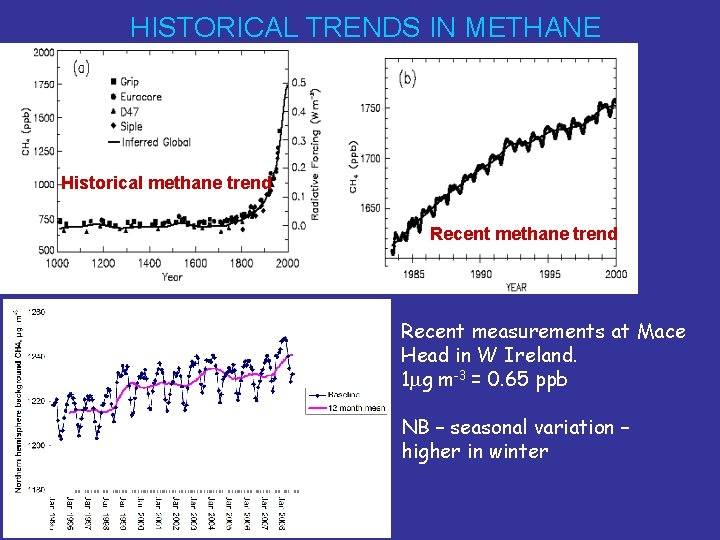
HISTORICAL TRENDS IN METHANE Historical methane trend Recent measurements at Mace Head in W Ireland. 1 mg m-3 = 0. 65 ppb NB – seasonal variation – higher in winter
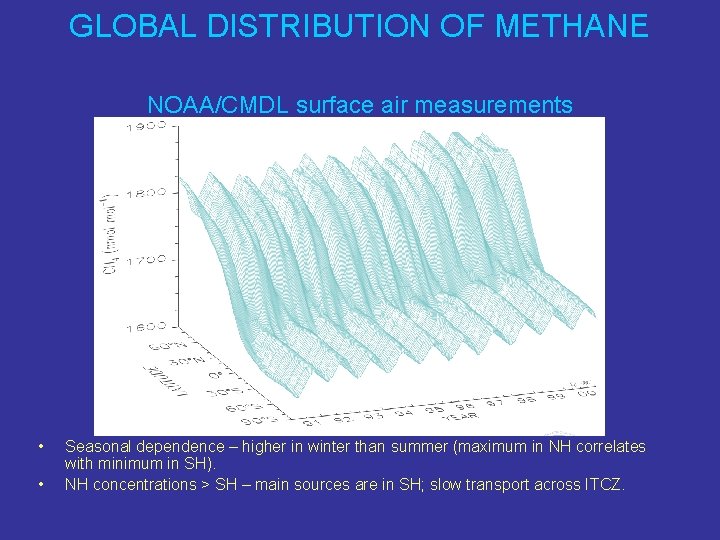
GLOBAL DISTRIBUTION OF METHANE NOAA/CMDL surface air measurements • • Seasonal dependence – higher in winter than summer (maximum in NH correlates with minimum in SH). NH concentrations > SH – main sources are in SH; slow transport across ITCZ.

