ATMO 469 b569 b CHEE 469 b569 b

ATMO 469 b/569 b, CHEE 469 b/569 b Air Pollution II: Aerosols Chemical Composition: New Frontiers Jan. 24, 2007 Dr. Song GAO (songatmo@email. arizona. edu)

Typical Particle Diameters (mm) Photochemical aerosols Tobacco smoke 0. 01 -1 Coal fly ash 1 -50 Flour dust 15 -20 Pollens 15 -70 0. 25 Human hair: ~25 -100 mm

Aerosols: Basic Properties • Size • Number • Microstructure • Chemistry (composition & transformation)

Organic Compounds in Ambient Aerosols • Highly complex ensemble - species with various functional groups, polarity, volatility, solubility. • Speciation of organic aerosols is a formidable analytical task.

• These 107 compounds together comprise < 12% of the total organic mass in fine aerosols in the SE United States. • Commonly in ambient aerosols, only 10 ~ 15% of the total organic matter (OM, OC) can be resolved into individual compounds.

Atmospheric Aerosols: tiny solid or liquid particles suspended in the air • Perturb the Earth’s radiation balance both directly and indirectly. • Probably partially counteract greenhouse gases in climatic effects. • Have the largest uncertainty of all climate forcings [IPCC, 2001].

Aerosols: Largest Uncertainty in Climate Forcings

Global Scale: Volcanoes, Aerosols & Climate Large volcano eruptions provide dramatic evidence of the ability of aerosols to affect global climate.

Importance of Organic Compounds in Atmospheric Aerosols • Comprise a substantial fraction of aerosol mass: - In polluted areas, 25 ~ 65% can be organic in nature. [Wolff et al. , 1991; Chow et al. , 1994; Novakov et al. , 1997] - In MBL, at least 10% of aerosol mass is organic [ACE-1]. • Often present in a single particle with inorganics. [Murphy et al. , 1998; Noble and Prather, 1996]

Potential Roles in Global Climate (I): light absorption • Not only black carbon, but also certain organic carbon in aerosols, can absorb solar radiation: [Pöschl 2003, 2005; Kirchstetter et al. , 2004] • “Light-absorbing brown or yellow carbon”?

Potential Roles in Global Climate (II): hygroscopic growth; CCN activation • Classical Köhler theory: based on inorganic salts as the solutes. • Some organics activate in accordance with classical Köhler theory, while others deviate from it. • Organics can alter the microstructure of aerosol particles. (Surfactants kinetic limitations of CCN/IN activation) • Modified Köhler equations (considering organics) are needed to accurately describe atmospheric processes [Laaksonen et al. , 1997; Charlson et al. , 2001].

Potential Roles in Human Health • Epidemiological studies: fine aerosols are correlated with severe health effects, including enhanced mortality, cardiovascular, respiratory, and allergic diseases. • Toxicological studies: model and real aerosols can cause pulmonary toxicity.

Experimental Studies – Smog Chamber • Inject model hydrocarbons, ozone and/or other reactants (& light). • Measure aerosol size distribution, number concentration. • Collect aerosol samples on filters and analyze composition with chromatography and mass spectrometry.

Structures of Model Hydrocarbons Precursor Hydrocarbon Molecular Structure Precursor Hydrocarbon cyclopentene 1 -methyl cyclopentene cyclohexene 1 -methyl cyclohexene cycloheptene 3 -methyl cyclohexene cyclooctene α-pinene Molecular Structure

Multiple Analytical Techniques Employed Liquid Chromatography – Mass Spectrometry • HP 1100 Series HPLC – single quadrupole MS (ESI source) Ion Trap Mass Spectrometry • Finnigan LCQ ion trap MS (ESI source) Laser Desorption/Ionization Mass Spectrometry • Voyager-DE PRO time-of-flight (TOF) MS (MALDI source) High-Resolution Mass Spectrometry • Waters LCT Premier TOF MS (ESI source) • JEOL JMS-600 H double-focusing, magnetic sector MS (FAB ionization)

