ATMOSPHERES Atmospheric Basics Layer of gas surrounding a

ATMOSPHERES

Atmospheric Basics Ø Layer of gas surrounding a planet. Ø Usually very thin for terrestrial planets (exception Venus). Ø Affects conditions on the planet. Ø We would like to understand how each of the terrestrial planets ended up having such different atmospheres. Venus’s thick atmosphere

All terrestrial planets probably had minimal (primary: H, He) atmospheres at some point after they formed These original atmospheres were swept away from the terrestrial planets early in their life. Current atmospheres are secondary atmospheres, formed primarily by outgassing (mostly carbon dioxide - CO 2)

Holding onto an atmosphere requires gravity Ø The strength of gravity determines the escape velocity from the planet. Ø The temperature and composition of an atmosphere determines the velocities of atoms and molecules in the atmosphere. (At a given temperature, H and He will have higher velocities than more massive elements or molecules; recall that K. E. = 1/2 mv 2)

Holding onto an atmosphere requires gravity Ø The strength of gravity determines the escape velocity from the planet. Ø The temperature and composition of an atmosphere determines the velocities of atoms and molecules in the atmosphere. Ø If the constituents of an atmosphere are moving faster than escape velocity, then a planet or moon will be unable to hold onto an atmosphere.

Larger (stronger gravity), cooler (slower moving molecules) planets can hold onto atmospheres better than smaller (weaker gravity), hotter (faster moving molecules) planets

● Moon and Mercury “Airless” worlds Ø gravity too weak to hold onto an atmosphere Ø “black Ø sky” The little atmosphere that exists consists of particles of the solar wind that are temporarily trapped.

● Mars Ø Very little atmosphere today (mainly CO 2) Ø Mars had standing and running water on its surface in the past. Ø Therefore, it must have had a more substantial atmosphere in the past Ø Does it have water today? Yes - frozen in polar ice caps and beneath its soil

● Venus Ø Densest atmosphere of all Terrestrials Ø Mostly CO 2 Ø Temperature at surface hot enough to melt lead Ø Pressure at the surface ~ 90 times that on Earth Ø Perpetual cloud cover, sulfuric acid rain Weather forecast “awful” all the time. Ø

● Earth Ø A moderate atmosphere today Ø Mostly nitrogen (N 2), with some oxygen (O 2: arises from photosynthetic life), carbon dioxide (CO 2), etc. Ø Enough to enable liquid water to exist (temperature and pressure adequate) Ø Together the air & water produce erosion

The Jovian planets (high gravity, cool/cold atmospheres) have very substantial atmospheres. ●

LAYERING OF ATMOSPHERES Structure is created within an atmosphere through interactions of atmospheric gasses with light

Interactions of light with atmospheric gasses • X rays • ionize atoms & molecules • dissociate molecules • absorbed by almost all gases • Ultraviolet (UV) • dissociate some molecules • absorbed well by O 3 & H 2 O • Visible (V) • passes right through gases • some photons are scattered • Infrared (IR) • absorbed by greenhouse gases
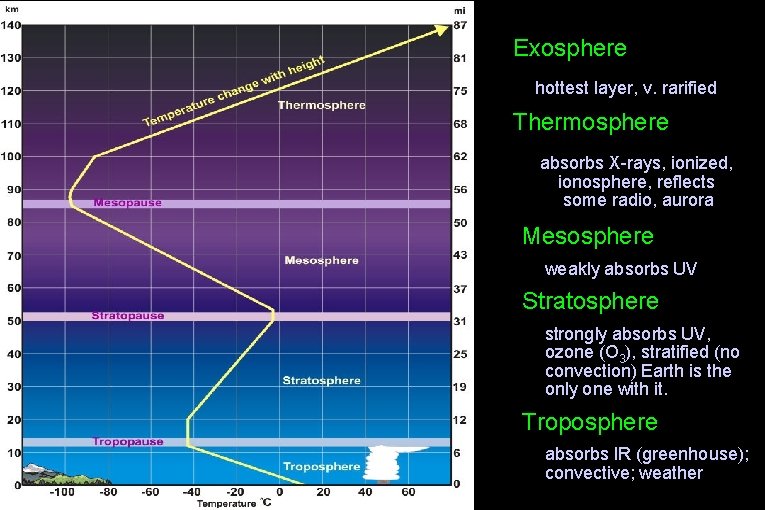
Exosphere hottest layer, v. rarified Thermosphere absorbs X-rays, ionized, ionosphere, reflects some radio, aurora Mesosphere • weakly absorbs UV Stratosphere • strongly absorbs UV, ozone (O 3), stratified (no convection) Earth is the only one with it. Troposphere • absorbs IR (greenhouse); convective; weather

