Atkins de Paula Physical Chemistry CHAPTER NUMBER 4

Atkins/ de Paula: Physical Chemistry CHAPTER NUMBER 4: Physical Transformations of Pure Substances Art Power. Points © Peter Atkins & Julio De Paula 2010
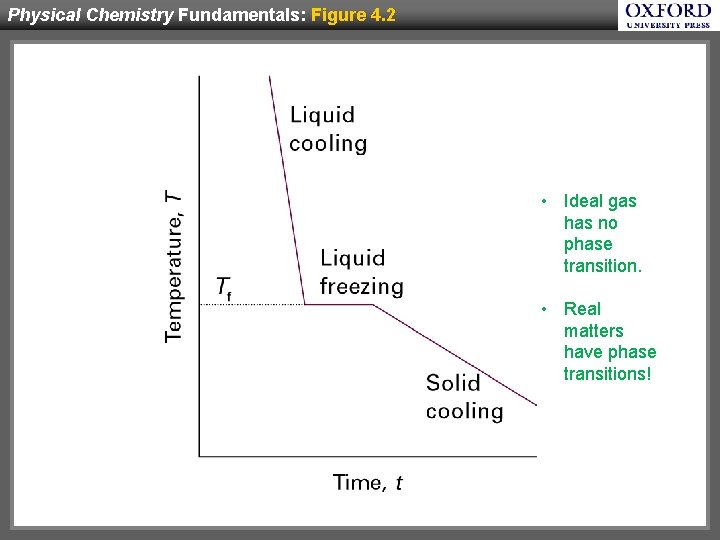
Physical Chemistry Fundamentals: Figure 4. 2 • Ideal gas has no phase transition. • Real matters have phase transitions!
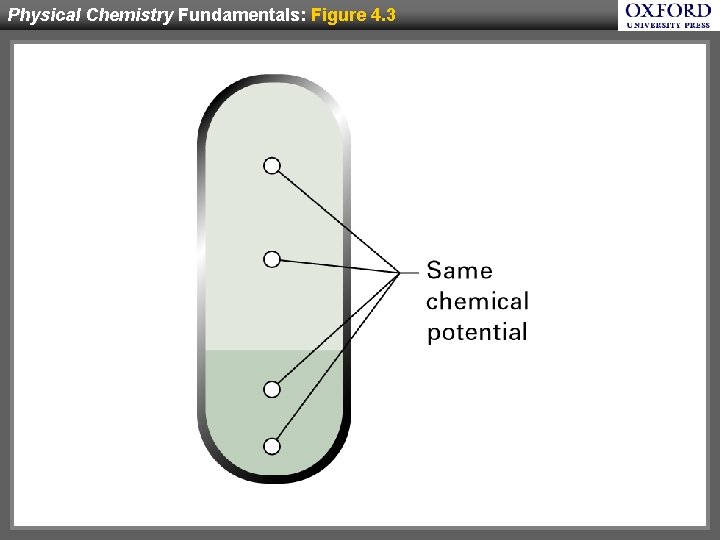
Physical Chemistry Fundamentals: Figure 4. 3
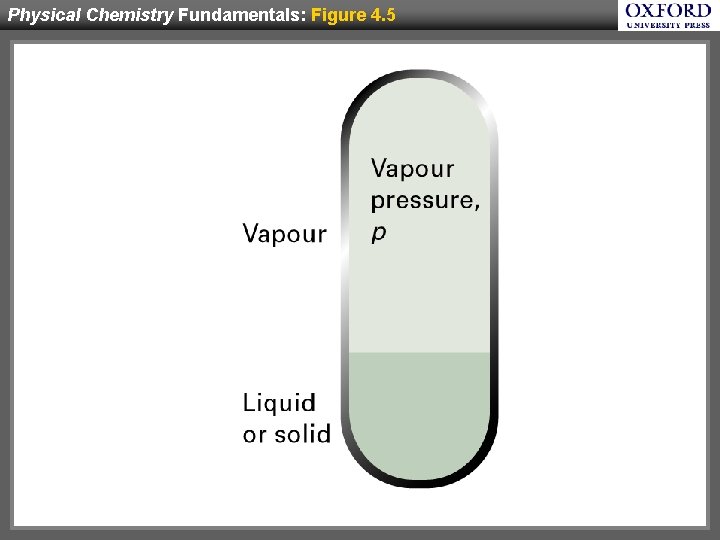