• Cycloolefins can be oxidized to form a variety of compounds, some of which have low enough vapor pressures to condense into the aerosol phase. • Secondary Organic Aerosol (SOA) Formation • Oxidation reaction mechanism : example. Gao et al. (2004) J. Phys. Chem. paper.
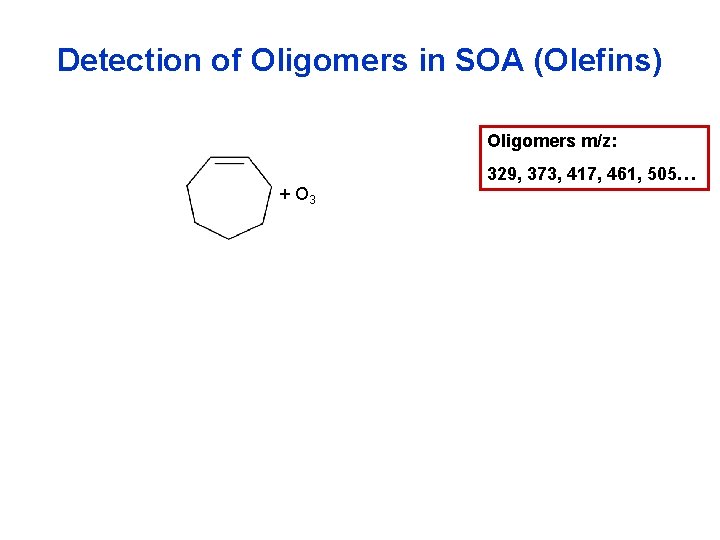
Detection of Oligomers in SOA (Olefins) Oligomers m/z: 329, 373, 417, 461, 505… + O 3
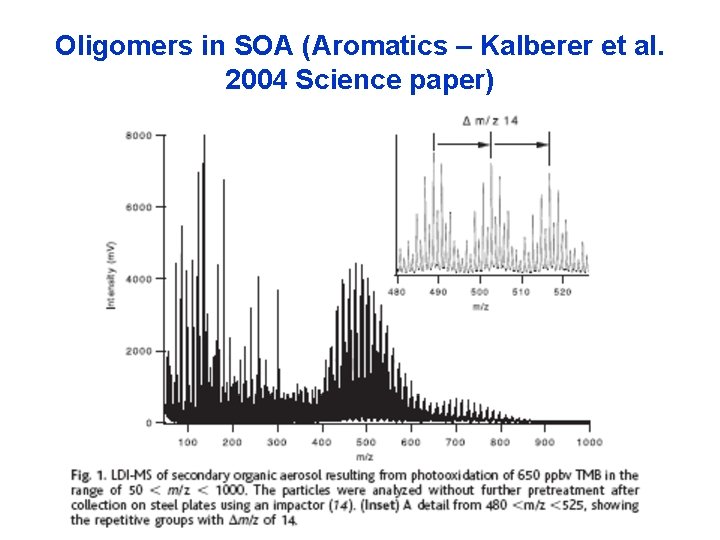
Oligomers in SOA (Aromatics – Kalberer et al. 2004 Science paper)
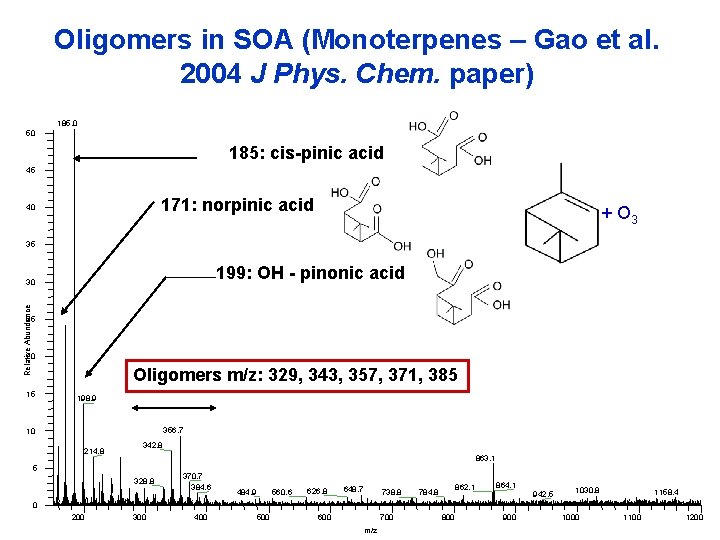
Oligomers in SOA (Monoterpenes – Gao et al. 2004 J Phys. Chem. paper) 185. 0 50 185: cis-pinic acid 45 171: norpinic acid 40 + O 3 35 199: OH - pinonic acid Relative Abundance 30 25 20 Oligomers m/z: 329, 343, 357, 371, 385 15 198. 9 356. 7 10 214. 8 342. 8 863. 1 5 328. 8 370. 7 384. 6 484. 9 560. 6 626. 8 648. 7 738. 8 862. 1 784. 8 864. 1 942. 5 1030. 8 1158. 4 0 200 300 400 500 600 700 m/z 800 900 1000 1100 1200
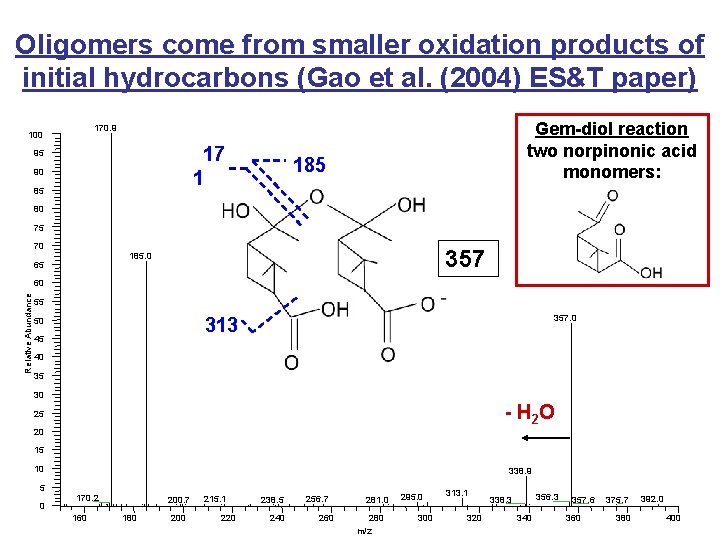
Oligomers come from smaller oxidation products of initial hydrocarbons (Gao et al. (2004) ES&T paper) Gem-diol reaction two norpinonic acid monomers: 170. 9 100 17 1 95 90 85 185 80 75 70 357 185. 0 65 Relative Abundance 60 55 357. 0 313 50 45 40 35 30 - H 2 O 25 20 15 10 338. 9 5 0 170. 2 160 200. 7 180 200 215. 1 220 238. 5 240 256. 7 260 281. 0 280 m/z 295. 0 300 313. 1 320 356. 3 338. 3 340 357. 6 360 375. 7 380 392. 0 400