A magnetic field creates “magnetosphere” that deflects away solar wind particles. In the absence of a “magnetosphere”, the solar wind will slowly strip away an atmosphere.

The greenhouse effect Ø Planets heat up by absorbing the Sun’s visible light Ø Planets cool off by radiating infrared out to space Ø Greenhouse gasses trap infrared radiation in troposphere (lowest level of atmosphere), thereby heating the lower atmosphere. ● greenhouse gasses (e. g. , H 2 O, CO 2, CH 4 - methane) transmit visible light but absorb infrared light
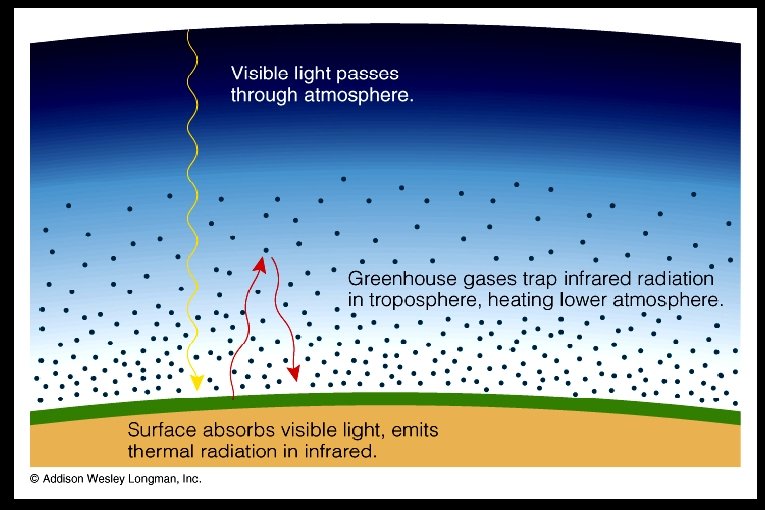
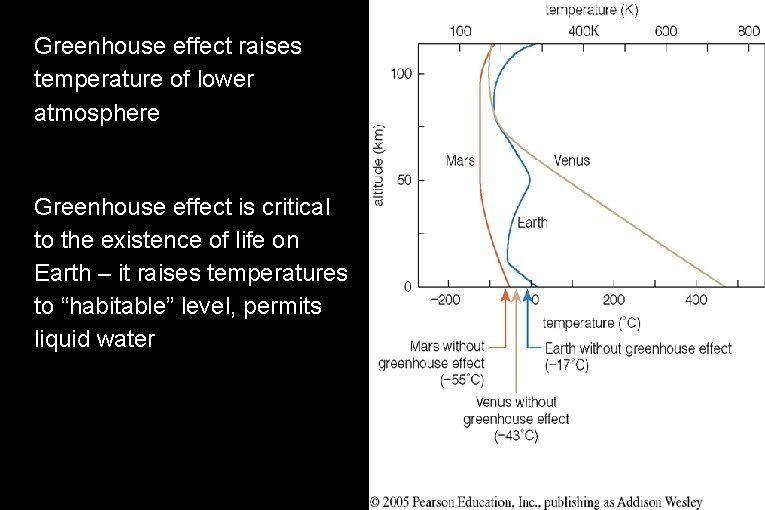
● ● Greenhouse effect raises temperature of lower atmosphere Greenhouse effect is critical to the existence of life on Earth – it raises temperatures to “habitable” level, permits liquid water