GLOBAL BUDGET OF CO
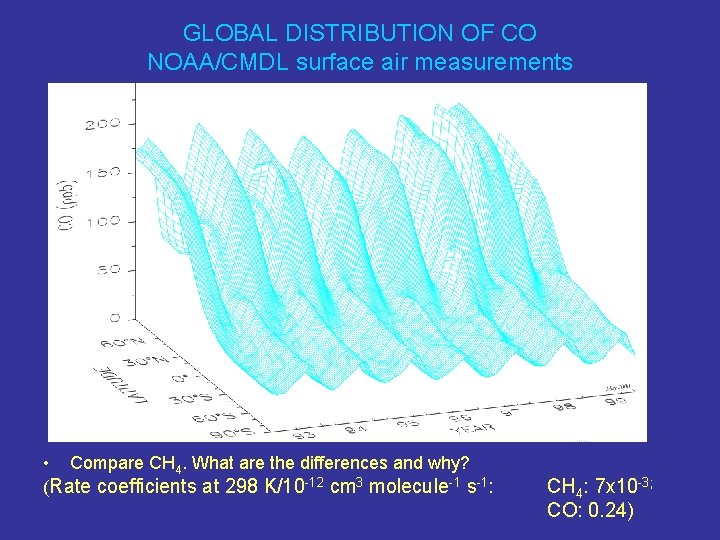
GLOBAL DISTRIBUTION OF CO NOAA/CMDL surface air measurements • Compare CH 4. What are the differences and why? (Rate coefficients at 298 K/10 -12 cm 3 molecule-1 s-1: CH 4: 7 x 10 -3; CO: 0. 24)

Global VOC emissions (Tg yr-1) Anthropogenic: fuel production and distribution 17; fuel consumption 49; road transport 36; chemical industry 2; solvents 20; waste burning 8, other 10. Total 142 Tg yr-1 Biogenic: isoprene 503; monoterpenes 127; other reactive VOCs 260, unreactive VOCs 260; Total 1150 Tg yr-1 Typical atmospheric lifetimes (for [OH] = 1 x 106 molecule cm-3) t = 1/k[OH] CH 4 6 yr isoprene 2. 7 h CO 48 days ethane 46 days benzene 6 days ethene 30 h