• Particle phase acidity has an explicit effect on oligomer formation and SOA yield: Higher acidity faster oligomer formation larger oligomers higher SOA yield. • Oligomers can form from α-pinene ozonolysis with or without pre-existing seed: The organic acids produced from hydrocarbon oxidation itself can readily facilitate oligomer formation in SOA.

Summary • Oligomers comprise a substantial fraction of secondary organic aerosols generated in chamber studies. • Acid-catalyzed heterogeneous reactions are the proposed pathways to form these oligomers. • As a consequence, some “semi-volatile” compounds can stay in the aerosol phase and increase the global aerosol burden.

Open Questions Ø Not yet clear whether oligomers are abundant in ambient aerosols. Ø Models to formulate the global burden and distribution of aerosols need to be reevaluated to account for these new compounds and their formation pathways. Ø New analytical methods need to be developed to fully understand aerosol composition, esp. organic species.

References • Seinfeld, J. H. ; Pankow, J. F. Annual Rev. Phys. Chem. 2003, 54, 121 – 140. • Iinuma, Y. ; Böge, O. ; Gnauk, T. ; Herrmann, H. Atmos. Environ. 2004, 38, 761 – 773. • Kalberer, M. ; Paulsen, D. ; Sax, M. ; Steinbacher, M. ; Dommen, J. ; Prevot, A. S. H. ; Fisseha, R. ; Weingartner, E. ; Frankevich, V. ; Zenobi, R. ; Baltensperger, U. Science 2004, 303, 1659 - 1662. • “Low-molecular-weight and oligomeric components in secondary organic aerosol from the ozonolysis of cycloalkenes and α-pinene”, Gao, S. , Keywood, M. , Ng, N. L. , Surratt, J. , Varutbangkul, V. , Bahreini, R. , Flagan, R. C. , Seinfeld, J. H. , J. Phys. Chem. A, 108 (46), 10147 – 10164, doi: 10. 1021/jp 047466 e, 2004. • “Particle phase acidity and oligomer formation in secondary organic aerosol”, Gao, S. , Ng, N. L. , Keywood, M. , Varutbangkul, V. , Bahreini, R. , Nenes, A. , He, J. , Yoo, K. Y. , Beauchamp, J. L. , Hodyss, R. P. , Flagan, R. C. , Seinfeld, J. H. , Environ. Sci. Technol. , dio: 10. 1021/es 049125 k, 2004. • “Characterization of polar organic components in fine aerosols in the Southeastern United States”, Gao, S. , J. Surratt, E. Knipping, E. Edgerton, M. Shahgholi and J. H. Seinfeld, J. Geophys. Res. , 111 (D 14): Art. No. D 14314, 2006.
- Slides: 24