Physical Chemistry Fundamentals: Figure 4. 5
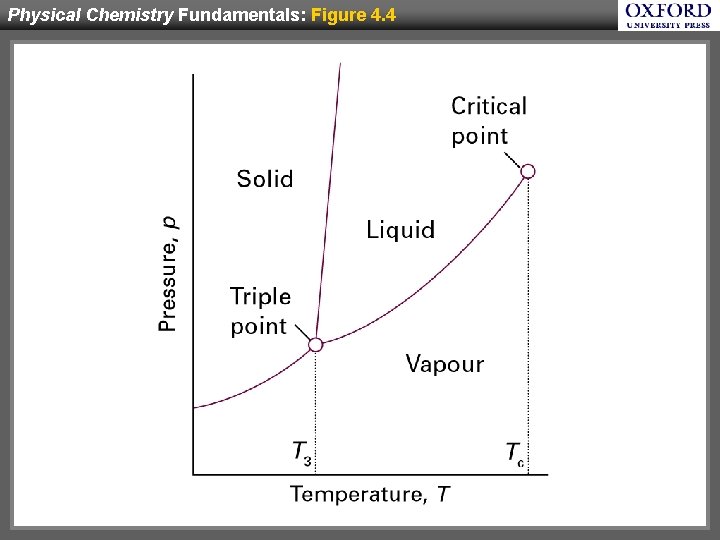
Physical Chemistry Fundamentals: Figure 4. 4

Physical Chemistry Fundamentals: Figure 4. 7 Phase rule: 1. consider all possibility of molarity in one phase. Minus x. A+x. B=1. Thus, P(C-1). 2. 2. how many equalities there, for µ(a)= µ (b)? C(P-1). 3. 3 P(C-1)-C(P-1) +2.
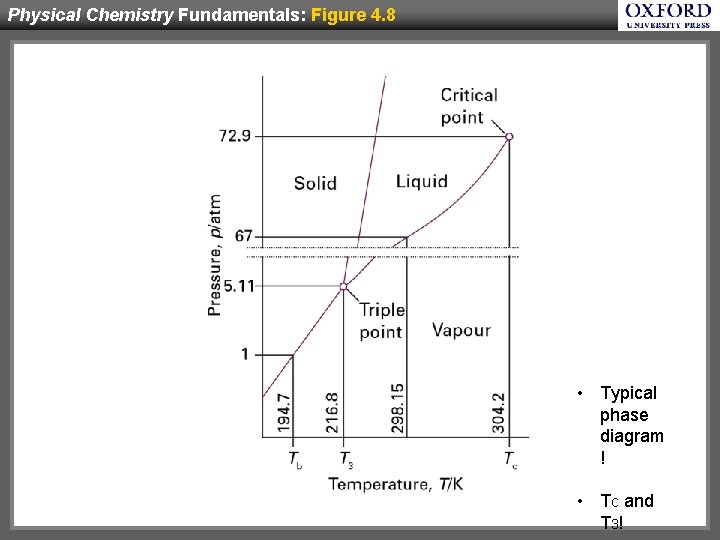
Physical Chemistry Fundamentals: Figure 4. 8 • Typical phase diagram ! • Tc and T 3!
