ASTRAL2 study SOFVEL vs SOF RBV in genotype
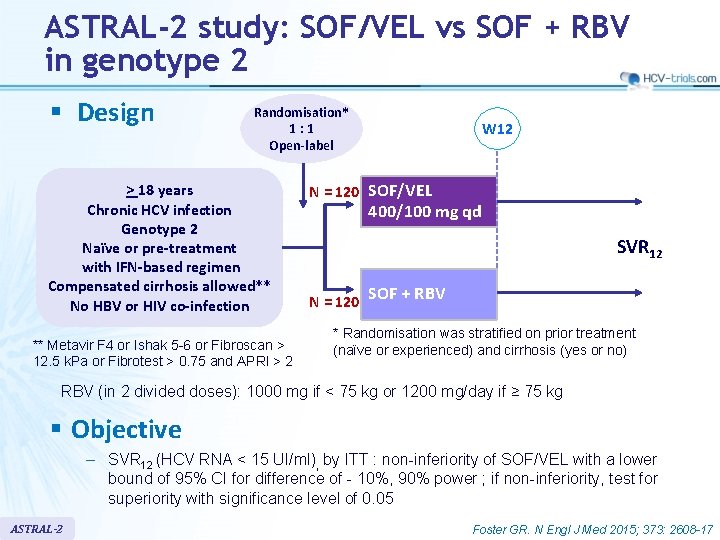
ASTRAL-2 study: SOF/VEL vs SOF + RBV in genotype 2 § Design Randomisation* 1: 1 Open-label > 18 years Chronic HCV infection Genotype 2 Naïve or pre-treatment with IFN-based regimen Compensated cirrhosis allowed** No HBV or HIV co-infection ** Metavir F 4 or Ishak 5 -6 or Fibroscan > 12. 5 k. Pa or Fibrotest > 0. 75 and APRI > 2 W 12 N = 120 SOF/VEL 400/100 mg qd SVR 12 N = 120 SOF + RBV * Randomisation was stratified on prior treatment (naïve or experienced) and cirrhosis (yes or no) RBV (in 2 divided doses): 1000 mg if < 75 kg or 1200 mg/day if ≥ 75 kg § Objective – SVR 12 (HCV RNA < 15 UI/ml), by ITT : non-inferiority of SOF/VEL with a lower bound of 95% CI for difference of - 10%, 90% power ; if non-inferiority, test for superiority with significance level of 0. 05 ASTRAL-2 Foster GR. N Engl J Med 2015; 373: 2608 -17
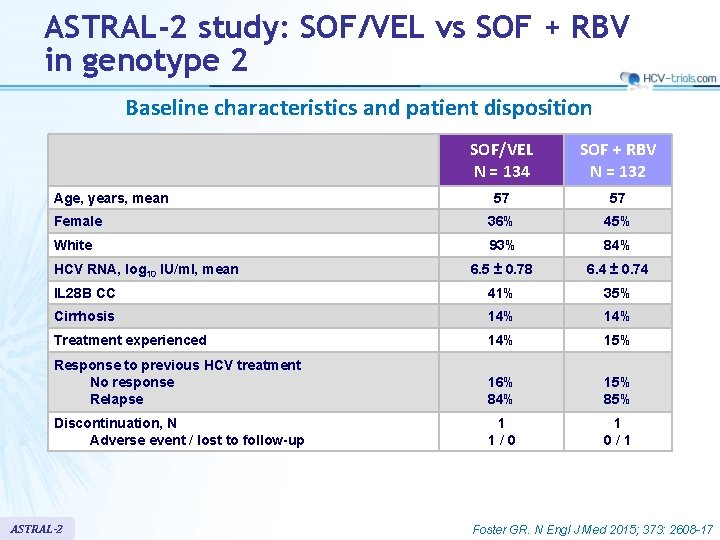
ASTRAL-2 study: SOF/VEL vs SOF + RBV in genotype 2 Baseline characteristics and patient disposition SOF/VEL N = 134 SOF + RBV N = 132 57 57 Female 36% 45% White 93% 84% 6. 5 ± 0. 78 6. 4 ± 0. 74 IL 28 B CC 41% 35% Cirrhosis 14% Treatment experienced 14% 15% Response to previous HCV treatment No response Relapse 16% 84% 15% 85% Discontinuation, N Adverse event / lost to follow-up 1 1 / 0 1 0 / 1 Age, years, mean HCV RNA, log 10 IU/ml, mean ASTRAL-2 Foster GR. N Engl J Med 2015; 373: 2608 -17