EVOLUTION OF ATMOSPHERES

The dominant gasses arising from outgassing were most carbon dioxide and water vapor, with minor amounts of nitrogen, sulfer, argon, … Each terrestrial planet’s outgassed atmosphere was roughly the same at the beginning. Why do they differ now?

Mercury is too small and too hot to hold onto an atmosphere.

Mars lost much of its atmosphere because of its small size. The atmosphere it retains resembles its original atmosphere in composition (essentially CO 2). The fate of its water is still a matter of debate. There appears to be substantial amounts of subsurface frozen water.

Venus and the Earth: started with more or less identical atmospheres. Their atmospheres have subsequently followed very different paths. WHY?

Slightly higher temperatures at Venus’ distance from the Sun made it difficult for water to stay in liquid state. Ø Liquid water exists in abundance on the Earth Ø Carbon dioxide dissolves in oceans Ø Photosynthetic life creates oxygen (oxygen has a short lifetime in the atmosphere - must be constantly replenished).

Evolution of Atmospheres: Earth vs. Venus because of the Earth’s temperature: On Earth there are oceans Original CO 2 has dissolved into oceans and is tied up in carbonate rocks, rocks (carbonates) keep levels of CO 2 just balanced in atmosphere keeps planet WARM but not HOT if planet were hotter, CO 2, H 2 O would be boiled out of oceans and baked out of rocks more CO 2, H 2 O enter Atmosphere

Liquid water may have existed early in Evolution of Atmospheres: Venus’ history – but most vaporized into Earth vs. Venus atmosphere: T was hotter on Venus H 2 O vapor is a greenhouse gas - trapped energy thus making planet hotter; eventually T so high that water boils ‘runaway’ because more H 20 goes into the atmosphere as it evaporates; no water left on planet to dissolve CO 2 – out of balance! eventually stabilized when H 20 broken down by UV sunlight (H escaped to space, O reacted with minerals) and there was no further CO 2 to bake out of the Venus surface

● ● ● This is called the runaway greenhouse effect It happened on Venus because Venus is closer to the Sun. We do not think it can happen on the Earth. So - Earth has less atmosphere because most of our CO 2 is frozen in rocks (e. g. , limestone)

What Determines a Planet’s Surface Temperature? In the absence of the Greenhouse Effect: Ø the planet's distance from the Sun Ø the planet’s overall reflectivity • the higher the albedo (reflectivity), the less light absorbed planet cooler

What Determines a Planet’s Surface Temperature? ● With a greenhouse effect. Ø Ø it increases the energy (heat) in lower atmosphere, keeping the surface warmer It works like a blanket

Greenhouse Effect on the Planets ● Greenhouse Effect warms Venus, Earth, & Mars Ø on Venus: it is very strong Ø on Earth: it is moderate Ø on Mars: it is weak Ø avg. temp. on Venus & Earth would be freezing without it

We are now mostly finished with planetary environments, so. . Before proceeding to life… A brief visit to a missed topic… The Origin of the Earth’s Moon

The Earth-Moon double planet does not fit well into the nebular theory planetesimal accretion predicts both should have the same chemical composition. They don’t - there are subtle but significant differences Moon is composed of less dense material than Earth

The general view is that the Earth’s moon was created as a result of the impact of a large object, perhaps as large as Mars, with the Earth very early in its existence. The moon was formed from the debris of this collision, which included lower density “mantle” material from the Earth.

“Life” v How is life defined? What is needed for life? How hard it is for life to form? What environments are suitable for life?
- Slides: 35