Global budget for NOx • Global sources (Tg N yr-1): Fossil fuel combustion 21; Biomass burning: 12 Soils 6 Lightning 3 Ammonia oxidation 3 Aircraft 0. 5 Transport from strat 0. 1 • Coupling (rapid - ~ 1 minute in the day NO + O 3 → NO 2 + O 2 NO 2 + Light → NO + O; O + O 2 + M → O 3 + M Also HO 2 + NO → NO 2 + OH • Loss OH + NO 2 + M → HNO 3 + M Rainout of HNO 3 • Lifetime of NOx is about 1 day. NOx is a key component in ozone formation. Can it be transported to regions where it is not strongly emitted?
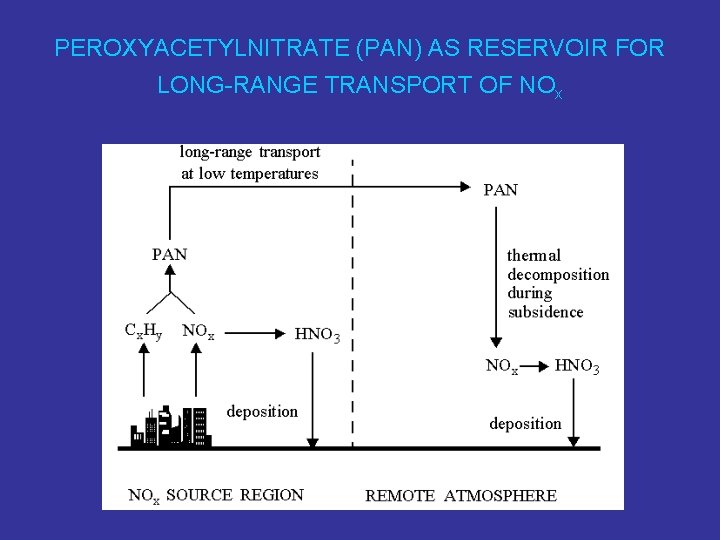
PEROXYACETYLNITRATE (PAN) AS RESERVOIR FOR LONG-RANGE TRANSPORT OF NOx
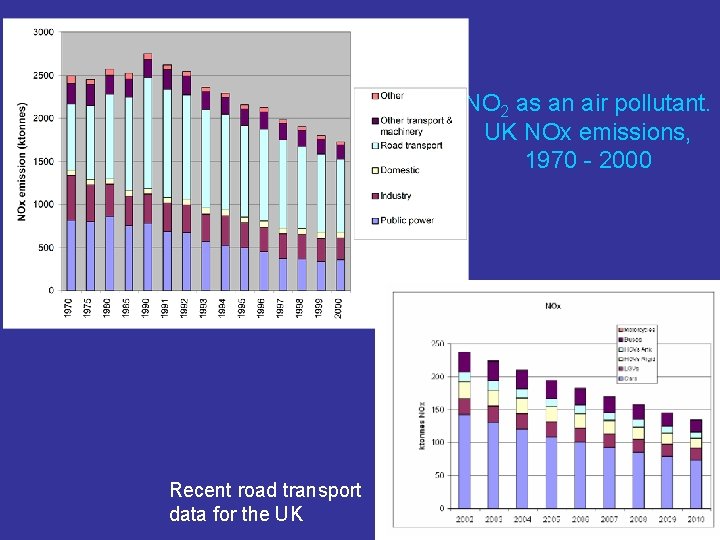
NO 2 as an air pollutant. UK NOx emissions, 1970 - 2000 Recent road transport data for the UK

Spatial distribution of NOx emissions

EU Air quality objectives – limit values legally binding NO 2 1 hour mean 200 mg m-3 (105 ppb); (not to be exceeded >18 times per year) annual mean 40 mg m-3 (21 ppb) To be achieved by 2010. Possible to apply for derogation to 2015