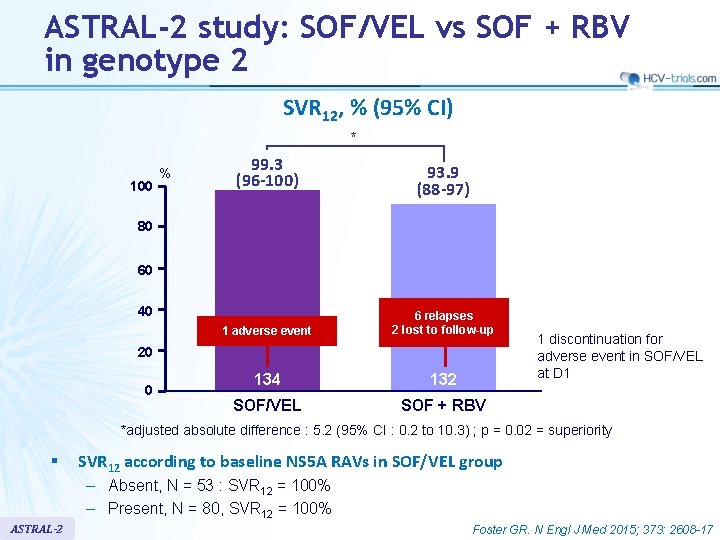
ASTRAL-2 study: SOF/VEL vs SOF + RBV in genotype 2 SVR 12, % (95% CI) * 100 % 99. 3 (96 -100) 93. 9 (88 -97) 80 60 40 1 adverse event 6 relapses 2 lost to follow-up 134 132 SOF/VEL SOF + RBV 20 0 1 discontinuation for adverse event in SOF/VEL at D 1 *adjusted absolute difference : 5. 2 (95% CI : 0. 2 to 10. 3) ; p = 0. 02 = superiority § SVR 12 according to baseline NS 5 A RAVs in SOF/VEL group – Absent, N = 53 : SVR 12 = 100% – Present, N = 80, SVR 12 = 100% ASTRAL-2 Foster GR. N Engl J Med 2015; 373: 2608 -17
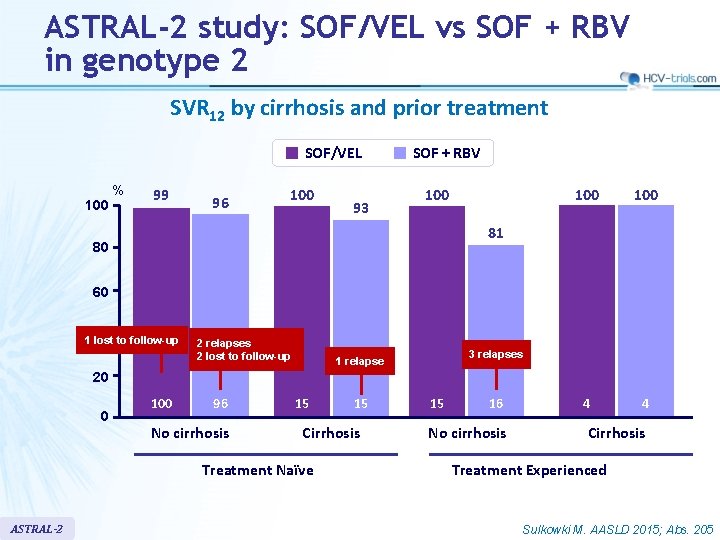
ASTRAL-2 study: SOF/VEL vs SOF + RBV in genotype 2 SVR 12 by cirrhosis and prior treatment SOF/VEL 100 % 99 96 100 93 SOF + RBV 100 100 4 4 81 80 60 40 1 lost to follow-up 2 relapses 2 lost to follow-up 3 relapses 1 relapse 20 0 100 96 No cirrhosis 15 Cirrhosis Treatment Naïve ASTRAL-2 15 15 16 No cirrhosis Cirrhosis Treatment Experienced Sulkowki M. AASLD 2015; Abs. 205
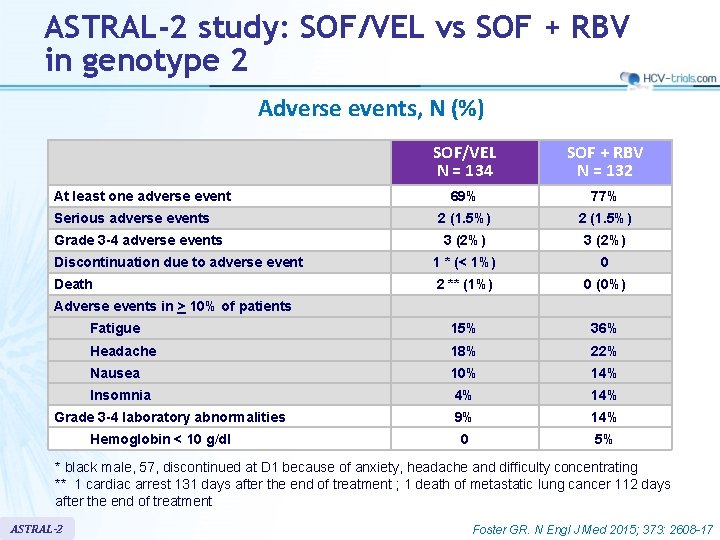
ASTRAL-2 study: SOF/VEL vs SOF + RBV in genotype 2 Adverse events, N (%) SOF/VEL N = 134 SOF + RBV N = 132 69% 77% 2 (1. 