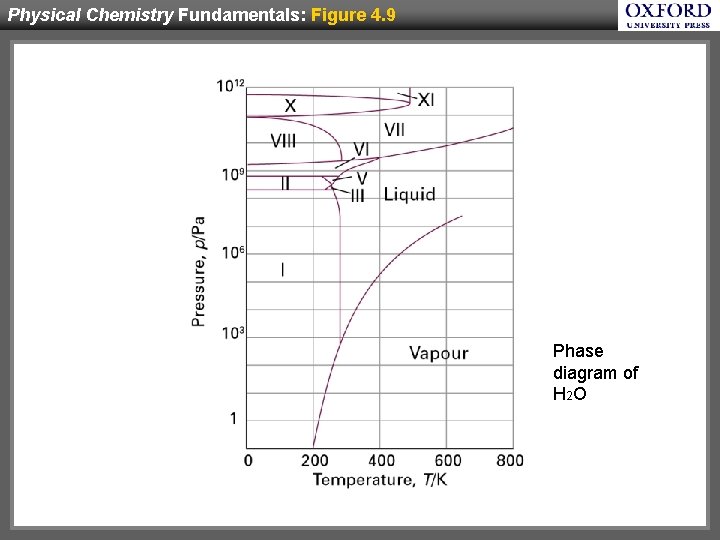
Physical Chemistry Fundamentals: Figure 4. 9 Phase diagram of H 2 O
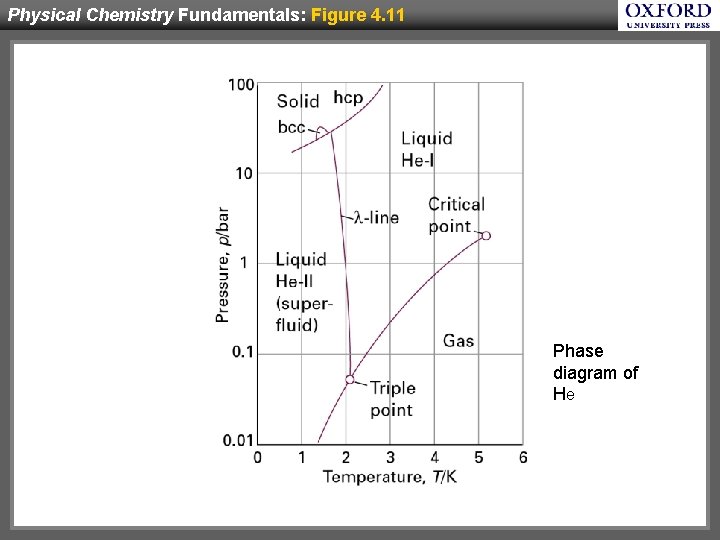
Physical Chemistry Fundamentals: Figure 4. 11 Phase diagram of He
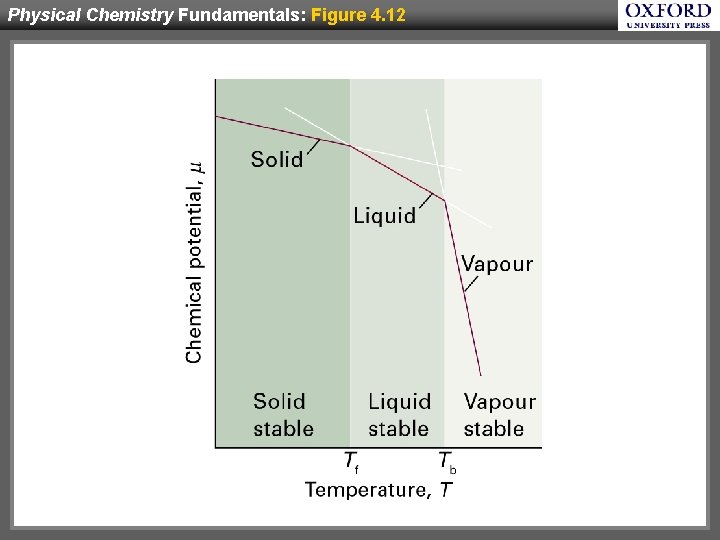
Physical Chemistry Fundamentals: Figure 4. 12
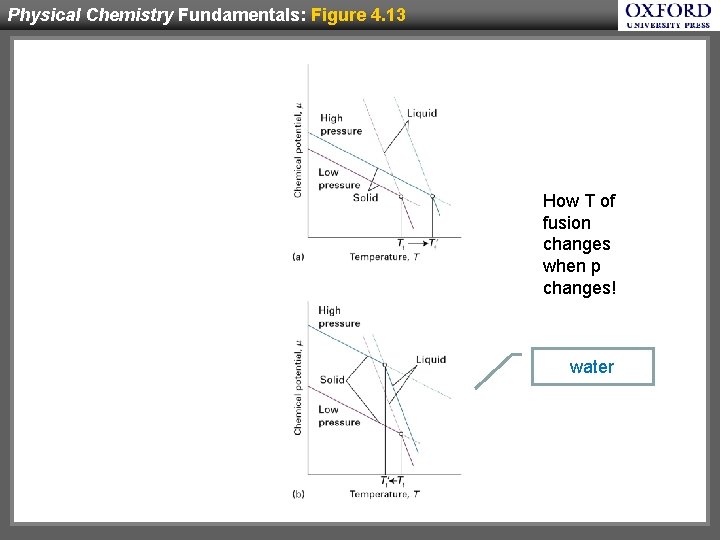
Physical Chemistry Fundamentals: Figure 4. 13 Hw How T of fusion changes when p changes! water

Main Concepts • Phase diagram why it looks like that way? 4 lines meet? Forbidden. • At mountain, Tb lowers! Why? What is deeper reason? p up, Tb up. Tf is also up but, for H 2 O, down. • Introduction of chemical potential! (why? Because