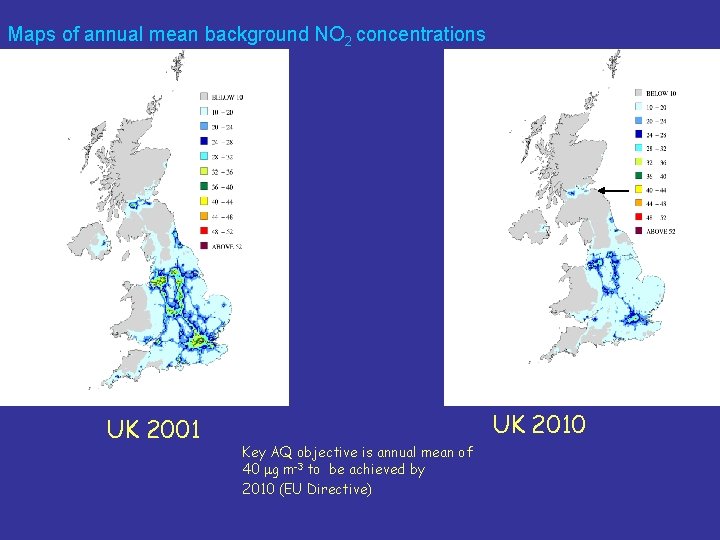
Maps of annual mean background NO 2 concentrations UK 2001 UK 2010 Key AQ objective is annual mean of 40 mg m-3 to be achieved by 2010 (EU Directive)
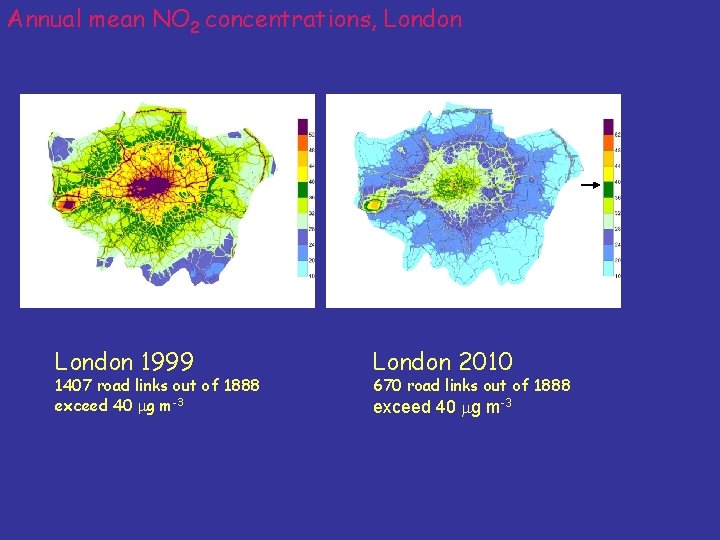
Annual mean NO 2 concentrations, London 1999 1407 road links out of 1888 exceed 40 mg m-3 London 2010 670 road links out of 1888 exceed 40 mg m-3

Hungarian air quality network http: //www. kvvm. hu/olm/index. php
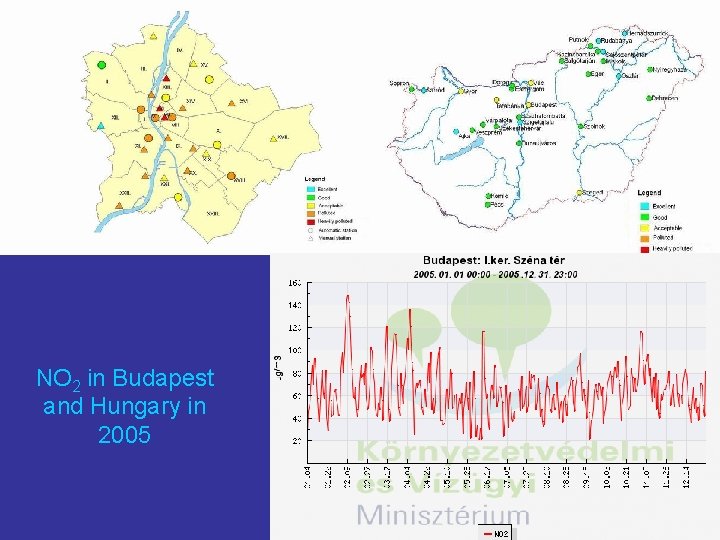
NO 2 in Budapest and Hungary in 2005
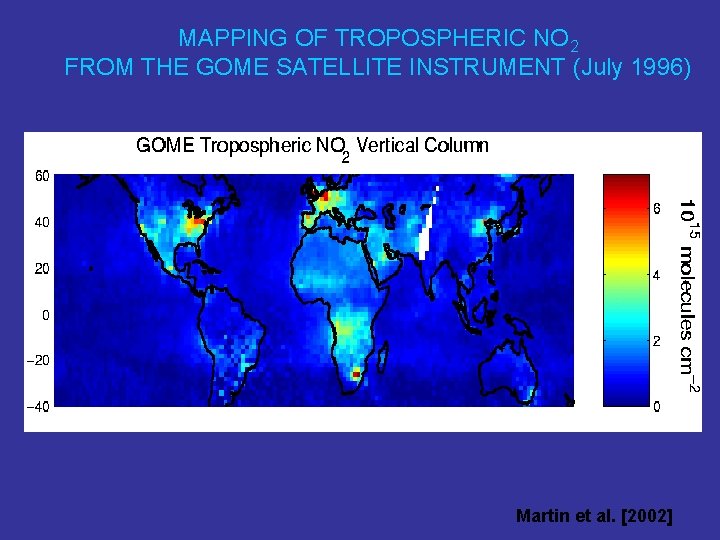
MAPPING OF TROPOSPHERIC NO 2 FROM THE GOME SATELLITE INSTRUMENT (July 1996) Martin et al. [2002]
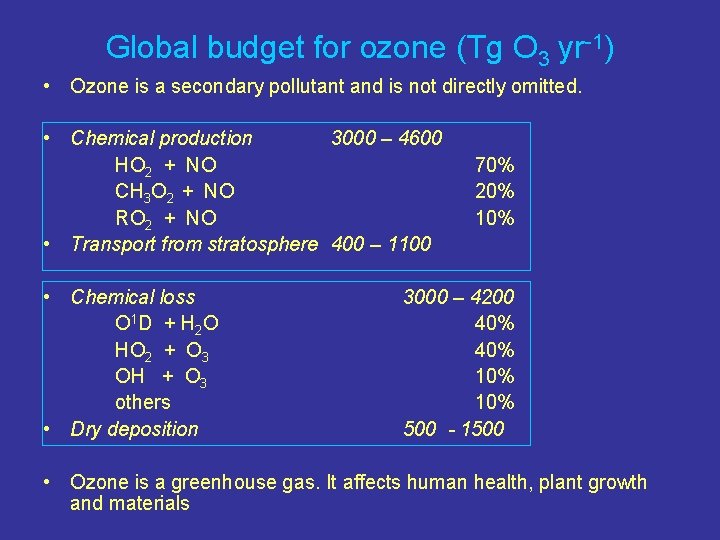