5%) 3 (2%) Discontinuation due to adverse event 1 * (< 1%) 0 Death 2 ** (1%) 0 (0%) Fatigue 15% 36% Headache 18% 22% Nausea 10% 14% Insomnia 4% 14% 9% 14% 0 5% At least one adverse event Serious adverse events Grade 3 -4 adverse events Adverse events in > 10% of patients Grade 3 -4 laboratory abnormalities Hemoglobin < 10 g/dl * black male, 57, discontinued at D 1 because of anxiety, headache and difficulty concentrating ** 1 cardiac arrest 131 days after the end of treatment ; 1 death of metastatic lung cancer 112 days after the end of treatment ASTRAL-2 Foster GR. N Engl J Med 2015; 373: 2608 -17

ASTRAL-2 study: SOF/VEL vs SOF + RBV in genotype 2 § Summary – Treatment with SOF/VEL for 12 weeks resulted in a 99% SVR 12 rate in patients with HCV genotype 2 infection – SOF/VEL for 12 weeks was statistically superior to SOF + RBV for 12 weeks (p = 0. 02) – No patients who received SOF/VEL experienced virologic failure in a population that included patients with cirrhosis and previous treatment failure • Limitation : small number of black patients – Among patients with HCV genotype 2 who received SOF/VEL , the presence of baseline NS 5 A or NS 5 B RAVs was not associated with virologic failure – SOF/VEL was well tolerated and, compared with SOF + RBV, lacked toxicities commonly associated with RBV – SOF/VEL for 12 weeks provides a single tablet, once daily, highly effective, RBV-free treatment for patients with HCV genotype 2 infection ASTRAL-2
- Slides: 6