two phases may have different moles! d. G=(µ 2 –µ 1)dn (page 137, 9 th Ed. ) • Phase rule • Dependence of vapor pressure on external pressure. dµ(l)=dµ(g), d µ =Vm dp at const. T. • Clayperon equation and its application to derive the curves in phase diagram

Physical Chemistry Fundamentals: Figure 4. 14 How does vapour pressure depend on DP!
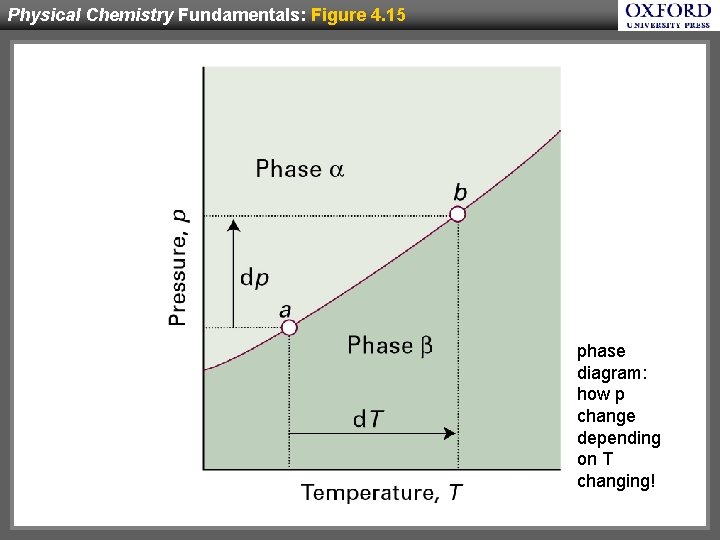
Physical Chemistry Fundamentals: Figure 4. 15 phase diagram: how p change depending on T changing!
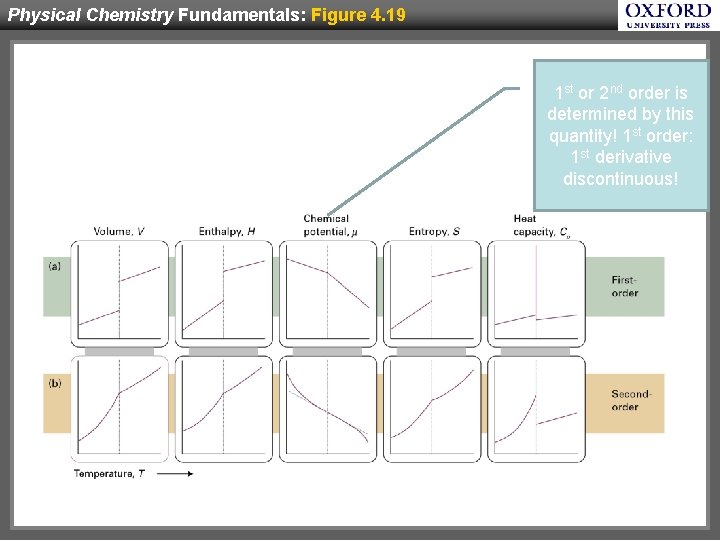
Physical Chemistry Fundamentals: Figure 4. 19 1 st or 2 nd order is determined by this quantity! 1 st order: 1 st derivative discontinuous!
- Slides: 15