Global budget for ozone (Tg O 3 yr-1) • Ozone is a secondary pollutant and is not directly omitted. • Chemical production 3000 – 4600 HO 2 + NO CH 3 O 2 + NO RO 2 + NO • Transport from stratosphere 400 – 1100 • Chemical loss O 1 D + H 2 O HO 2 + O 3 OH + O 3 others • Dry deposition 70% 20% 10% 3000 – 4200 40% 10% 500 - 1500 • Ozone is a greenhouse gas. It affects human health, plant growth and materials
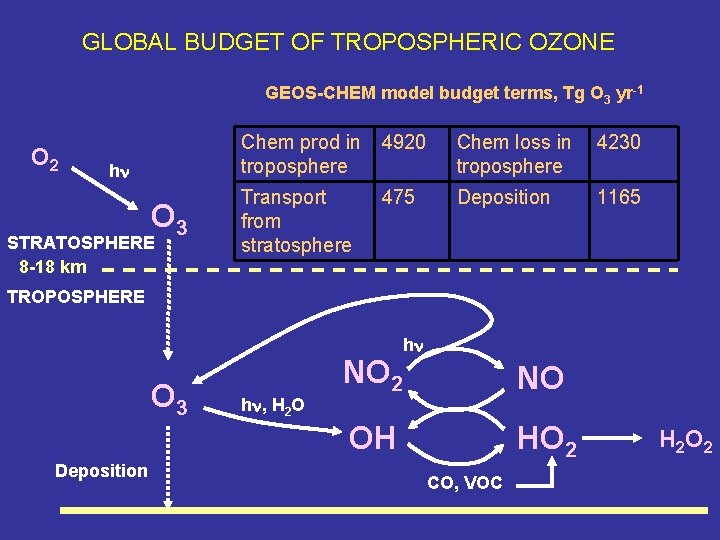
GLOBAL BUDGET OF TROPOSPHERIC OZONE GEOS-CHEM model budget terms, Tg O 3 yr-1 O 2 hn O 3 STRATOSPHERE 8 -18 km Chem prod in troposphere 4920 Chem loss in troposphere 4230 Transport from stratosphere 475 Deposition 1165 TROPOSPHERE O 3 hn, H 2 O NO 2 hn NO OH Deposition HO 2 CO, VOC H 2 O 2
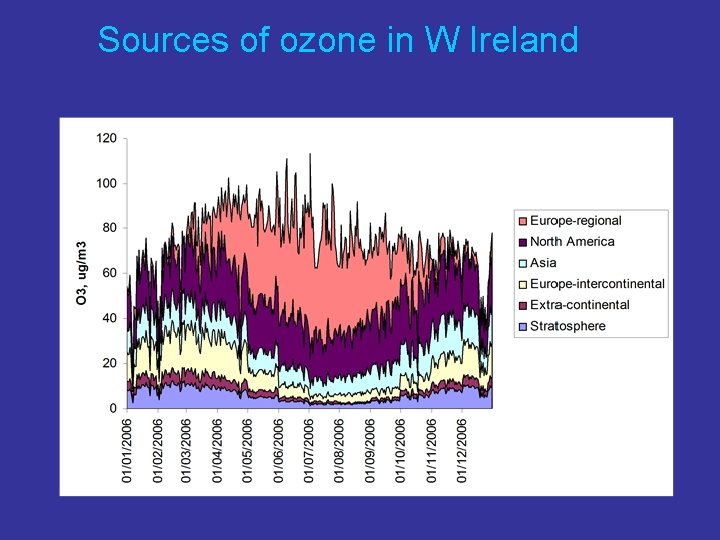
Sources of ozone in W Ireland
- Slides: 26